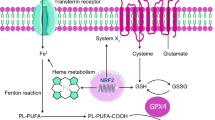
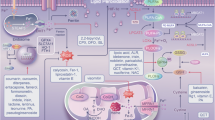
Abstract
In recent years, it has been identified that excess iron contributes to the development of various pathologies and their complications. Kidney diseases do not escape the toxic effects of iron, and ferroptosis is identified as a pathophysiological mechanism that could be a therapeutic target to avoid damage or progression of kidney disease. Ferroptosis is cell death associated with iron-dependent oxidative stress. To study the effects of iron overload (IOL) in the kidney, numerous animal models have been developed. The methodological differences between these models should reflect the IOL-generating mechanisms associated with human IOL diseases. A careful choice of animal model should be considered for translational purposes.
Similar content being viewed by others
References
Adams P, Barton J, Guo H, Alter D, Speechley M (2015) Serum ferritin is a biomarker for liver mortality in the hemochromatosis and iron overload screening study. Ann Hepatol 14:348–353. https://doi.org/10.1016/S1665-2681(19)31274-8
Aggarwal SK, Kinter M, Fitzgerald RL, Herold DA (1994) Mass spectrometry of trace elements in biological samples. Crit Rev Clin Lab Sci 31:35–87. https://doi.org/10.3109/10408369409084673
Aldwairi M, Yassin M (2020) Iatrogenic iron overload in an end stage renal disease patient. Case Rep Oncol 13:760–763. https://doi.org/10.1159/000507979
Asano T, Komatsu M, Yamaguchi-Iwai Y, Ishikawa F, Mizushima N, Iwai K (2011) Distinct mechanisms of ferritin delivery to lysosomes in iron-depleted and iron-replete cells. Mol Cell Biol 31:2040–2052. https://doi.org/10.1128/MCB.01437-10
Barroso C, Carvalho P, Nunes M, Gonçalves JFM, Rodrigues PNS, Neves JV (2021) The era of antimicrobial peptides: use of hepcidins to prevent or treat bacterial infections and iron disorders. Front Immunol 12:754437. https://doi.org/10.3389/fimmu.2021.754437
Boyd E, Shanas M (1963) The acute oral toxicity of reduced iron. Can Med Assoc J 89:171–175 (PMID: 14014682)
Boyd M, Shanas M (1967) Studies on the low toxicity of reduced iron, B.P. 1932. Can Med Assoc J 96:1141–1146 (PMID: 6021563)
Breborowicz A, Polubinska A, Górna K, Breborowicz M, Oreopoulos D (2006) Iron sucrose induced morphological and functional changes in the rat kidney. Transl Res 148:257–262. https://doi.org/10.1016/j.trsl.2006.04.008
Budak H, Kocpinar E, Gonul N, Ceylan H, Erol H, Erdogan O (2014) Stimulation of gene expression and activity of antioxidant related enzyme in Sprague Dawley rat kidney induced by long-term iron toxicity. Comp Biochem Physiol C Toxicol Pharmacol 166:44–50. https://doi.org/10.1016/j.cbpc.2014.07.002
Cavdar Z, Oktan M, Ural C, Calisir M, Kocak A, Heybeli C, Yildiz S, Arici A, Ellidokuz H, Celik A, Yilmaz O, Sarioglu S, Cavdar C (2020) Renoprotective effects of alpha lipoic acid on iron overload-induced kidney injury in rats by suppressing Nadph oxidase 4 and p38 MAPK signaling. Biol Trace Elem Res 193:483–493. https://doi.org/10.1007/s12011-019-01733-3
Cavdar Z, Oktan M, Ural C, Kocak A, Calisir M, Heybeli C, Yildiz S, Ozbal S, Arslan S, Ergur B, Yilmaz O, Cavdar C (2021) Alpha lipoic acid attenuates iron induced oxidative acute kidney injury in rats. Biotech Histochem 96:409–417. https://doi.org/10.1080/10520295.2020.1812001
Conrad M, Friedmann-Angeli JP (2015) Glutathione peroxidase 4 (Gpx4) and ferroptosis: what’s so special about it? Mol Cell Oncol 30:e995047. https://doi.org/10.4161/23723556.2014.99504
Desmots F, Rissel M, Pigeon C, Loyer P, Loréal O, Guillouzo A (2002) Differential effects of iron overload on GST isoform expression in mouse liver and kidney and correlation between GSTA4 induction and overproduction of free radicles. Free Radic Biol Med 32:93–101. https://doi.org/10.1016/S0891-5849(01)00767-5
Dimitriou E, Kairis M, Sarafidou J, Michelakakis H (2000) Iron overload and kidney lysosomes. Biochim Biophys Acta 1501:138–148. https://doi.org/10.1016/s0925-4439(00)00019-3
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS, Morrison B 3rd, Stockwell BR (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149:1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Doll S, Freitas FP, Shah R, Aldrovandi M, da Silva MC, Ingold I, Goya Grocin A, Xavier da Silva TN, Panzilius E, Scheel CH, Mourão A, Buday K, Sato M, Wanninger J, Vignane T, Mohana V, Rehberg M, Flatley A, Schepers A, Kurz A, White D, Sauer M, Sattler M, Tate EW, Schmitz W, Schulze A, O’Donnell V, Proneth B, Popowicz GM, Pratt DA, Angeli JPF, Conrad M (2019) FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575:693–698. https://doi.org/10.1038/s41586-019-1707-0
Enerbäck L, Lundin P (1965) The effect of overloading with complex iron preparations on the morphology and function of the rat kidney. Toxicol Appl Pharmacol 7:525–534. https://doi.org/10.1016/0041-008x(65)90037-2
Erichsen K, Ulvik RJ, Grimstad T, Berstad A, Berge RK, Hausken T (2005) Effects of ferrous sulphate and non-ionic iron-polymaltose complex on markers of oxidative tissue damage in patients with inflammatory bowel disease. Aliment Pharmacol Ther 22:831–838. https://doi.org/10.1111/j.1365-2036.2005.02652.x
Eybl V, Kotyzová D, Cerná P, Koutensky J (2008) Effect of melatonin, curcumin, quercetin, and resveratrol on acute ferric nitrilotriacetate (Fe-NTA)-induced renal oxidative damage in rats. Hum Exp Toxicol 27:347–353. https://doi.org/10.1177/0960327108094508
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch A, Eggenhofer E, Basavarajappa D, Rådmark O, Kobayashi S, Seibt T, Beck H, Neff F, Esposito I, Wanke R, Förster H, Yefremova O, Heinrichmeyer M, Bornkamm GW, Geissler EK, Thomas SB, Stockwell BR, O’Donnell VB, Kagan VE, Schick JA, Conrad M (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16:1180–1191. https://doi.org/10.1038/ncb3064
Galleano M, Farre S, Turrens J, Puntarulo S (1994) Resistance of rat kidney mitochondrial membranes to oxidation induced by acute iron overload. Toxicology 88:141–149. https://doi.org/10.1016/0300-483X(94)90116-3
Gao W, Li X, Gao Z, Li H (2014) Iron increases diabetes-induced kidney injury and oxidative stress in rats. Biol Trace Elem Res 160:368–375. https://doi.org/10.1007/s12011-014-0021-9
Geisser P, Burckhardt S (2011) The pharmacokinetics and pharmacodynamics of iron preparations. Pharmaceutics 3:12–33. https://doi.org/10.3390/pharmaceutics3010012
Gholampour F, Saki N (2019) Hepatic and renal protective effects of quercetin in ferrous sulfate induced toxicity. Gen Physiol Biophys 38:27–38. https://doi.org/10.4149/gpb_2018038
Guan S, Ma J, Zhang Y, Gao Y, Zhang Y, Zhang X, Wang N, Xie Y, Wang J, Zhang J, Chu L (2013) Danshen (Salvia miltiorrhiza) injection suppresses kidney injury induced by iron overload in mice. PLoS One 8:e74318. https://doi.org/10.1371/journal.pone.0074318
Ikeda K, Sun F, Tanaka K, Tokumaru S, Kojo S (1998) Increase of lipid hydroperoxides in the rat liver and kidney after administering ferric nitrilotriacetate. Biosci Biotechnol Biochem 62:1438–1439. https://doi.org/10.1271/bbb.62.1438
Iqbal M, Athar M (1998) Attenuation of iron-nitrilotriacetate (Fe-NTA)-mediated renal oxidative stress, toxicity and hyperproliferative response by the prophylactic treatment of rats with garlic oil. Food Chem Toxicol 36:485–495. https://doi.org/10.1016/S0278-6915(98)00008-8
Ishizaka N, Saito K, Noiri E, Sata M, Mori I, Ohno M, Nagai R (2004) Iron dextran causes renal iron deposition but not renal dysfunction in angiotensin II-treated and untreated rats. Nephron Physiol 98:107–113. https://doi.org/10.1159/000081559
Italia K, Colah R, Ghosh K (2015) Experimental animal model to study iron overload and iron chelation and review of other such models. Blood Cells Mol Dis 55:194–199. https://doi.org/10.1016/j.bcmd.2015.06.003
Johnson A, Becker K, Zager R (2010) Parenteral iron formulations differentially affect MCP-1, HO-1, and NGAL gene expression and renal responses to injury. Am J Physiol Renal Physiol 299:426–435. https://doi.org/10.1152/ajprenal.00248.2010
Kadkhodaee M, Gol A (2004) The role of nitric oxide in iron-induced rat renal injury. Hum Exp Toxicol 23:533–536. https://doi.org/10.1191/0960327104ht485oa
Kang H, Han M, Xue J, Baek Y, Chang J, Hu S, Nam H, Jo M, El Fakhri G, Hutchens M, Choi H, Kim J (2019) Renal clearable nanochelators for iron overload therapy. Nat Commun 10:5134. https://doi.org/10.1038/s41467-019-13143-z
Kawabata T, Ogino T, Mori M, Awai M (1992) Effects of nicotinamide and its isomers on iron-induced renal damage. Acta Pathol Jpn 42:469–475. https://doi.org/10.1111/j.1440-1827.1992.tb03091.x
Kozlov A, Bini A, Gallesi D, Giovannini F, Iannone A, Masini A, Meletti E, Tomasi A (1996) ‘Free’ iron, as detected by electron paramagnetic resonance spectroscopy, increases unequally in different tissues during dietary iron overload in the rat. Biometals 9:98–103. https://doi.org/10.1007/BF00188097
Kudo H, Suzuki S, Watanabe A, Kikuchi H, Sassa S, Sakamoto S (2008) Effects of colloidal iron overload on renal and hepatic siderosis and the femur in male rats. Toxicology 246:143–147. https://doi.org/10.1016/j.tox.2008.01.004
Kulaksiz H, Theilig F, Bachmann S, Gehrke SG, Rost D, Janetzko A, Cetin Y, Stremmel W (2005) The iron-regulatory peptide hormone hepcidin: expression and cellular localization in the mammalian kidney. J Endocrinol 184:361–370. https://doi.org/10.1677/joe.1.05729
Li Y, Chien S, Chu P, Liu M (2012) Prophylactic and therapeutic effects of a subcutaneous injection of sesame oil against iron-induced acute renal injury in mice. JPEN J Parenter Enteral Nutr 36:344–348. https://doi.org/10.1177/0148607111415530
Lim C, Vaziri N (2004a) Iron and oxidative stress in renal insufficiency. Am J Nephrol 24:569–575. https://doi.org/10.1159/000082201
Lim C, Vaziri N (2004b) The effects of iron dextran on the oxidative stress in cardiovascular tissues of rats with chronic renal failure. Kidney Int 65:1802–1809. https://doi.org/10.1111/j.1523-1755.2004.00580.x
Linder-Horowitz M, Ruettinger R, Munro H (1970) Iron induction of electrophoretically different ferritins in rat liver, heart and kidney. Biochim Biophys Acta 200:442–448. https://doi.org/10.1016/0005-2795(70)90100-5
Mai TT, Hamaï A, Hienzsch A, Cañeque T, Müller S, Wicinski J, Cabaud O, Leroy C, David A, Acevedo V, Ryo A, Ginestier C, Birnbaum D, Charafe-Jauffret E, Codogno P, Mehrpour M, Rodriguez R (2017) Salinomycin kills cancer stem cells by sequestering iron in lysosomes. Nat Chem 9:1025–1033. https://doi.org/10.1038/nchem.2778
Márquez-Ibarra A, Huerta M, Villalpando-Hernández S, Ríos-Silva M, Díaz-Reval M, Cruzblanca H, Mancilla E, Trujillo X (2016) The effects of dietary iron and capsaicin on hemoglobin, blood glucose, insulin tolerance, cholesterol, and triglycerides, in healthy and diabetic wistar rats. PLoS One 11:e0152625. https://doi.org/10.1371/journal.pone.0152625
Martin-Sanchez D, Fontecha-Barriuso M, Martinez-Moreno JM, Ramos AM, Sanchez-Niño MD, Guerrero-Hue M, Moreno JA, Ortiz A, Sanz AB (2020) Ferroptosis and kidney disease. Nefrologia 40:384–394. https://doi.org/10.1016/j.nefro.2020.03.005
Mohammad G, Matakidou A, Robbins P, Lakhal-Littleton S (2021) The kidney hepcidin/ferroportin axis controls iron reabsorption and determines the magnitude of kidney and systemic iron overload. Kidney Int 100:559–569. https://doi.org/10.1016/j.kint.2021.04.034
Moulouel B, Houamel D, Delaby C, Tchernitchko D, Vaulont S, Letteron P, Thibaudeau O, Puy H, Gouya L, Beaumont C, Karim Z (2013) Hepcidin regulates intrarenal iron handling at the distal nephron. Kidney Int 84:756–766. https://doi.org/10.1038/ki.2013.142
Müller S, Sindikubwabo F, Cañeque T, Lafon A, Versini A, Lombard B, Loew D, Wu TD, Ginestier C, Charafe-Jauffret E, Durand A, Vallot C, Baulande S, Servant N, Rodriguez R (2020) CD44 regulates epigenetic plasticity by mediating iron endocytosis. Nat Chem 12:929–938. https://doi.org/10.1038/s41557-020-0513-5
Nankivell B, Chen J, Boadle R, Harris D (1994) The role of tubular iron accumulation in the remnant kidney. J Am Soc 4:1598–1607. https://doi.org/10.1681/ASN.V481598
Navidi-Shishaone M, Mohhebi S, Nematbakhsh M, Roozbehani S, Talebi A, Pezeshki Z, Eshraghi-Jazi F, Mazaheri S, Shirdavani S, Gharagozloo M, Moaeidi B (2014) Co-administration of silymarin and deferoxamine against kidney, liver and heart iron deposition in male iron overload rat model. Int J Prev Med 5:110–116
NDA 21–135, Venofer (Iron sucrose injection) https://www.accessdata.fda.gov/drugsatfda_docs/label/2000/21135lbl.pdf., 2000 (Accessed 20 October 2022)
Nematbakhsh M, Pezeshki Z, Moaeidi B, Eshraghi-Jazi F, Talebi A, Nasri H, Baradaran S, Gharagozloo M, Safari T, Haghighi M (2013) Protective role of silymarin and deferoxamine against iron dextran-induced renal iron deposition in male rats. Int J Prev Med 4:286–292
Nemeth E, Ganz T (2021) Hepcidin-Ferroportin Interaction Controls Systemic Iron Homeostasis. Int J Mol Sci 22:6493. https://doi.org/10.3390/ijms22126493
Oliveira F, Rocha S, Fernandes R (2014) Iron metabolism: from health to disease. J Clin Lab Anal 28:210–218. https://doi.org/10.1002/jcla.21668
Ortega L, Ladero J, Carreras M, Alvarez T, Taxonera C, Oliván M, Sanz-Esponera J, Díaz-Rubio M (2005) A computer-assisted morphometric quantitative analysis of iron overload in liver biopsies. A comparison with histological and biochemical methods. Pathol Res Pract 201:673–677. https://doi.org/10.1016/j.prp.2005.07.002
Paller M, Hedlund B (1988) Role of iron in postischemic renal injury in the rat. Kidney Int 34:474–480. https://doi.org/10.1038/ki.1988.205
Pan S, Qian Z, Cui S, Zhao D, Lan W, Wang X, Chen X (2020) Local hepcidin increased intracellular iron overload via the degradation of ferroportin in the kidney. Biochem Biophys Res Commun 522:322–327. https://doi.org/10.1016/j.bbrc.2019.11.066
Pereira M, Pereira M, Sousa J (1998) Adsorptive stripping measurements of iron accumulation in mice kidney using microelectrodes and histological features. J Trace Elem Med Biol 12:50–55. https://doi.org/10.1016/S0946-672X(98)80021-7
Pigeon C, Ilyin G, Courselaud B, Leroyer P, Turlin B, Brissot P, Loréal O (2001) A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload. J Biol Chem 276:7811–7819. https://doi.org/10.1074/jbc.M008923200
Plays M, Müller S, Rodriguez R (2021) Chemistry and biology of ferritin. Metallomics 13:021. https://doi.org/10.1093/mtomcs/mfab021
Ramanathan G, Olynyk J, Ferrari P (2017) Diagnosing and preventing iron overload. Hemodial Int 21:58–67. https://doi.org/10.1111/hdi.12555
Refaat B, Abdelghany A, BaSalamah M, El-Boshy M, Ahmad J, Idris S (2018) Acute and chronic iron overloading differentially modulates the expression of cellular iron-homeostatic molecules in normal rat kidney. J Histochem Cytochem 66:825–839. https://doi.org/10.1369/0022155418782696
Saito K, Ishizaka N, Aizawa T, Sata M, Iso-O N, Noiri E, Ohno M, Nagai R (2004) Role of aberrant iron homeostasis in the upregulation of transforming growth factor-beta1 in the kidney of angiotensin II-induced hypertensive rats. Hypertens Res 27:599–607. https://doi.org/10.1291/hypres.27.599
Sajjad S, Garcia M, Malik A, Van Thiel D (2008) Hepatic iron quantitation and its relationship with disease measures and histologically assessed iron content. Dig Dis Sci 53:1390–1394. https://doi.org/10.1007/s10620-007-9993-z
Schümann K, Solomons NW, Romero-Abal ME, Orozco M, Weiss G, Marx J (2012) Oral administration of ferrous sulfate, but not of iron polymaltose or sodium iron ethylenediaminetetraacetic acid (NaFeEDTA), results in a substantial increase of non-transferrin-bound iron in healthy iron-adequate men. Food Nutr Bull 33:128–136. https://doi.org/10.1177/156482651203300206
Scindia Y, Leeds J, Swaminathan S (2019) Iron homeostasis in healthy kidney and its role in acute kidney injury. Semin Nephrol 39:76–84. https://doi.org/10.1016/j.semnephrol.2018.10.006
Seiler A, Schneider M, Förster H, Roth S, Wirth EK, Culmsee C, Plesnila N, Kremmer E, Rådmark O, Wurst W, Bornkamm GW, Schweizer U, Conrad M (2008) Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death. Cell Metab 8:237–248. https://doi.org/10.1016/j.cmet.2008.07.005
Somers G (1947) Relative oral toxicity of some therapeutic iron preparations. Br Med J 2:201–203. https://doi.org/10.1136/bmj.2.4518.201
Suzuki N, Matsuo-Tezuka Y, Sasaki Y, Sato K, Miyauchi K, Kato K, Saito S, Shimonaka Y, Hirata M, Yamamoto M (2018) Iron attenuates erythropoietin production by decreasing hypoxia-inducible transcription factor 2α concentrations in renal interstitial fibroblasts. Kidney Int 94:900–911. https://doi.org/10.1016/j.kint.2018.06.028
Toblli J, Cao G, Oliveri L, Angerosa M (2009) Differences between original intravenous iron sucrose and iron sucrose similar preparations. Arzneimittelforschung 59:176–190. https://doi.org/10.1055/s-0031-1296383
Toblli J, Cao G, Olivieri L, Angerosa M (2010) Comparison of the renal, cardiovascular and hepatic toxicity data of original intravenous iron compounds. Nephrol Dial Transplant 25:3631–3640. https://doi.org/10.1093/ndt/gfq260
Wang X, Zheng X, Zhang J, Zhao S, Wang Z, Wang F, Shang W, Barasch J, Qiu A (2018) Physiological functions of ferroportin in the regulation of renal iron recycling and ischemic acute kidney injury. Am J Physiol Renal Physiol 315:1042–1057. https://doi.org/10.1152/ajprenal.00072.2018
Wareing M, Ferguson C, Delannoy M, Cox A, McMahon R, Green R, Riccardi D, Smith C (2003) Altered dietary iron intake is a strong modulator of renal DMT1 expression. Am J Physiol Renal Physiol 285:1050–1059. https://doi.org/10.1152/ajprenal.00064.2003
Weiss A, Spektor L, Cohen L, Lifshitz L, Magid Gold I, ZhanG D, Truman-Rosentsvit M, Leichtmann-Bardoogo Y, Nyska A, Addadi S, Rouault T, Meyron-Holtz E (2018) Orchestrated regulation of iron trafficking proteins in the kidney during iron overload facilitates systemic iron retention. PLoS One 13:e0204471. https://doi.org/10.1371/journal.pone.0204471
Wu Z, Paller M (1994) Iron loading enhances susceptibility to renal ischemia in rats. Ren Fail 16:471–480. https://doi.org/10.3109/08860229409045078
Xu L, Liu Y, Chen X, Zhong H, Wang Y (2023) Ferroptosis in life: to be or not to be. Biomed Pharmacother 159:114241. https://doi.org/10.1016/j.biopha.2023.114241
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji AF, Clish CB, Brown LM, Girotti AW, Cornish VW, Schreiber SL, Stockwell BR (2014) Regulation of ferroptotic cancer cell death by GPX4. Cell 16(156):317–331. https://doi.org/10.1016/j.cell.2013.12.010
Yeung C, Zhu L, Glahn R, Miller D (2004) Iron absorption from NaFeEDTA is downregulated in iron-loaded rats. J Nutr 134:2270–2274. https://doi.org/10.1093/jn/134.9.2270
Yun S, Chu D, He X, Zhang W, Feng C (2020) Protective effects of grape seed proanthocyanidins against iron overload-induced renal oxidative damage in rats. J Trace Elem Med Biol 57:126407. https://doi.org/10.1016/j.jtemb.2019.126407
Zager R, Johnson A, Frostad K (2016) Combined iron sucrose and protoporphyrin treatment protects against ischemic and toxin-mediated acute renal failure. Kidney Int 90:67–76. https://doi.org/10.1016/j.kint.2016.01.022
Zainal T, Weindruch R, Szweda L, Oberley T (1999) Localization of 4-hydroxy-2-nonenal-modified proteins in kidney following iron overload. Free Radic Biol Med 26:1181–1193. https://doi.org/10.1016/S0891-5849(98)00312-8
Zhang Y, Gao Z, Liu J, Xu Z (2011) Protective effects of baicalin and quercetin on an iron-overloaded mouse: comparison of liver, kidney and heart tissues. Nat Prod Res 25:1150–1160. https://doi.org/10.1080/14786419.2010.495070
Funding
This research has not received specific aid from public sector agencies, the commercial sector, or non-profit entities.
Author information
Authors and Affiliations
Contributions
MRS and YC: designed the study, AGOM, OMC, EMZ, JABB and II: carried out the data collection. MRS, YC, MH and XT: analyzed the data. AGOM, OMC, EMZ, JABB and II: made the tables. MRS, YC, MH, and XT: drafted and revised the article. All authors are agreeing with and have approved this version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ríos-Silva, M., Cárdenas, Y., Ortega-Macías, A.G. et al. Animal models of kidney iron overload and ferroptosis: a review of the literature. Biometals 36, 1173–1187 (2023). https://doi.org/10.1007/s10534-023-00518-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-023-00518-5