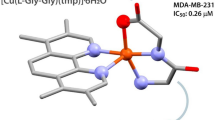
Abstract
A group of bidentate nitrogen and sulfur donor pyrazole derivative ligands abbreviated as Na[RNCS(Pz)], Na[RNCS(PzMe2)], Na[RNCS(PzMe3)], Na[RNCS(PzPhMe)], Na[RNCS(PzPh2)], where (R = Et, Ph), and their Cu (II) complexes were synthesized and characterized by spectroscopic and physicochemical methods. The crystal structure of [Cu(PhNCSPzMe3)2] was determined by X-ray crystallography analysis and the results described a distorted square planar coordination geometry for this complex. Also, the cyclic voltammetry investigations indicated that the synthesized copper complex is an electrochemically active species. Moreover, the cytotoxic activity of all of the twenty synthesized compounds was evaluated using MTT assay against the MCF-7 (human breast carcinoma) cell lines, in vitro. Cu (II) complexes indicate significant cytotoxicity against the MCF-7 cell lines as compared with the free ligands. The docking studies showed that the copper complexes have better interactions with EGFR and CDK2 proteins, compared to the free ligands, and most of the studied compounds have a higher value of binding energy relative to the studied controls. The results of QSAR analysis suggest that dipole moment is in direct correlation with the obtained IC50 values, and it strongly impact the anticancer effects generated by the compounds. Our findings suggest that the developed copper complexes can be good candidates for further evaluations as chemotherapeutic agents in the treatment of cancer.
Graphical abstract










Similar content being viewed by others
References
Aghdam EM, Barzegar A, Hejazi MS (2014) Evolutionary origin and conserved structural building blocks of riboswitches and ribosomal RNAs: riboswitches as probable target sites for aminoglycosides interaction. Adv Pharm Bull 4:225
Aguilar-Martinez M, Cuevas G, Jimenez-Estrada M, González I, Lotina-Hennsen B, Macias-Ruvalcaba N (1999) An experimental and theoretical study of the substituent effects on the redox properties of 2-[(R-phenyl) amine]-1, 4-naphthalenediones in acetonitrile. J Org Chem 64:3684–3694
Anjum R et al (2019) Synthesis, characterization, and in vitro anticancer activity of copper and zinc bis (thiosemicarbazone) complexes. Inorg Chem 58:13709–13723
Ansari A, Ali A, Asif M (2017) Biologically active pyrazole derivatives. New J Chem 41:16–41
Bahron H, Khaidir SS, Tajuddin AM, Ramasamy K, Yamin BM (2019) Synthesis, characterization and anticancer activity of mono-and dinuclear Ni (II) and Co (II) complexes of a Schiff base derived from o-vanillin. Polyhedron 161:84–92
Banerjee S, Chakravarty AR (2015) Metal complexes of curcumin for cellular imaging, targeting, and photoinduced anticancer activity. Acc Chem Res 48:2075–2083
Baumann M, Krause M, Dikomey E, Dittmann K, Dörr W, Kasten-Pisula U, Rodemann HP (2007) EGFR-targeted anti-cancer drugs in radiotherapy: preclinical evaluation of mechanisms. Radiother Oncol 83:238–248
Bayarı S, Ataç A, Yurdakul Ş (2003) Coordination behaviour of nicotinamide: an infrared spectroscopic study. J Mol Struct 655:163–170
Bhosale M, Yadav A, Magdum C, Mohite S (2020) Microwave assisted synthesis, molecular docking studies and anticancer screening of some 1, 3, 4-thiadiazole derivatives. J Univ Shanghai Sci Technol 22:520–534
Blumberger J, Bernasconi L, Tavernelli I, Vuilleumier R, Sprik M (2004) Electronic structure and solvation of copper and silver ions: a theoretical picture of a model aqueous redox reaction. J Am Chem Soc 126:3928–3938
Bruijnincx PC, Sadler PJ (2008) New trends for metal complexes with anticancer activity. Curr Opin Chem Biol 12:197–206
Cava C, Castiglioni I (2020) Integration of molecular docking and in vitro studies: a powerful approach for drug discovery in breast cancer. Appl Sci 10:6981
Chakraborty A et al (2015) Synthesis, X-ray structure and in vitro cytotoxicity studies of Cu (I/II) complexes of thiosemicarbazone: special emphasis on their interactions with DNA. Dalton Trans 44:6140–6157
Cheung-Ong K, Giaever G, Nislow C (2013) DNA-damaging agents in cancer chemotherapy: serendipity and chemical biology. Chem Biol 20:648–659. https://doi.org/10.1016/j.chembiol.2013.04.007
Cronin J et al (2011) Epidermal growth factor receptor (EGFR) is overexpressed in high-grade dysplasia and adenocarcinoma of the esophagus and may represent a biomarker of histological progression in Barrett’s esophagus (BE). Am Coll Gastroenterol 106:46–56
Dantu AS, Shankarguru P, Ramya DD, Vedha HB (2012) Evaluation of in vitro anticancer activity of hydroalcoholic extract of Tabernaemontana divaricata. Asian J Pharm Clin Res 5:59–61
Datar PA, Khedkar SA, Malde AK, Coutinho EC (2006) Comparative residue interaction analysis (CoRIA): a 3D-QSAR approach to explore the binding contributions of active site residues with ligands. J Comput-Aided Mol Des 20:343–360
Dhar S, Senapati D, Das PK, Chattopadhyay P, Nethaji M, Chakravarty AR (2003) Ternary copper complexes for photocleavage of DNA by red light: direct evidence for sulfur-to-copper charge transfer and d−d band involvement. J Am Chem Soc 125:12118–12124
Feazell RP, Nakayama-Ratchford N, Dai H, Lippard SJ (2007) Soluble single-walled carbon nanotubes as longboat delivery systems for platinum (IV) anticancer drug design. J Am Chem Soc 129:8438–8439
Frezza M, Hindo S, Chen D, Davenport A, Schmitt S, Tomco D, Dou QP (2010) Novel metals and metal complexes as platforms for cancer therapy. Curr Pharm Des 16:1813–1825. https://doi.org/10.2174/138161210791209009
Galluzzi L et al (2012) Molecular mechanisms of cisplatin resistance. Oncogene 31:1869–1883. https://doi.org/10.1038/onc.2011.384
Gao Y, Wang H, Zhong A, Yu T (2020) Expression and prognosis of CyclinA and CDK2 in patients with advanced cervical cancer after chemotherapy. Cell Mol Biol 66:85–91
Geersing A, Ségaud N, van der Wijst MG, Rots MG, Roelfes G (2018) Importance of metal-ion exchange for the biological activity of coordination complexes of the biomimetic ligand N4Py. Inorg Chem 57:7748–7756
Ghasemian M, Mahdavi M, Zare P, Feizi MAH (2015) Spiroquinazolinone-induced cytotoxicity and apoptosis in K562 human leukemia cells: alteration in expression levels of Bcl-2 and Bax. J Toxicol Sci 40:115–126
Ghorbanpour M, Soltani B, Ziegler CJ, Jamshidi-Ghaleh K (2021) Novel pyrazolate-bridged binuclear Ni (II), Cu (II) and Zn (II) complexes: synthesis, X-ray crystal structure and nonlinear optical studies. Inorg Chim Acta 514:119957
Ghorbanpour M, Soltani B, Molavi O, Aghdam EM (2022) In vitro and in silico studies on the anticancer and antimicrobial activity of Cu (II), Ni (II) and Co (II) complexes with bis (pyrazolyl) borate derivative ligand. Pharm Sci. https://doi.org/10.34172/PS.2022.3
Goodman V, Brewer G, Merajver S (2004) Copper deficiency as an anti-cancer strategy. Endocr Relat Cancer 11:255–263
Herrick RS, Wrona I, McMicken N, Jones G, Ziegler CJ, Shaw J (2004) Preparation and characterization of rhenium (I) compounds with amino ester derivatized diimine ligands. Investigations of luminescence. Crystal structures of Re (CO) 3Cl (pyca-β-Ala-OEt) and Re (CO) 3Cl (pyca-l-Asp (OMe)-OMe). J Organomet Chem 689:4848–4855
Hossaini Sadr M, Soltani B, Jalili A, Nejadghafar F, Kia R, Engle JT, Ziegler CJ (2012) Synthesis and characterization of copper (II) complexes incorporating pyrazolyl-derived N, S-donor bidentate ligands. Trans Metal Chem 37:611–617
Jafari-Gharabaghlou D et al (2018) Combination of metformin and phenformin synergistically inhibits proliferation and hTERT expression in human breast cancer cells Iranian. J Basic Med Sci 21:1167
Konkankit CC, Marker SC, Knopf KM, Wilson JJ (2018) Anticancer activity of complexes of the third row transition metals, rhenium, osmium, and iridium. Dalton Trans 47:9934–9974
Kumar KA, Jayaroopa P (2013) Pyrazoles: synthetic strategies and their pharmaceutical applications-an overview. Int J Pharm Tech Res 5:1473–1486
Kumar V, Kaur K, Gupta GK, Sharma AK (2013) Pyrazole containing natural products: synthetic preview and biological significance. Eur J Med Chem 69:735–753
Li Y, Yang Z, Zhou M, Li Y (2017) Synthesis and crystal structure of new monometallic Ni (II) and Co (II) complexes with an asymmetrical aroylhydrazone: effects of the complexes on DNA/protein binding property, molecular docking, and in vitro anticancer activity. RSC Adv 7:49404–49422
Lv P-C, Li H-Q, Sun J, Zhou Y, Zhu H-L (2010) Synthesis and biological evaluation of pyrazole derivatives containing thiourea skeleton as anticancer agents. Bioorg Med Chem 18:4606–4614
Ma Z, Molavi O, Haddadi A, Lai R, Gossage RA, Lavasanifar A (2008) Resveratrol analog trans 3, 4, 5, 4′-tetramethoxystilbene (DMU-212) mediates anti-tumor effects via mechanism different from that of resveratrol. Cancer Chemother Pharmacol 63:27–35
Ma DL, He HZ, Leung KH, Chan DSH, Leung CH (2013) Bioactive luminescent transition-metal complexes for biomedical applications. Angew Chem Int Ed 52:7666–7682
Machado I et al (2014) New metal complexes of NNO tridentate ligands: effect of metal center and co-ligand on biological activity. Inorg Chim Acta 420:39–46
Malik MA, Dar OA, Gull P, Wani MY, Hashmi AA (2018) Heterocyclic Schiff base transition metal complexes in antimicrobial and anticancer chemotherapy. MedChemComm 9:409–436
Marzano C, Pellei M, Tisato F, Santini C (2009) Copper complexes as anticancer agents. Anti-Cancer Agents Med Chem 9:185–211
Mohammadian J, Sabzichi M, Molavi O, Shanehbandi D, Samadi N (2016) Combined treatment with stattic and docetaxel alters the Bax/Bcl-2 gene expression ratio in human prostate cancer cells. Asian Pac J Cancer Prev 17:5031
Monteiro DC, Phillips RM, Crossley BD, Fielden J, Willans CE (2012) Enhanced cytotoxicity of silver complexes bearing bidentate N-heterocyclic carbene ligands. Dalton Trans 41:3720–3725
Mota A, Lotfi AS, Barzin J, Hatam M, Adibi B, Khalaj Z, Massumi M (2014) Human bone marrow mesenchymal stem cell behaviors on PCL/gelatin nanofibrous scaffolds modified with a collagen IV-derived RGD-containing peptide. Cell J 16:1
Muhammad N, Guo Z (2014) Metal-based anticancer chemotherapeutic agents. Curr Opin Chem Biol 19:144–153
Naim MJ, Alam O, Farah Nawaz M, Alam J, Alam P (2016) Current status of pyrazole and its biological activities. J Pharm Bioallied Sci 8:2
Norouzi S, Farahani M, Ebrahimi SN (2021) The integration of pharmacophore-based 3D-QSAR modeling and virtual screening in identification of natural product inhibitors against SARS-COV-2. Pharm Sci 27:S94–S108. https://doi.org/10.34172/PS.2020.98
Palanimuthu D, Shinde SV, Somasundaram K, Samuelson AG (2013) In vitro and in vivo anticancer activity of copper bis (thiosemicarbazone) complexes. J Med Chem 56:722–734
Pandey RK, Kumbhar BV, Srivastava S, Malik R, Sundar S, Kunwar A, Prajapati VK (2017) Febrifugine analogues as Leishmania donovani trypanothione reductase inhibitors: binding energy analysis assisted by molecular docking, ADMET and molecular dynamics simulation. J Biomol Struct Dyn 35:141–158
Phosrithong N, Ungwitayatorn J (2010) Molecular docking study on anticancer activity of plant-derived natural products. Med Chem Res 19:817–835
Rajendiran V et al (2007) Mixed-ligand copper (II)-phenolate complexes: effect of coligand on enhanced DNA and protein binding, DNA cleavage, and anticancer activity. Inorg Chem 46:8208–8221
Rao VS, Srinivas K (2011) Modern drug discovery process: an in silico approach. J Bioinf Seq Anal 3:89–94
Rasulev BF, Abdullaev ND, Syrov VN, Leszczynski J (2005) A quantitative structure-activity relationship (QSAR) study of the antioxidant activity of flavonoids QSAR & combinatorial. Science 24:1056–1065
Ray S, Mohan R, Singh JK, Samantaray MK, Shaikh MM, Panda D, Ghosh P (2007) Anticancer and antimicrobial metallopharmaceutical agents based on palladium, gold, and silver N-heterocyclic carbene complexes. J Am Chem Soc 129:15042–15053
Sadr MH, Jalili AR, Razmi H, Ng SW (2005) A new family of pyrazolyl-based anionic bidentate ligands and crystal structure of bis (N-phenyl-2-pyrazolyl-1-carboximidothioato) copper (II). J Organomet Chem 690:2128–2132
Sadr MH, Soltani B, Jalili A, Nejadghafar F, Kia R, Engle JT, Ziegler CJ (2012) Synthesis and characterization of copper (II) complexes incorporating pyrazolyl-derived N, S-donor bidentate ligands. Transit Met Chem 37:611–617
Said MA, Al-unizi A, Al-Mamary M, Alzahrani S, Lentz D (2020) Easy coordinate geometry indexes, τ4 and τ5 and HSA study for unsymmetrical Pd (II), Fe (II), Zn (II), Mn (II), Cu (II) and VO (IV) complexes of a tetradentate ligand: synthesis, characterization, properties, and antioxidant activities. Inorg Chim Acta 505:119434
Santini C, Pellei M, Gandin V, Porchia M, Tisato F, Marzano C (2014) Advances in copper complexes as anticancer agents. Chem Rev 114:815–862
Soenmez M, Bayram MR, Çelebı M (2009) Synthesis and characterization of heterocyclic Schiff base and its complexes with Cu (II), Ni (II), Co (II), Zn (II), and Cd (II). J Coord Chem 62:2728–2735
Soltani B, Ghorbanpour M, Ziegler CJ, Ebadi-Nahari M, Mohammad-Rezaei R (2020) Nickel (II) and cobalt (II) complexes with bidentate nitrogen-sulfur donor pyrazole derivative ligands: syntheses, characterization, X-ray structure, electrochemical studies, and antibacterial activity. Polyhedron 180:114423
Sorrell TN, Jameson DL (1983) Synthesis, structure, and reactivity of monomeric two-coordinate copper (I) complexes. J Am Chem Soc 105:6013–6018
Sreejaya S, Santhy K (2013) Cytotoxic properties of Acorus calamus in MCF-7 breast cancer cells. Int J Curr Res Acad Rev 1:106–111
Sun J et al (2013) Synthesis, biological evaluation and molecular docking studies of pyrazole derivatives coupling with a thiourea moiety as novel CDKs inhibitors. Eur J Med Chem 68:1–9
Tabbì G, Giuffrida A, Bonomo RP (2013) Determination of formal redox potentials in aqueous solution of copper (II) complexes with ligands having nitrogen and oxygen donor atoms and comparison with their EPR and UV–Vis spectral features. J Inorg Biochem 128:137–145
Tadesse S, Caldon EC, Tilley W, Wang S (2018) Cyclin-dependent kinase 2 inhibitors in cancer therapy: an update. J Med Chem 62:4233–4251
Tadesse S, Anshabo AT, Portman N, Lim E, Tilley W, Caldon CE, Wang S (2020) Targeting CDK2 in cancer: challenges and opportunities for therapy. Drug Discov Today 25:406–413
Tahghighi A (2014) Importance of metal complexes for development of potential leishmanicidal agents. J Organomet Chem 770:51–60
Tardito S, Marchio L (2009) Copper compounds in anticancer strategies. Curr Med Chem 16:1325–1348
Tigreros A, Portilla J (2020) Recent progress in chemosensors based on pyrazole derivatives. RSC Adv 10:19693–19712
Tisato F, Marzano C, Porchia M, Pellei M, Santini C (2010) Copper in diseases and treatments, and copper-based anticancer strategies. Med Res Rev 30:708–749
Veerasamy R, Rajak H, Jain A, Sivadasan S, Varghese CP, Agrawal RK (2011) Validation of QSAR models-strategies and importance. Int J Drug Des Discov 3:511–519
Verma J, Khedkar VM, Coutinho EC (2010) 3D-QSAR in drug design-a review. Curr Top Med Chem 10:95–115
Walker TL et al (2015) Blue copper protein analogue: synthesis and characterization of copper complexes of the N 2 S 2 macrocycle 1, 8-dithia-4, 11-diazacyclotetradecane. Dalton Trans 44:20200–20206
Wu EL, Han K, Zhang JZ (2008) Selectivity of neutral/weakly basic P1 group inhibitors of thrombin and trypsin by a molecular dynamics study. Chem A Eur J 14:8704–8714
Yang L, Powell DR, Houser RP (2007) Structural variation in copper (I) complexes with pyridylmethylamide ligands: structural analysis with a new four-coordinate geometry index, τ 4. Dalton Trans. https://doi.org/10.1039/B617136B
Yang X, Liu S, Liu G, Zhong H (2017) A DFT study on the structure–reactivity relationship of aliphatic oxime derivatives as copper chelating agents and malachite flotation collectors. J Ind Eng Chem 46:404–415
Zhu H, Zhang S, Li M, Shao Y, Zhu Z (2010) Electrochemical sensor for melamine based on its copper complex. Chem Commun 46:2259–2261
Acknowledgements
We would like to thank University of Azarbaijan Shahid Madani for supporting this work. Single crystal X-ray diffraction experiments were performed at the University of Akron. The optimization of the structure of the compounds were performed in the Molecular Simulation laboratory of Azarbaijan Shahid Madani University. We thank them for their helpful collaboration.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix: supplementary data
Appendix: supplementary data
CCDC 2059101 contain the supplementary crystallographic data for 18. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghorbanpour, M., Soltani, B., Mota, A. et al. Copper (II) complexes with N, S donor pyrazole-based ligands as anticancer agents. Biometals 35, 1095–1111 (2022). https://doi.org/10.1007/s10534-022-00426-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-022-00426-0