Abstract
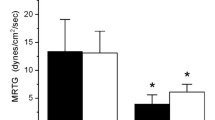
While the administration of antivenom to treat hemotoxic snake bite injury remains the gold standard of therapy, we have demonstrated that modifying human fibrinogen with iron and carbon monoxide renders it resistant to fibrinogenolytic snake venom enzymes. In order to translate these findings into a possible biometal-based therapy complementary to antivenom administration, a preclinical model that possesses fibrinogen that closely mimics the human molecule in response to iron and carbon monoxide needed to be identified. The goal of this investigation was to determine if a swine model could serve in this capacity by assessing the thrombelastographic response of porcine plasma to iron and carbon monoxide exposure, without or with further exposure to the fibrinogenolytic venom of the viper Crotalus atrox. Using plasma obtained from eight swine, it was determined that their plasma responded to iron and carbon monoxide in a manner similar to that of human plasma by displaying enhanced coagulation kinetics. However, in sharp contrast to the response seen with human plasma, only iron significantly protected porcine plasma coagulation kinetics from C. atrox venom degradation. Therefore the pig is an animal beyond humans that could derive benefit from the biometal-focused therapy of iron infusion to protect against venom mediated compromise of coagulation. Thus, future investigation to assess the effects of iron administration to attenuate the effects of fibrinogenolytic envenomation with a pig model is justified.



Similar content being viewed by others
References
Doering CJ, Wagg CR, Caulkett NA, McAllister RK, Brookfield CE, Paterson JM, Warren AL, Smith BL, Boysen SR (2014) Comparison of arterial and venous whole blood clot initiation, formation, and strength by thromboelastography in anesthetized swine. Blood Coagul Fibrinolysis 25:20–24
Hack JB, Orogbemi B, Deguzman JM, Brewer KL, Meggs WJ, O’Rourke D (2010) A localizing circumferential compression device delayed death after artificial eastern diamondback rattlesnake envenomation to the torso of an animal model in a pilot study. J Med Toxicol 6:207–211
Kessler U, Grau T, Gronchi F, Berger S, Brandt S, Bracht H, Marcucci C, Zachariou Z, Jakob SM (2011) Comparison of porcine and human coagulation by thrombelastometry. Thromb Res 128:477–482
Meggs WJ, Courtney C, O’Rourke D, Brewer KL (2010) Pilot studies of pressure-immobilization bandages for rattlesnake envenomations. Clin Toxicol 48:61–63
Nielsen VG, Boyer LV (2016) Iron and carbon monoxide attenuate degradation of plasmatic coagulation by Crotalus atrox venom. Blood Coagul Fibrinolysis 27:506–510
Nielsen VG, Jacobsen WK (2016) Iron modulates the alpha chain of fibrinogen. Biometals 29:235–238
Nielsen VG, Pretorius E (2014) Iron and carbon monoxide enhance coagulation and attenuate fibrinolysis by different mechanisms. Blood Coagul Fibrinolysis 25:695–702
Nielsen VG, Cerruti MA, Valencia OM, Amos Q (2016a) Decreased snake venom metalloproteinase effects via inhibition of enzyme and modification of fibrinogen. Biometals 29:913–919
Nielsen VG, Redford DT, Boyle PK (2016b) Effect of iron and carbon monoxide on fibrinogenase-like degradation of plasmatic coagulation by venoms of six Agkistrodon species. Basic Clin Pharmacol Toxicol 118:390–395
Nielsen VG, Sánchez EE, Redford DT (2017) Characterization of the rabbit as an in vitro and in vivo model to assess the effects of fibrinogenolytic activity of snake venom on coagulation. Basic Clin Pharmacol Toxicol. doi:10.1111/bcpt.12848
Zentai C, Solomon C, van der Meijden PE, Spronk HM, Schnabel J, Rossaint R, Grottke O (2016) Effects of fibrinogen concentrate on thrombin generation, thromboelastometry parameters, and laboratory coagulation testing in a 24-hour porcine trauma model. Clin Appl Thromb Hemost 22:749–759
Grant support
This investigation was supported by the CSU Clinical Pathology Research and Development Funds and the Department of Anesthesiology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Olver, C.S., Nielsen, V.G. Iron protects porcine plasma coagulation kinetics from degradation by Crotalus atrox venom. Biometals 30, 677–683 (2017). https://doi.org/10.1007/s10534-017-0035-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-017-0035-9