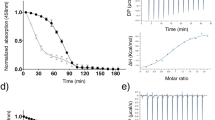
Abstract
Human mitochondrial protein mitoNEET is a novel target of type II diabetes drug pioglitazone, and contains a redox active [2Fe–2S] cluster that is hosted by a unique ligand arrangement of three cysteine and one histidine residues. Here we report that zinc ion can compete for the [2Fe–2S] cluster binding site in human mitoNEET and potentially modulate the physiological function of mitoNEET. When recombinant mitoNEET is expressed in Escherichia coli cells grown in M9 minimal media, purified mitoNEET contains very little or no iron–sulfur clusters. Addition of exogenous iron or zinc ion in the media produces mitoNEET bound with a [2Fe–2S] cluster or zinc, respectively. Mutations of the amino acid residues that hosting the [2Fe–2S] cluster in mitoNEET diminish the zinc binding activity, indicating that zinc ion and the [2Fe–2S] cluster may share the same binding site in mitoNEET. Finally, excess zinc ion effectively inhibits the [2Fe–2S] cluster assembly in mitoNEET in E. coli cells, suggesting that zinc ion may impede the function of mitoNEET by blocking the [2Fe–2S] cluster assembly in the protein.





Similar content being viewed by others
References
Agar JN, Krebs C, Frazzon J, Huynh BH, Dean DR, Johnson MK (2000) IscU as a scaffold for iron–sulfur cluster biosynthesis: sequential assembly of [2Fe–2S] and [4Fe–4S] clusters in IscU. Biochemistry 39:7856–7862
Arif W, Xu S, Isailovic D, Geldenhuys WJ, Carroll RT, Funk MO (2011) Complexes of the outer mitochondrial membrane protein mitoNEET with resveratrol-3-sulfate. Biochemistry 50:5806–5811
Bae JB, Park JH, Hahn MY, Kim MS, Roe JH (2004) Redox-dependent changes in RsrA, an anti-sigma factor in Streptomyces coelicolor: zinc release and disulfide bond formation. J Mol Biol 335:425–435
Bak DW, Zuris JA, Paddock ML, Jennings PA, Elliott SJ (2009) Redox characterization of the FeS protein mitoNEET and impact of thiazolidinedione drug binding. Biochemistry 48:10193–10195
Baxter EL, Jennings PA, Onuchic JN (2011) Interdomain communication revealed in the diabetes drug target mitoNEET. Proc Natl Acad Sci USA 108:5266–5271
Beyersmann D, Haase H (2001) Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals 14:331–341
Colca JR, McDonald WG, Waldon DJ, Leone JW, Lull JM, Bannow CA, Lund ET, Mathews WR (2004) Identification of a novel mitochondrial protein (“mitoNEET”) cross-linked specifically by a thiazolidinedione photoprobe. Am J Physiol Endocrinol Metab 286:E252–E260
Cowart RE, Singleton FL, Hind JS (1993) A comparison of bathophenanthrolinedisulfonic acid and ferrozine as chelators of iron(II) in reduction reactions. Anal Biochem 211:151–155
Cuajungco MP, Lees GJ (1997) Zinc metabolism in the brain: relevance to human neurodegenerative disorders. Neurobiol Dis 4:137–169
Duce JA, Tsatsanis A, Cater MA, James SA, Robb E, Wikhe K, Leong SL, Perez K, Johanssen T, Greenough MA, Cho HH, Galatis D, Moir RD, Masters CL, McLean C, Tanzi RE, Cappai R, Barnham KJ, Ciccotosto GD, Rogersh JT, Bush AI (2010) Iron-export ferroxidase activity of beta-amyloid precursor protein is inhibited by zinc in Alzheimer’s disease. Cell 142:857–867
Hou X, Liu R, Ross S, Smart EJ, Zhu H, Gong W (2007) Crystallographic studies of human mitoNEET. J Biol Chem 282:33242–33246
Klinge S, Nunez-Ramirez R, Llorca O, Pellegrini L (2009) 3D Architecture of DNA Pol alpha reveals the functional core of multi-subunit replicative polymerases. EMBO J 28:1978–1987
Koh J-Y, Suh SW, Gwag BJ, He YY, Hsu CY, Choi DW (1996) The role of zinc in selective neuronal death after transient global cerebral ischemia. Science 272:1013–1016
Lemire J, Mailloux R, Appanna VD (2008) Zinc toxicity alters mitochondrial metabolism and leads to decreased ATP production in hepatocytes. J Appl Toxicol 28:175–182
Lill R (2009) Function and biogenesis of iron–sulphur proteins. Nature 460:831–838
Lin J, Zhou T, Ye K, Wang J (2007) Crystal structure of human mitoNEET reveals distinct groups of iron–sulfur proteins. Proc Natl Acad Sci USA 104:14640–14645
Lin J, Zhang L, Lai S, Ye K (2011) Structure and molecular evolution of CDGSH iron-sulfur domains. PLoS ONE 6:e24790
Liu J, Oganesyan N, Shin DH, Jancarik J, Yokota H, Kim R, Kim SH (2005) Structural characterization of an iron–sulfur cluster assembly protein IscU in a zinc-bound form. Proteins 59:875–881
Netz DJA, Stith CM, Stümpfig M, Köpf G, Vogel D, Genau HM, Stodola JL, Lill R, Burgers PMJ, Pierik AJ (2012) Eukaryotic DNA polymerases require an iron–sulfur cluster for the formation of active complexes. Nat Chem Biol 8:125–132
Paddock ML, Wiley SE, Axelrod HL, Cohen AE, Roy M, Abresch EC, Capraro D, Murphy AN, Nechushtai R, Dixon JE, Jennings PA (2007) MitoNEET is a uniquely folded 2Fe 2S outer mitochondrial membrane protein stabilized by pioglitazone. Proc Natl Acad Sci USA 104:14342–14347
Pagani MA, Casamayor A, Serrano R, Atrian S, Arino J (2007) Disruption of iron homeostasis in Saccharomyces cerevisiae by high zinc levels: a genome-wide study. Mol Microbiol 65:521–537
Ramelot TA, Cort JR, Goldsmith-Fischman S, Kornhaber GJ, Xiao R, Shastry R, Acton TB, Honig B, Montelione GT, Kennedy MA (2004) Solution NMR structure of the iron–sulfur cluster assembly protein U (IscU) with zinc bound at the active site. J Mol Biol 344:567–583
Raulfs EC, O’Carroll IP, Dos Santos PC, Unciuleac MC, Dean DR (2008) In vivo iron–sulfur cluster formation. Proc Natl Acad Sci USA 105:8591–8596
Rouault TA, Tong WH (2008) Iron–sulfur cluster biogenesis and human disease. Trends Genet 24:398–407
Sharpley MS, Hirst J (2006) The inhibition of mitochondrial complex I (NADH:ubiquinone oxidoreductase) by Zn2+. J Biol Chem 281:34803–34809
Tirrell TF, Paddock ML, Conlan AR, Smoll EJ Jr, Nechushtai R, Jennings PA, Kim JE (2009) Resonance Raman studies of the (His)(Cys)(3) 2Fe–2S cluster of mitoNEET: comparison to the (Cys)(4) mutant and implications of the effects of pH on the labile metal center. Biochemistry 48:4747–4752
Wang W, Huang H, Tan G, Si F, Liu M, Landry AP, Lu J, Ding H (2010) In vivo evidence for the iron binding activity of an iron–sulfur cluster assembly protein IscA in Escherichia coli. Biochem J 432:429–436
Wiley SE, Murphy AN, Ross SA, van der Geer P, Dixon JE (2007a) MitoNEET is an iron-containing outer mitochondrial membrane protein that regulates oxidative capacity. Proc Natl Acad Sci USA 104:5318–5323
Wiley SE, Paddock ML, Abresch EC, Gross L, van der Geer P, Nechushtai R, Murphy AN, Jennings PA, Dixon JE (2007b) The outer mitochondrial membrane protein mitoNEET contains a novel redox-active 2Fe–2S cluster. J Biol Chem 282:23745–23749
Yang J, Bitoun JP, Ding H (2006) Interplay of IscA and IscU in biogenesis of iron–sulfur clusters. J Biol Chem 281:27956–27963
Zheng L, Cash VL, Flint DH, Dean DR (1998) Assembly of iron–sulfur clusters. Identification of an iscSUA-hscBA-fdx gene cluster from Azotobacter vinelandii. J Biol Chem 273:13264–13272
Zhou T, Lin J, Feng Y, Wang J (2010) Binding of reduced nicotinamide adenine dinucleotide phosphate destabilizes the iron–sulfur clusters of human mitoNEET. Biochemistry 49:9604–9612
Zuris JA, Harir Y, Conlan AR, Shvartsman M, Michaeli D, Tamir S, Paddock ML, Onuchic JN, Mittler R, Cabantchik ZI, Jennings PA, Nechushtai R (2011) Facile transfer of [2Fe–2S] clusters from the diabetes drug target mitoNEET to an apo-acceptor protein. Proc Natl Acad Sci USA 108:13047–13052
Zuris JA, Ali SS, Yeh H, Nguyen TA, Nechushtai R, Paddock ML, Jennings PA (2012) NADPH inhibits [2Fe–2S] cluster protein transfer from diabetes drug target mitoNEET to an apo-acceptor protein. J Biol Chem 287:11649–11655
Acknowledgments
Research reported in this publication was partially supported by the National Cancer Institute of the National Institutes of Health under award number R01CA107494, and by the Chinese Natural Science Foundation grants (31228006 and 31200587).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, G., Landry, A.P., Dai, R. et al. Competition of zinc ion for the [2Fe–2S] cluster binding site in the diabetes drug target protein mitoNEET. Biometals 25, 1177–1184 (2012). https://doi.org/10.1007/s10534-012-9580-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-012-9580-4