Abstract
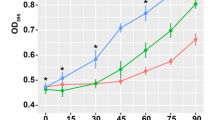
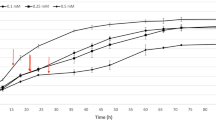
In this work we investigated the adaptive response of E. faecalis to Cu and the role of CopY, a Cu-dependent repressor, in the regulation of Cu metabolism. In doing so, we examined the whole-genome transcriptional response of E. faecalis wild-type (WT) and a ΔcopY strain exposed to non-toxic Cu excess. The results indicated that after Cu exposure, most of the genes that displayed a significant change in their expression levels in the WT strain (135 of the 145 up-regulated genes and 115 of the 142 down-regulated genes) were also differentially expressed in the E. faecalis ΔcopY strain. This extensive overlap in the transcriptional response, suggested that additional transcription factors mediate the response of E. faecalis to Cu. As a first step to analyze this possibility, we selected among the up-regulated genes five genes encoding putative transcriptional regulators and determined their expression levels at different times after Cu exposure. The temporal expression of these regulators was different from that of copY, which reached its maximum at the earliest time measured. Nevertheless, transcription elongation factor GreA, and members of Rrf2, Cro/CI and SorC/DeoR transcription factor families were induced shortly after Cu exposure, suggesting that these proteins are able to complement the role of CopY in the regulatory network activated by Cu. To our knowledge, this is the first report on the global transcriptional response to Cu in a member of this taxonomic group.



Similar content being viewed by others
References
Arredondo M, Núñez MT (2005) Iron and copper metabolism. Mol Aspects Med 26:313–327
Bissig KD, Wunderli-Ye H, Duda PW, Solioz M (2001) Structure-function analysis of purified Enterococcus hirae CopB copper ATPase: effect of Menkes/Wilson disease mutation homologues. Biochem J 357:217–223
Chen Y, Staddon JH, Dunny GM (2007) Specificity determinants of conjugative DNA processing in the Enterococcus faecalis plasmid pCF10 and the Lactococcus lactis plasmid pRS01. Mol Microbiol 63:1549–1564
de Freitas J, Wintz H, Kim JH, Poynton H, Fox T, Vulpe C (2003) Yeast, a model organism for iron and copper metabolism studies. Biometals 16:185–197
del Pozo T, Cambiazo V, González M (2010) Gene expression profiling analysis of copper homeostasis in Arabidopsis thaliana. Biochem Biophys Res Commun. doi:10.1016/j.bbrc.2010.01.111
Frangipani E, Slaveykova VI, Reimmann C, Haas D (2008) Adaptation of aerobically growing Pseudomonas aeruginosa to copper starvation. J Bacteriol 190:6706–6717
Gonzalez M, Tapia L, Alvarado M, Tornero JD, Fernández R (1999) Intracellular determination of elements in mammalian cultured cells by total reflection X-ray fluorescence spectrometry. J Anal At Spectrom 14:885–888
Keller G, Bird A, Winge DR (2005) Independent metalloregulation of Ace1 and Mac1 in Saccharomyces cerevisiae. Eukaryot Cell 4:1863–1871
Kershaw CJ, Brown NL, Constantinidou C, Patel MD, Hobman JL (2005) The expression profile of Escherichia coli K-12 in response to minimal, optimal and excess copper concentrations. Microbiology 151:1187–1198
Kristich CJ, Chandler JR, Dunny GM (2007) Development of a host-genotype-independent counter selectable marker and a high-frequency conjugative delivery system and their use in genetic analysis of Enterococcus faecalis. Plasmid 57:131–144
Leenhouts K, Buist G, Bolhuis A, ten Berge A, Kiel J, Mierau I, Dabrowska M, Venema G, Kok J (1996) A general system for generating unlabelled gene replacements in bacterial chromosomes. Mol Gen Genet 253:217–224
Linder MC, Hazegh-Azam M (1996) Copper biochemistry and molecular biology. Am J Clin Nutr 63:797S–811S
Lu ZH, Dameron CT, Solioz M (2003) The Enterococcus hirae paradigm of copper homeostasis: copper chaperone turnover, interactions, and transactions. Biometals 16:137–143
Macomber L, Imlay JA (2009) The iron-sulfur clusters of dehydratases are primary intracellular targets of copper toxicity. Proc Natl Acad Sci USA 106:8344–8349
O’Halloran TV (1993) Transition metals in control of gene expression. Science 261:715–725
Odermatt A, Solioz M (1995) Two trans-acting metalloregulatory proteins controlling expression of the copper-ATPases of Enterococcus hirae. J Biol Chem 270:4349–4354
Pecou E, Maass A, Remenik D, Briche J, Gonzalez M (2006) A mathematical model for copper homeostasis in Enterococcus hirae. Math Biosci 20:222–239
Portmann R, Poulsen KR, Wimmer R, Solioz M (2006) CopY-like copper inducible repressors are putative ‘winged helix’ proteins. Biometals 19:61–70
Rae TD, Schmidt PJ, Pufahl RA, Culotta VC, O’Halloran TV (1999) Undetectable intracellular free copper: the requirement of a copper chaperone for superoxide dismutase. Science 28:805–808
Reyes A, Leiva A, Cambiazo V, Mendez MA, Gonzalez M (2006) Cop-like operon: structure and organization in species of the Lactobacillales order. Biol Res 39:87–93
Ruiz-Garbajosa P, Bonten MJ, Robinson DA, Top J, Nallapareddy SR, Torres C, Coque TM, Cantón R, Baquero F, Murray BE, del Campo R, Willems RJ (2006) Multilocus sequence typing scheme for Enterococcus faecalis reveals hospital-adapted genetic complexes in a background of high rates of recombination. J Clin Microbiol 44:2220–2228
Rutherford JC, Bird AJ (2004) Metal-responsive transcription factors that regulate iron, zinc, and copper homeostasis in eukaryotic cells. Eukaryot Cell 3:1–13
Solioz M, Stoyanov JV (2003) Copper homeostasis in Enterococcus hirae. FEMS Microbiol Rev 27:183–195
Teitzel GM, Geddie A, De Long SK, Kirisits MJ, Whiteley M, Parsek MR (2006) Survival and growth in the presence of elevated copper: transcriptional profiling of copper-stressed Pseudomonas aeruginosa. J Bacteriol 188:7242–7256
Urbanski NK, Beresewicz A (2000) Generation of *OH initiated by interaction of Fe2+ and Cu+ with dioxygen; comparison with the Fenton chemistry. Acta Biochim Pol 47:951–962
Ward SK, Hoye EA, Talaat AM (2008) The global responses of Mycobacterium tuberculosis to physiological levels of copper. J Bacteriol 190:2939–2946
Yamamoto K, Ishiham A (2005) Transcriptional response of Escherichia coli to external copper. Mol Microbiol 56:215–227
Acknowledgements
This work was supported by grants Basal-CMM, Fondecyt 1071083 and 1090211 and Fondef D04I1257. AR-J was recipients of an ‘A. Steckel’ fellowship from INTA and MAL is a recipient of doctoral fellowship from Conicyt. The authors thank Arely González and Andres Aravena for COGs assignments and manual revision of V583 genome.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Reyes-Jara, A., Latorre, M., López, G. et al. Genome-wide transcriptome analysis of the adaptive response of Enterococcus faecalis to copper exposure. Biometals 23, 1105–1112 (2010). https://doi.org/10.1007/s10534-010-9356-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-010-9356-7