Abstract
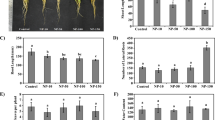
Characterized by a photo—catalysis property, nano-anatase TiO2 is closely related to photosynthesis of spinach. It could not only improve light absorbance, transformation from light energy to electron energy and active chemical energy, but also promote the activity of Rubiso activase of spinach. However, the relation between the activity of Rubiso activase and the growth of spinach promoted by nano-anatase TiO2 treatment remains largely unclear. In this study, we find that the amount and the activity of Rubiso activase are obviously increased by nano-anatase TiO2 treatment, which led to the great promotion of Rubsico carboxylation and the high rate of photosynthesis, thus improving of spinach growth. The significant enhancement of Rubiso activase activity of nano-anatase TiO2 treated spinach is also accompanied by conformational changes as determined by spectroscopic analysis. But bulk TiO2 effect is not as significant as nano-anatase TiO2, as the grain size of nano-anatase TiO2 (5 nm) is much smaller than that of bulk TiO2, which entered spinach cell more easily.





Similar content being viewed by others
References
Arnon DI (1949) Copper enzymes in isolated chloroplasts: polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15
Chua NH, Schmidt GW (1979) Transport of proteins into mitochondria and chloroplasts. J Cell Biol 81:461–483
Crabtree RH (1998) A new type of hydrogen bond. Science 282:2000–2001
Gao FQ, Hong FS, Liu C, Zheng L, Su MY (2006) Mechanism of Nano-anatase TiO2 on promoting photosynthetic carbon reaction of spinach: Inducing complex of Rubisco–Rubisco activase. Biol Trace Element Res 111:286–301
Han Y, Chen G, Wang Z (2000) The progress of studies on Rubisco activase. Chin Bull Botany (in Chinese) 17(4):306–311
Hong FS, Yang P, Gao FQ, Liu C, Zheng L, Yang F, Zhou J, (2005a) Effect of nano-TiO2 on spectral characterization of photosystem II particles from spinach. Chem Res Chin Univ 21(2):196–200
Hong FS, Yang F, Ma ZN, Zhou J, Liu C, Wu C, Yang P (2005b) Influences of nano-TiO2 on the chloroplast ageing of spinach under light. Biol Trace Element Res 104(3):249–260
Hong FS, Zhou J, Liu C, Yang F, Wu C, Zheng L, Yang P (2005c) Effect of Nano-TiO2 on Photochemical Reaction of Chloroplasts of Spinach. Biol. Trace Element Res 105:1–11 [1] Wang LJ, Guo ZM, Li TJ, Li M (1999) Biomineralized nanostructured materials and plant silicon nutrition. Prog Chem 11:119–128 (in Chinese)
Hong FS, Liu C, Zheng L, Wang XF, Wu K, Song WP, Lv SP, Tao Y, Zhao GW, (2005d) Formation of Complexes of Rubisco–Rubisco activase from La3+, Ce3+ treatment spinach. Sci China Ser B Chem 48(1):67–74
Jimenez ESD, Medrano L, Martinez-Barajas E (1995) Rubisco activae, a possible new member of the molecular chaperonefamily. Biochemistry 34:2826–2831
Keegstra K, Olsen LJ, Theg. SM (1989) Chloroplastic precursors and their transport across the envelope membranes. Annu Rev Plant Physiol Plant Mol Biol 40:471–501
Kim K, Portis AR (2005) Temperature dependence of photosynthesis in Arabidopsis plants with modifications in Rubisco activase and membrane fluidity. Plant Cell Physiol 46(3):522–530
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature 277:680–685
Lan Y, Meott KA (1991) Determination of apparent Km values for ribulose 1,5-bisphosphate carboxylase/oxygnase (Rubisco) activase using the spectrophotometric assay of Rubisco activity. Plant Physiol 95:604–609
Liu C, Hong FS, Wu K, Ma HB, Zhang XG, Hong CJ, Wu C, Gao FQ, Yang F, Zheng L, Wang XF, Liu T, Xie YN, Xu JH, Li ZR (2006) Effect of Nd3+ ion on carboxylation activity of ribulose-1,5-bisphosphate carboxylase/oxygenase of spinach. Biochem Biophys Res Commun 342(1):36–43
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Perczel A, Park K, Fasman GD (1992) Analysis of the circular dichroism spectrum of proteins using the convex constraint algorithm:A practical guide. Anal Biochem 203:83–89
Portis A Jr (1995) The regulation of Rubisco by Rubisco activase. J Expt Bot 46:1285–1291
Portis A Jr, Salvucci ME, Ogren WL (1986) Activation of ribulosebisphosphate carboxylase.oxygenase at physiological CO2 and ribulosebisphosphate concentrations by Rubisco activase. Plant Physiol 82:967–971
Robert L, Houtza, Portis A Jr (2003) The life of ribulose-1,5-bisphosphate carboxylase/oxygenase—posttranslational facts and mysteries. Arch Biochem Biophys 414:150–158
Robinson SP, Streusand VJ, Chatifield JM, Portis A Jr (1988) Purification and assay of Rubisco activase from leaves. Plant Physiol 88:1008–1014
Salvucci ME, Klein RR (1994) Site-Directed mutagenase of reaction lysyl residue(Lys-247) of Rubisco activase. Arck Biochem Biophys 314:178–185
Sambrook EF, Fritsch T (eds) (1989) Molecular cloning. Cold Spring Laboratory Press, NewYork
Spreitzer RJ (1999) Questions about the complexity of chloroplast ribulose-1,5-bisphosphate carboxylase/oxygenase. Photosynth Res 60:29–42
Sugiyama T, Nakayama N, Ogawa M, Akazawa T, Oda T (1968) Structure and function of chloroplast proteins: Effect of ρ-Chloromercuribenzoate treatment on the ribulose-1,5-bisphosphate carboxylase/oxygenase activity of Spinach leaf fraction protein. Arch Biochem Biophys 125:98–106
Tang RH, Jia JW, Li LR (1997) Purification and characterization of Rubisco activase from tobacco. Acta Phytophysiol Sin (in Chinese) 23:89–95
To KY, Cheng MC, Chen LFO, Chen SCG (1996) Introduction and expression of foreign DNA in isolated spinach chloroplasts by electroporation. Plant J 10:737–743
Wang WG, Li LR (1980) A simplified purification method of RuBP carboxylase from spinach leaves. Acta Phytophysiologia Sin (in Chinese) 40(3):256–262
Wang LJ, Guo ZM, Li TJ, Li M (1999) Biomineralized nanostructured materials and plant silicon nutrition. Prog Chem (in Chinese) 11:119–128
Yang P, Lu C, Hua N, Du Y (2002) Titanim dioxide nanoparticles co-doped with Fe3+ and Eu3+ ions for photocatalysis. Mater Lett 57:794–801
Zheng L, Hong FS, Lu SP, Liu C (2005) Effect of nano-TiO2 on strength of naturally aged seeds and growth of spinach. Biol Trace Element Res 104(1):82–93
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. 20671067, 30470150) and by the Jiangsu Province Universities Natural Science Foundation (grant no. 06KJB180094).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, F., Liu, C., Qu, C. et al. Was improvement of spinach growth by nano-TiO2 treatment related to the changes of Rubisco activase?. Biometals 21, 211–217 (2008). https://doi.org/10.1007/s10534-007-9110-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-007-9110-y