Abstract
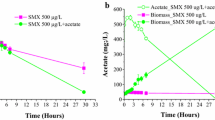
Sulfamethoxazole (SMX) has frequently been detected in aquatic environments. In natural environment, not only individual microorganism but also microbial consortia are involved in some biotransformation of pollutants. The competition for space under consortia causing cell–cell contact inhibition changes the cellular behaviors. Herein, the membrane bioreactor system (MBRS) was applied to improve SMX elimination thorough exchanging the cell-free broths (CFB). The removal efficiency of SMX was increased by more than 24% whether under the pure culture of A. faecalis or under the co-culture of A. faecalis and P. denitrificans with MBRS. Meanwhile, MBRS significantly inhibited the formation of HA-SMX, and Ac-SMX from parent compound. Additionally, the cellular growth under MBRS was obviously enhanced, indicating that the increases in the cellular growth under MBRS are possibly related to the decreases in the levels of HA-SMX and Ac-SMX compared to that without MBRS. The intracellular NADH/NAD+ ratios of A. faecalis under MBRS were increased whether thorough itself-recycle of CFB or exchanging CFB between the pure cultures of A. faecalis and P. denitrificans, suggesting that the enhancement in the bioremoval efficiencies of SMX under MBRS by A. faecalis is likely related to the increases in the NADH/NAD+ ratio. Taken together, the regulation of cell-to-cell communication is preferable strategy to improve the bioremoval efficiency of SMX.









Similar content being viewed by others
References
Alam MZ, Fakhru’L-Razi A, Abd-Aziz S, Molla AH (2003) Optimization of compatible mixed cultures for liquid state bioconversion of municipal wastewater sludge. Water Air Soil Pollut 149:113–126
Aydın S, Ince B, Ince O (2015) Inhibitory effect of erythromycin, tetracycline and sulfamethoxazole antibiotics on anaerobic treatment of a pharmaceutical wastewater. Water Sci Technol 71:1620–1628
Bonvin F, Omlin J, Rutler R, Schweizer WB, Alaimo PJ, Strathmann TJ, McNeill K, Kohn T (2013) Direct photolysis of human metabolites of the antibiotic sulfamethoxazole: evidence for abiotic back-transformation. Environ Sci Technol 47:6746–6755
Clara M, Strenn B, Ausserleitner M, Kreuzinger N (2004) Comparison of the behaviour of selected micropollutants in a membrane bioreactor and a conventional wastewater treatment plant. Water Sci Technol 50:29–36
Clement B, Behrens D, Amschler J, Matschke K, Wolf S, Havemeyer A (2005) Reduction of sulfamethoxazole and dapsone hydroxylamines by a microsomal enzyme system purified from pig liver and pig and human liver microsomes. Life Sci 77:205–219
Collado N, Buttiglieri G, Marti E, Ferrando-Climent L, Rodriguez-Mozaz S, Barceló D, Comas J, Rodriguez-Roda I (2013) Effects on activated sludge bacterial community exposed to sulfamethoxazole. Chemosphere 93:99–106
Cribb AE, Spielberg SP (1992) Sulfamethoxazole is metabolized to the hydroxylamine in humans. Clin Pharmacol Ther 51:522–526
Dai JJ, Cheng JS, Liang YQ, Jiang T, Yuan YJ (2014) Regulation of extracellular oxidoreduction potential enhanced (R, R)-2,3-butanediol production by Paenibacillus polymyxa CJX518. Bioresour Technol 167:433–440
Ebert BE, Kurth F, Grund M, Blank LM, Schmid A (2011) Response of Pseudomonas putida KT2440 to increased NADH and ATP demand. Appl Environ Microbiol 77:6597–6605
García-Galán MJ, Díaz-Cruz MS, Barceló D (2012) Removal of sulfonamide antibiotics upon conventional activated sludge and advanced membrane bioreactor treatment. Anal Bioanal Chem 404:1505–1515
Haack SK, Metge DW, Fogarty LR, Meyer MT, Barber LB, Harvey RW, Leblanc DR, Kolpin DW (2012) Effects on groundwater microbial communities of an engineered 30-day in situ exposure to the antibiotic sulfamethoxazole. Environ Sci Technol 46:7478–7486
Hai FI, Li X, Price WE, Nghiem LD (2011) Removal of carbamazepine and sulfamethoxazole by MBR under anoxic and aerobic conditions. Bioresour Technol 102:10386–10390
Herzog B, Lemmer H, Horn H, Müller E (2013) Characterization of pure cultures isolated from sulfamethoxazole-acclimated activated sludge with respect to taxonomic identification and sulfamethoxazole biodegradation potential. BMC Microbiol 13:276
Holtge S, Kreuzig R (2007) Laboratory testing of sulfamethoxazole and its metabolite acetyl-sulfamethoxazole in soil. Clean: Soil, Air, Water 35:104–110
Kagaya H, Miura M, Niioka T, Saito M, Numakura K, Habuchi T, Satoh S (2012) Influence of NAT2 polymorphisms on sulfamethoxazole pharmacokinetics in renal transplant recipients. Antimicrob Agents Chemother 56(2):825–829
Kemsawasd V, Branco P, Almeida MG, Caldeira J, Albergaria H, Arneborg N (2015) Cell-to-cell contact and antimicrobial peptides play a combined role in the death of Lachanchea thermotolerans during mixed-culture alcoholic fermentation with Saccharomyces cerevisiae. FEMS Microbiol Lett. https://doi.org/10.1093/femsle/fnv103
Kimura K, Hara H, Watanabe Y (2007) Elimination of selected acidic pharmaceuticals from municipal wastewater by an activated sludge system and membrane bioreactors. Environ Sci Technol 41:3708–3714
Kor-Bicakci G, Pala-Ozkok I, Rehma A, Jonas D, Ubay-Cokgor E, Orhon D (2014) Chronic impact of sulfamethoxazole on acetate utilization kinetics and population dynamics of fast growing microbial culture. Bioresour Technol 166:219–228
Laera G, Cassano D, Lopez A, Pinto A, Pollice A, Ricco G, Mascolo G (2012) Removal of organics and degradation products from industrial wastewater by a membrane bioreactor integrated with ozone or UV/H2O2 treatment. Environ Sci Technol 46:1010–1018
Larcher S, Yargeau V (2011) Biodegradation of sulfamethoxazole by individual and mixed bacteria. Appl Microbiol Biotechnol 91:211–218
Larcher S, Yargeau V (2013) The effect of ozone on the biodegradation of 17α-ethinylestradiol and sulfamethoxazole by mixed bacterial cultures. Appl Microbiol Biotechnol 97:2201–2210
Lavergne SN, Kurian JR, Bajad SU, Maki JE, Yoder AR, Guzinski MV, Graziano FM, Trepanier LA (2006) Roles of endogenous ascorbate and glutathione in the cellular reduction and cytotoxicity of sulfamethoxazole-nitroso. Toxicology 222:25–36
Li B, Zhang T (2010) Biodegradation and adsorption of antibiotics in the activated sludge process. Environ Sci Technol 44:3468–3473
Li X, Xu QM, Cheng JS, Yuan YJ (2016) Improving the bioremoval of sulfamethoxazole and alleviating cytotoxicity of its biotransformation by laccase producing system under coculture of Pycnoporus sanguineus and Alcaligenes faecalis. Bioresour Technol 220:333–340
Liu F, Wu J, Ying GG, Luo Z, Feng H (2012) Changes in functional diversity of soil microbial community with addition of antibiotics sulfamethoxazole and chlortetracycline. Appl Microbiol Biotechnol 95:1615–1623
Majewsky M, Wagner D, Delay M, Bräse S, Yargeau V, Horn H (2014) Antibacterial activity of sulfamethoxazole transformation products (TPs): general relevance for sulfonamide TPs modified at the para position. Chem Res Toxicol 27:1821–1828
Müller E, Schüssler W, Horn H, Lemmer H (2013) Aerobic biodegradation of the sulfonamide antibiotic sulfamethoxazole by activated sludge applied as co-substrate and sole carbon and nitrogen source. Chemosphere 92:969–978
Nie XP, Liu BY, Yu HJ, Liu WQ, Yang YF (2013) Toxic effects of erythromycin, ciprofloxacin and sulfamethoxazole exposure to the antioxidant system in Pseudokirchneriella subcapitata. Environ Pollut 172:23–32
Nissen P, Nielsen D, Arneborg N (2004) The relative glucose uptake abilities of non-Saccharomyces yeasts play a role in their coexistence with Saccharomyces cerevisiae in mixed cultures. Appl Microbiol Biotechnol 64(4):543–550
Ott G, Plitzko B, Krischkowski C, Reichmann D, Bittner F, Mendel RR, Kunze T, Clement B, Havemeyer A (2014) Reduction of sulfamethoxazole hydroxylamine (SMX-HA) by the mitochondrial amidoxime reducing component (mARC). Chem Res Toxicol 27:1687–1695
Pan S, Yan N, Liu X, Wang W, Zhang Y, Liu R, Rittmann BE (2014) How UV photolysis accelerates the biodegradation and mineralization of sulfadiazine (SD). Biodegradation 25:911–921
Parsek MR, Greenberg EP (2005) Sociomicrobiology: the connections between quorum sensing and biofilms. Trends Microbiol 13:27–33
Radke M, Lauwigi C, Heinkele G, Murdter TE, Letzel M (2009) Fate of the antibiotic sulfamethoxazole and its two major human metabolites in a water sediment test. Environ Sci Technol 43:3135–3141
Ricken B, Corvini PF, Cichocka D, Parisi M, Lenz M, Wyss D, Martínez-Lavanchy PM, Müller JA, Shahgaldian P, Tulli LG, Kohler HP, Kolvenbach BA (2013) Ipso-hydroxylation and subsequent fragmentation: a novel microbial strategy to eliminate sulfonamide antibiotics. Appl Environ Microbiol 79:5550–5558
Sacco JC, Clara LA (2010) Cytochrome b5 and NADH cytochrome b5 reductase: genotype-phenotype correlations for hydroxylamine reduction. Pharmacogenet Genom 20:26–37
Sahar E, Messalem R, Cikurel H, Aharoni A, Brenner A, Godehardt M, Jekel M, Ernst M (2011) Fate of antibiotics in activated sludge followed by ultrafiltration (CAS-UF) and in a membrane bioreactor (MBR). Water Res 45:4827–4836
Schröder HF, Tambosi JL, Sena RF, Moreira RF, José HJ, Pinnekamp J (2012) The removal and degradation of pharmaceutical compounds during membrane bioreactor treatment. Water Sci Technol 65:833–839
Schwarzenbach RP, Egli T, Hofstetter TB, von Gunten U, Wehrli B (2010) Global water pollution and human health. Annu Rev Environ Resour 35:109–136
Shen L, Yuan X, Shen W, He N, Wang Y, Lu H, Lu Y (2014) Positive impact of biofilm on reducing the permeation of ampicillin through membrane for membrane bioreactor. Chemosphere 97:34–39
Stadler LB, Su L, Moline CJ, Ernstoff AS, Aga DS, Love NG (2014) Effect of redox conditions on pharmaceutical loss during biological wastewater treatment using sequencing batch reactors. J Hazard Mater 282:106–115
Taillandier P, Lai QP, Julien-Ortiz A, Brandam C (2014) Interactions between Torulaspora delbrueckii and Saccharomyces cerevisiae in wine fermentation: influence of inoculation and nitrogen content. World J Microbiol Biotechnol 30(7):1959–1967
Tappe W, Hofmann D, Disko U, Koeppchen S, Kummer S, Vereecken H (2015) A novel isolated Terrabacter-like bacterium can mineralize 2-aminopyrimidine, the principal metabolite of microbial sulfadiazine degradation. Biodegradation 26:139–150
Wang Z, Zhang XH, Huang Y, Wang H (2015) Comprehensive evaluation of pharmaceuticals and personal care products (PPCPs) in typical highly urbanized regions across China. Environ Pollut 204:223–232
Xia S, Jia R, Feng F, Xie K, Li H, Jing D, Xu X (2012) Effect of solids retention time on antibiotics removal performance and microbial communities in an A/O-MBR process. Bioresour Technol 106:36–43
Xia Z, Xiao-chun W, Zhong-lin C, Hao X, Qing-fang Z (2015) Microbial community structure and pharmaceuticals and personal care products removal in a membrane bioreactor seeded with aerobic granular sludge. Appl Microbiol Biotechnol 99:425–433
Yang SF, Lin CF, Wu CJ, Ng KK, Lin AY, Hong PK (2012) Fate of sulfonamide antibiotics in contact with activated sludge–sorption and biodegradation. Water Res 46:1301–1308
Zhang R, Tang J, Li J, Zheng Q, Liu D, Chen Y, Zou Y, Chen X, Luo C, Zhang G (2013) Antibiotics in the offshore waters of the Bohai Sea and the Yellow Sea in China: occurrence, distribution and ecological risks. Environ Pollut 174:71–77
Zhang Q, Jia A, Wan Y, Liu H, Wang K, Peng H, Dong Z, Hu J (2014) Occurrences of three classes of antibiotics in a natural river basin: association with antibiotic-resistant Escherichia coli. Environ Sci Technol 48:14317–14325
Zhang YB, Zhou J, Xu QM, Cheng JS, Luo YL, Yuan YJ (2016) Exogenous cofactors for the improvements of sulfamethoxazole (SMX) bioremoval and biotransformation by Alcaligenes faecalis. Sci Total Environ 565:547–556
Acknowledgements
The authors are grateful for the financial support from the National Basic Research Program of China (973 Program) (Grant No. 2014CB745100), the National Natural Science Foundation of China (Grant No. 21576201).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tang, MH., Gao, N., Zhou, J. et al. Improvement of sulfamethoxazole (SMX) elimination and inhibition of formations of hydroxylamine-SMX and N4-acetyl-SMX by membrane bioreactor systems. Biodegradation 29, 245–258 (2018). https://doi.org/10.1007/s10532-018-9826-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-018-9826-0