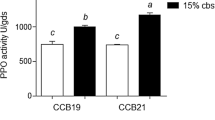
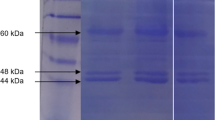
Abstract
The exploitation of food residual sources consists of a major factor in reducing the polluting load of food industry wastes and developing novel added-value products. Plant food residues including trimmings and peels might contain a range of enzymes capable of transforming bio-organic molecules with potential phytotoxicity, including hydrolases, peroxidases and polyphenoloxidases. Although the use of bacterial and fungal enzymes has gained interest in studies pertaining to bioremediation applications, plant enzymes have been given less attention or even disregarded. In this view, this study aimed at the investigating the use of a crude peroxidase preparation from onion solid by-products for oxidising caffeic acid, a widespread o-diphenol, whose various derivatives may occur in food industry wastes, such as olive mill waste waters. Increased enzyme activity was observed at a pH value of 5, but considerable activity was also retained for pH up to 7. Favourable temperatures for increased activity varied between 20°C and 40°C, 30°C being the optimal. Liquid chromatography-mass spectrometry analysis of a homogenate/H2O2-treated caffeic acid solution revealed the existence of a tetramer as major oxidation product. Based on the data generated, a putative pathway for the formation of the peroxidase-mediated caffeic acid tetramer was proposed.








Similar content being viewed by others
Abbreviations
- 4-AAP:
-
4-Aminoantipyrine
- BGP:
-
Bitter gourd peroxidase
- CA:
-
Caffeic acid
- CA-OP:
-
Caffeic acid oxidation product
- CouA:
-
p-Coumaric acid
- FA:
-
Ferulic acid
- HRP:
-
Horseradish peroxidase
- LC-MS:
-
Liquid chromatography-mass spectrometry
- OMWW:
-
olive mill waste water
- OSWH:
-
Onion solid waste homogenate
- SA:
-
Sinapic acid
- SBP:
-
Soybean peroxidase
- S.D.:
-
Standard deviation
- TMP:
-
Tomato peroxidase
- TNP:
-
Turnip peroxidase
References
Akhtar S, Husain Q (2006) Potential applications of immobilized bitter gourd (Momordica charantia) peroxidase in the removal of phenols from polluted water. Chemosphere 65:1228–1235
Akhtar S, Khan AA, Husain Q (2005) Potential of immobilized bitter gourd (Momordica charantia) peroxidases in the decolorization and removal of textile dyes from polluted wastewater and dying effluent. Chemosphere 60:291–301. doi:10.1016/j.chemosphere.2004.12.017
Arrieta-Baez D, Stark RE (2006) Modeling suberization with peroxidase-catalyzed polymerisation of hydroxycinnamic acids: cross-coupling and dimerization reactions. Phytochemistry 67:743–753. doi:10.1016/j.phytochem.2006.01.026
Azbar N, Bayram A, Filibeli A, Muezzinoglu A, Sengul F, Ozer A (2004) A review of waste management options in olive oil production. Crit Rev Environ Sci Technol 34:209–247. doi:10.1080/10643380490279932
Bassi A, Geng Z, Gijzen M (2004) Enzymatic removal of phenol and chlorophenols using soybean seed hulls. Eng Life Sci 4:125–130. doi:10.1002/elsc.200420021
Bódalo A, Gómez JL, Gómez E, Bastida J, Máximo MF (2006) Comparison of commercial peroxidases for removing phenol from water solutions. Chemosphere 63:626–632. doi:10.1016/j.chemosphere.2005.08.007
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Caza N, Bewtra JK, Biswas N, Taylor KE (1999) Removal of phenolic compounds from synthetic wastewater using soybean peroxidase. Water Res 33:3012–3018. doi:10.1016/S0043-1354(98)00525-9
Cheng J, Yu SM, Zuo P (2006) Horseradish peroxidase immobilized on aluminium-pillared interlayered clay for the catalytic oxidation of phenolic wastewater. Water Res 40:283–290. doi:10.1016/j.watres.2005.11.017
Dalal S, Gupta MN (2007) Treatment of phenolic wastewater by horseradish peroxidase immobilized by bioaffinity layering. Chemosphere 67:741–747. doi:10.1016/j.chemosphere.2006.10.043
De Marco E, Savarese M, Paduano A, Sacchi R (2007) Characterization and fractionation of phenolic compounds extracted from olive oil mill wastewaters. Food Chem 104:858–867. doi:10.1016/j.foodchem.2006.10.005
DellaGreca M, Previtera L, Temussi F, Zarrelli A (2004) Low-molecular-weight components of olive oil mill waste-waters. Phytochem Anal 15:184–188. doi:10.1002/pca.766
Derat E, Shaik S (2006) An efficient proton-coupled electron-transfer process during oxidation of ferulic acid by horseradish peroxidase: coming full cycle. J Am Chem Soc 128:13940–13949. doi:10.1021/ja065058d
Duarte-Vázquez MA, Ortega-Tovar MA, García-Almendarez BE, Regalado C (2002) Removal of aqueous phenolic compounds from a model system by oxidative polymerisation with turnip (Brassica napus L. var purple top white globe) peroxidase. J Chem Technol Biotechnol 78:42–47. doi:10.1002/jctb.740
Flock C, Bassi A, Gijzen M (1999) Removal of aqueous phenols and 2-chlorophenol with purified soybean peroxidase and raw soybean hulls. J Chem Technol Biotechnol 74:303–309. doi :10.1002/(SICI)1097-4660(199904)74:4<303::AID-JCTB38>3.0.CO;2-B
Geng Z, Rao J, Bassi AS, Gijzen M, Krishnamoorthy N (2001) Investigation of biocatalytic properties of soybean seed hull peroxidase. Catal Today 64:233–238. doi:10.1016/S0920-5861(00)00527-7
González PS, Capozucca CE, Tigier HA, Milrad SR, Agostini E (2006) Phytoremediation of phenol from wastewater, by peroxidases of tomato hairy root cultures. Enzyme Microb Technol 39:647–653. doi:10.1016/j.enzmictec.2005.11.014
Hapiot P, Neudeck A, Pinson J, Fulcrand H, Neta P, Rolando C (1996) Oxidation of caffeic acid and related hydroxycinnamic acids. J Electroanal Chem 405:169–176. doi:10.1016/0022-0728(95)04412-4
Hewson WD, Dunford HB (1976) Oxidation of p-cresol by horseradish peroxidase compound I. J Biol Chem 251:6036–6042
Huang Q, Weber WJ (2004) Peroxidase-catalyzed coupling of phenol in the presence of model inorganic and organic solid phases. Environ Sci Technol 38:5238–5245. doi:10.1021/es049826h
Huang Q, Weber WJ (2005) Transformation and removal of bisphenol A from aqueous phase via peroxidase-mediated oxidative coupling reactions: efficacy, products, and pathways. Environ Sci Technol 39:6029–6036. doi:10.1021/es050036x
Karam J, Nicell JA (1997) Potential applications of enzymes in waste treatment. J Chem Technol Biotechnol 69:141–153. doi :10.1002/(SICI)1097-4660(199706)69:2<141::AID-JCTB694>3.0.CO;2-U
Kennedy K, Alemany K, Warith M (2002) Optimisation of soybean peroxidase treatment of 2, 4-dichlorophenol. Water SA 28:149–158
Liu H-L, Wan X, Huang X-F, Kong L-Y (2007) Biotransformation of sinapic acid catalysed by Momordica charantia peroxidase. J Agric Food Chem 55:1003–1008. doi:10.1021/jf0628072
Mantzavinos D, Kalogerakis N (2005) Treatment of olive mill effluents. Part I. Organic matter degradation by chemical and biological processes-an overview. Environ Int 31:289–295. doi:10.1016/j.envint.2004.10.005
Obied HK, Allen MS, Bedgood DR Jr, Prenzler PD, Robards K (2005) Investigation of Australian olive mill waste for recovery of biophenols. J Agric Food Chem 53:9911–9920. doi:10.1021/jf0518352
Oudgenoeg G, Hilhorst R, Piersma SR, Boeriu CG, Gruppen H, Hessing M et al (2001) Peroxidase-mediated cross-linking of a tyrosine-containing peptide with ferulic acid. J Agric Food Chem 49:2503–2510. doi:10.1021/jf000906o
Pati S, Losito I, Palmisano F, Zambonin PG (2006) Characterization of caffeic acid enzymatic oxidation by-products by liquid chromatography coupled to electrospray ionisation tandem mass spectrometry. J Chrom A 1102:184–192. doi:10.1016/j.chroma.2005.10.041
Petrucci R, Astolfi P, Greci L, Firuzi O, Saso L, Marrosu G (2007) A spectroelectrochemical and chemical study on oxidation of hydroxycinnamic acids in aprotic medium. Electrochim Acta 52:2461–2470. doi:10.1016/j.electacta.2006.08.053
Ralph J, Bunzel M, Marita JM, Hatfield RD, Lu F, Kim H et al (2004) Peroxidase-dependent cross-linking reactions of p-hydroxycinnamates in plant cell walls. Phytochem Rev 3:79–96. doi:10.1023/B:PHYT.0000047811.13837.fb
Singh A, Billingsley KA, Ward OP (2000) Transformation of polychlorinated biphenyls with oxidative enzymes. Bioproc Eng 23:421–425. doi:10.1007/s004499900121
Takahama U, Hirota S (2000) Deglucosidation of quercetin glucosides to the aglycone and formation of antifungal agents by peroxidase-dependent oxidation of quercetin on browning of onion scales. Plant Cell Physiol 41:1021–1029. doi:10.1093/pcp/pcd025
Tatsumi T, Wada S, Ichikawa H (1996) Removal of chlorophenols from wastewater by immobilized horseradish peroxidase. Biotechnol Bioeng 51:126–130. doi :10.1002/(SICI)1097-0290(19960705)51:1<126::AID-BIT15>3.0.CO;2-O
Tong Z, Qingxiang Z, Hui H, Qin L, Yi Z (1997) Removal of toxic phenol and 4-chlorophenol from waste water by horseradish peroxidase. Chemosphere 34:893–903. doi:10.1016/S0045-6535(97)00015-5
Tong Z, Qingxiang Z, Hui H, Qin L, Yi Z (1998) Kinetic study on the removal of toxic phenol and chlorophenol from waste water by horseradish peroxidase. Chemosphere 37:1571–1577. doi:10.1016/S0045-6535(98)00140-4
Veitch NC (2004) Structural determinants of plant peroxidase function. Phytochem Rev 3:3–18. doi:10.1023/B:PHYT.0000047799.17604.94
Wagner M, Nicell JA (2002) Detoxification of phenolic solutions with horseradish peroxidase and hydrogen peroxide. Water Res 36:4041–4052. doi:10.1016/S0043-1354(02)00133-1
Wilberg K, Assenhaimer C, Rubio J (2002) Removal of aqueous phenol catalysed by a low purity soybean peroxidase. J Chem Technol Biotechnol 77:851–857. doi:10.1002/jctb.646
Wright H, Nicell JA (1999) Characterization of soybean peroxidase for the treatment of aqueous phenols. Biores Tech 70:69–79
Wu Y, Taylor KE, Biswas N, Bewtra JK (1997) Comparison of additives in the removal of phenolic compounds by peroxidase-catalyzed polymerisation. Water Res 31:2699–2704. doi:10.1016/S0043-1354(97)00119-X
Yamada K, Shibuya T, Noda M, Uchiyama N, Kashiwada A, Matsuda K et al (2007) Influence of position of substituent groups on removal of chlorophenols and cresols by horseradish peroxidase and determination of optimum conditions. Biosci Biotechnol Biochem 71:2503–2510. doi:10.1271/bbb.70298
Yu B-B, Han X-Z, Lou H-X (2007) Oligomers of resveratrol and ferulic acid prepared by peroxidase-catalyzed oxidation and their protective effects on cardiac injury. J Agric Food Chem 55:7753–7757. doi:10.1021/jf0711486
Zhang G, Nicell JA (2000) Treatment of aqueous pentachlorophenol by horseradish peroxidase and hydrogen peroxide. Water Res 34:1629–1637. doi:10.1016/S0043-1354(99)00326-7
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agha, A.E., Abbeddou, S., Makris, D.P. et al. Biocatalytic properties of a peroxidase-active cell-free extract from onion solid wastes: caffeic acid oxidation. Biodegradation 20, 143–153 (2009). https://doi.org/10.1007/s10532-008-9208-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-008-9208-0