Abstract
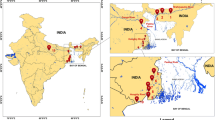
The Malayan tapir (Tapirus indicus) is an endangered species in Southeast Asia (SEA). Over the years, there has only been a few reports on its population genetic structure and evolutionary history in SEA. In particular, while the captive Malayan tapir population in Thailand has received fairly more research in recent years, there has not been any wide-scale population genetic study in the Malay Peninsula since the last decade. In this study, we report on the genetic diversity and attempt to make inferences on the phylogeography of the Malayan tapir in the Malay Peninsula, using the mitochondrial DNA control region, including also data from previous studies. We identified 12 novel haplotypes and two distinct Malayan tapir clades with a divergence time of 14.6 million years ago (mya) in the Peninsular Malaysia (i.e. southern Malay Peninsula) samples, as opposed to only one clade found in the Thai (i.e. Thailand western border and northern Malay Peninsula) captive individuals, with an almost two-fold higher nucleotide diversity compared to the latter. The co-occurrence of the two lineages in southern Malay Peninsula suggested past historical events of population isolation, migration and admixture as a result of episodic glacial periods and paleoenvironmental changes in SEA. Only one haplotype shared between the populations of the two regions suggested further gene flow restriction at the narrow corridor of the Malay Peninsula as of present day.


Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available in GenBank (MT519685-MT519728) and in this published article and its supplementary information file. Additional information is available from the corresponding author on reasonable request.
References
Abdul J (2000) The status of Malayan tapir (Tapirus indicus) in Peninsular Malaysia. Tapir Conserv 10:15
Allendorf FW, Luikart G, Aitken SN (2013) Conservation and the genetics of populations, 2nd edn. Wiley-Blackwell, Hoboken
Arnason U, Adegoke JA, Gullberg A et al (2008) Mitogenomic relationships of placental mammals and molecular estimates of their divergences. Gene 421:37–51. https://doi.org/10.1016/j.gene.2008.05.024
Ashley MV, Norman JE, Stross L (1996) Phylogenetic analysis of the Perissodactyla family Tapiridae using mitochondrial cytochrome c oxidase (COII) sequences. J Mamm Evol 3:315–325
Bandelt HJ, Forster P, Röhl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48
Cranbrook EO, Piper PJ (2013) Paleontology to policy: the Quaternary history of Southeast Asian tapirs (Tapiridae) in relation to large mammal species turnover, with a proposal for conservation of Malayan tapir by reintroduction to Borneo. Integr Zool 8:95–120. https://doi.org/10.1111/j.1749-4877.2012.00319.x
Darriba D, Toboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772
de Thoisy B, da Silva A, Ruiz-Garcia M et al (2010) Population history, phylogeography, and conservation genetics of the last Neotropical mega-herbivore, the lowland tapir (Tapirus terrestris). BMC Evol Biol 10:278. https://doi.org/10.1186/1471-2148-10-278
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567
Fritz SA, Bininda-Emonds ORP, Purvis A (2009) Geographical variation in predictors of mammalian extinction risk: big is bad, but only in the tropics. Ecol Lett 12:538–549. https://doi.org/10.1111/j.1461-0248.2009.01307.x
Fu X-Y (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 147:915–925
García MJ, Medici EP, Naranjo EJ et al (2012) Distribution, habitat and adaptability of the genus Tapirus. Integr Zool 7:346–355. https://doi.org/10.1111/j.1749-4877.2012.00317.x
Gonçalves da Silva A, Lalonde DR, Quse V et al (2010) Genetic approaches refine ex situ lowland tapir (Tapirus terrestris) conservation. J Hered 101:581–590
Gorog AJ, Sinaga MH, Engstrom MD (2004) Vicariance or dispersal? Historical biogeography of three Sunda shelf murine rodents (Maxomys surifer, Leopoldamys sabanus and Maxomys whiteheadi). Biol J Linn Soc 81:91–109. https://doi.org/10.1111/j.1095-8312.2004.00281.x
Guindon S, Gascuel O (2003) A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Syst Biol 52:696–704
Higgins D, Thompson J, Gibson T et al (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Hughes JB, Round PD, Woodruff DS (2003) The Indochinese-Sundaic faunal transition at the Isthmus of Kra: an analysis of resident forest bird species distributions: Avian transition at the Isthmus of Kra. J Biogeogr 30:569–580. https://doi.org/10.1046/j.1365-2699.2003.00847.x
Humphreys AM, Barraclough TG (2014) The evolutionary reality of higher taxa in mammals. Proc R Soc B Biol Sci 281:20132750. https://doi.org/10.1098/rspb.2013.2750
IUCN (International Union for Conservation of Nature) (2008) Tapirus indicus. The IUCN Red List of Threatened Species. Version 2021-1. Available at http://www.iucnredlist.org. Accessed 25 on Mar 2021
Jama M, Zhang Y, Aman RA, Ryder OA (1993) Sequence of the mitochondrial control region, tRNA thr, tRNS pro and tRNA phe genes from the black rhinoceros, Diceros bicornis. Nucleic Acids Res 21:4392–4392. https://doi.org/10.1093/nar/21.18.4392
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular Evolutionary Genetics Analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lowe A, Harris S, Ashton P (2009) Ecological genetics design, analysis, and application. Wiley, New York
Magintan D, Traeholt C, Karuppanannan KV (2012) Displacement of the Malayan Tapir (Tapirus indicus) in Peninsular Malaysia from 2006 to 2010. Tapir Conserv 21:13–17
Mantel NA (1967) The detection of disease clustering and generalised regression approach. Cancer Res 27:209–220
Mello B (2018) Estimating TimeTrees with MEGA and the TimeTree resource. Mol Biol Evol 35:2334–2342. https://doi.org/10.1093/molbev/msy133
Muangkram Y, Wajjwalku W, Salakij C et al (2013) Use of mitochondrial cytochrome b sequences to determine the origin of captive Asian tapirs Tapirus indicus: implications for conservation. Endanger Species Res 21:97–103. https://doi.org/10.3354/esr00509
Muangkram Y, Wajjwalku W, Kaolim N et al (2016) The complete mitochondrial genome of the Asian tapirs (Tapirus indicus): the only extant Tapiridae species in the old world. Mitochondrial DNA Part A 27:413–415. https://doi.org/10.3109/19401736.2014.898283
Muangkram Y, Amano A, Wajjwalku W et al (2017) Genetic diversity of the captive Asian tapir population in Thailand, based on mitochondrial control region sequence data and the comparison of its nucleotide structure with Brazilian tapir. Mitochondrial DNA Part A 28:597–601. https://doi.org/10.3109/24701394.2016.1149828
Norton JE, Ashley MV (2004a) Genetic variability and population differentiation in captive Baird’s Tapirs (Tapirus bairdii). Zoo Biol 23:521–531. https://doi.org/10.1002/zoo.20031
Norton JE, Ashley MV (2004b) Genetic variability and population structure among wild Baird’s tapirs. Anim Conserv 7:211–220. https://doi.org/10.1017/S1367943004001295
Ogata M, Watanabe S, Ogawa H (2009) Genetic variation of Asian tapir. Jpn Soc Zoo Wildl Med 14:73–76
Paradis E, Claude J, Strimmer K (2004) APE: analysis of phylogenetics and evolution in R language. Bioinformatics 20:289–290
Parnell J (2013) The biogeography of the Isthmus of Kra region: a review. Nord J Bot 31:001–015. https://doi.org/10.1111/j.1756-1051.2012.00121.x
Pinho GM, Gonçalves da Silva A, Hrbek T et al (2014) Kinship and social behavior of Lowland Tapirs (Tapirus terrestris) in a Central Amazon landscape. PLoS ONE 9:1–10. https://doi.org/10.1371/journal.pone.0092507
Pitra C, Veits J (2000) Use of mitochondrial DNA sequences to test the Ceratomorpha (Perissodactyla: Mammalia) hypothesis. J Zool Syst Evol Res 38:65–72
Rovie-Ryq J, Traeholt C, Elagupillay S et al (2008) Sequence variation in Malayan Tapir (Tapirus indicus) inferred using partial sequences of the cytochrome b segment of the mitochondrial DNA. J Wildl Parks 25:16–18
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC et al (2017) DnaSP 6: DNA sequence polymorphism analysis of large datasets. Mol Biol Evol 34:3299–3302. https://doi.org/10.1093/molbev/msx248
Ruiz-García M, Vásquez C, Pinedo-Castro M et al (2012) Phylogeography of the Mountain Tapir (Tapirus pinchaque) and the Central American Tapir (Tapirus bairdii) and the origins of the three Latin-American tapirs by means of mtCyt-B sequences. In: Anamthawat-Jónsson K (ed) Current topics in phylogenetics and phylogeography of terrestrial and aquatic systems. InTech, Rijeka, pp 83–116
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Tamura K, Battistuzzi FU, Billing-Ross P et al (2012) Estimating divergence times in large molecular phylogenies. Proc Natl Acad Sci 109:19333–19338. https://doi.org/10.1073/pnas.1213199109
Tamura K, Tao Q, Kumar S (2018) Theoretical foundation of the RelTime method for estimating divergence times from variable evolutionary rates. Mol Biol Evol 35:1770–1782. https://doi.org/10.1093/molbev/msy044
Tougard C (2001) Biogeography and migration routes of large mammal faunas in South-East Asia during the Late Middle Pleistocene: focus on the fossil and extant faunas from Thailand. Palaeogeogr Palaeoclimatol Palaeoecol 168:337–358
Traeholt C, Novarino W, bin Saaban S, et al (2016) Tapirus indicus. The IUCN Red List of Threatened Species 2016: e.T21472A45173636. Available at http://www.iucnredlist.org/details/21472/0. Accessed on 18 Jul 2016
Voris HK (2000) Maps of Pleistocene sea levels in Southeast Asia: shorelines, river systems and time durations. J Biogeogr 27:1153–1167. https://doi.org/10.1046/j.1365-2699.2000.00489.x
Woodruff DS, Turner LM (2009) The Indochinese-Sundaic zoogeographic transition: a description and analysis of terrestrial mammal species distributions. J Biogeogr 36:803–821. https://doi.org/10.1111/j.1365-2699.2008.02071.x
Acknowledgements
This project was supported by the Ministry of Education Malaysia’s Fundamental Research Grant Scheme (FRGS); project code: FRGS/1/2014/SG03/UPM/02/8); project vote no: 5524552, and Malaysian Nature Society Tapir Project Grant awarded to Geetha Annavi. We would like to thank Donny Yawah (PERHILITAN), Mat Naim Bin Haji Ramli (National Zoo of Malaysia) and Kavitha Jayaseelan (National Zoo of Malaysia) for their assistance in collecting blood samples at the Sungai Dusun WCC and the National Zoo of Malaysia respectively. We also thank Mdm Noor Azleen binti Mohd Kulaimi (PERHILITAN) for aiding in the collection of tapir samples from the NWFL, PERHILITAN. We would also like to thank the staff from PERHILITAN, National Zoo of Malaysia and Sungai Dusun WCC for providing information on the Malayan tapir samples. All scientific activities were conducted under the permit from PERHILITAN (permit ref. NRE 600-2/2/21 JILID 2(42)). All the sampling procedures for the whole blood samples were approved by the Institutional Animal Care and Use Committee, Universiti Putra Malaysia (ethical approval ref.: UPM/IACUC/AUP-R033/2016). All methods were performed in accordance with the Universiti Putra Malaysia Code of Practice for the Care and Use of Animals for Scientific Purposes.
Funding
This study was supported by the Malaysian Ministry of Education’s Fundamental Research Grant Scheme (FRGS) (Project Code: FRGS/1/2014/SG03/UPM/02/8; Project Vote No. 5524552), and Malaysian Nature Society Tapir Project Grant awarded to Geetha Annavi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All the sampling procedures for the whole blood samples were approved by the Institutional Animal Care and Use Committee, Universiti Putra Malaysia (Ethical Approval Ref.: UPM/IACUC/AUP-R033/2016). All methods were performed in accordance with the Universiti Putra Malaysia Code of Practice for the Care and Use of Animals for Scientific Purposes.
Additional information
Communicated by Pradeep Kumar Divakar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lim, Q.L., Yong, C.S.Y., Ng, W.L. et al. Genetic diversity and phylogenetic relationships of Malayan tapir (Tapirus indicus) populations in the Malay Peninsula based on mitochondrial DNA control region. Biodivers Conserv 30, 2433–2449 (2021). https://doi.org/10.1007/s10531-021-02202-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-021-02202-x