Abstract
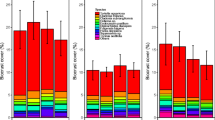
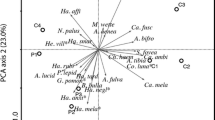
The species-rich calcareous grassland communities in Europe are gradually disappearing due to lack of management such as grazing or cultivation, resulting in decalcification and reduction of gaps in the vegetation. In this study, experimental soil perturbation (deep and shallow) was performed in degenerated sandy grassland in plots with a size of 8 × 8 m, using a randomised block design. The hypothesis that soil perturbation that inverts the soil layers decreases nutrient availability, creates vegetation gaps and thereby selects for desirable species was tested through comparisons with untreated controls as well as with nearby target habitats. The deep perturbation was designed to bring CaCO3 up to the surface, whereas the shallow perturbation tested the effect of disturbance alone. The effects of soil perturbation on soil chemistry, vegetation and beetle communities were analysed for the 2 years following the treatments. Increased pH and calcium concentration, and decreased nitrogen and phosphorus availability, showed that deep perturbation was successful in restoring the soil chemistry to levels similar to those of the target habitat. Perturbated plots were rapidly colonised by the acid tolerant grass Corynephorus canescens, but the slow colonisation of the threatened calcicole species Koeleria glauca was an indication that the vegetation could be evolving towards the target vegetation. Six red-listed beetle species associated with open, dry grasslands were found, out of which four were found only in perturbated plots, although this could not be statistically tested. In conclusion, it may take many years or even decades for the establishment of desirable flora, and seeding could therefore be a suitable method of increasing the rate of succession.





Similar content being viewed by others
References
Bobbink R (1991) Effects of nutrient enrichment in Dutch chalk grassland. J Appl Ecol 28:28–41
Bobbink R, Willems JH (1987) Increasing dominance of Brachypodium pinnatum (L) Beauv in chalk grasslands—a threat to a species-rich ecosystem. Biol Conserv 40:301–314
Bobbink R, Hornung M, Roelofs JGM (1998) The effects of air-borne nitrogen pollutants on species diversity in natural and semi-natural European vegetation. J Ecol 86:717–738
Bossuyt B, Butaye J, Honnay O (2006) Seed bank composition of open and overgrown calcareous grassland soils—a case study from Southern Belgium. J Environ Manag 79:364–371
Braak CJF (1988–2005) CANOCO, version 4.54
Carroll JA, Caporn SJM, Johnson D et al (2003) The interactions between plant growth, vegetation structure and soil processes in semi-natural acidic and calcareous grasslands receiving long-term inputs of simulated pollutant nitrogen deposition. Environ Pollut 121:363–376
de Graaf MCC, Verbeek PJM, Bobbink R et al (1998) Restoration of species-rich dry heaths: the importance of appropriate soil conditions. Acta Bot Neerl 47:89–111
Desender K, Bosmans R (1998) Ground beetles (Coleoptera, Carabidae) on set-aside fields in the Campine region and their importance for nature conservation in Flanders (Belgium). Biodivers Conserv 7:1485–1493
Dolman PM, Sutherland WJ (1992) The ecological changes of Breckland grass heaths and the consequences of management. J Appl Ecol 29:402–413
Dolman PM, Sutherland WJ (1994) The use of soil disturbance in the management of Breckland grass heaths for nature conservation. J Environ Manag 41:123–140
Eichberg C, Storm C, Kratochwil A (2006) A different method for seed bank analysis: validation and application to successional stages of Koelerio-Corynephoretea inland sand vegetation. Phytocoenologia 36:161–189
Eichberg C, Storm C, Schwabe A (2007) Endozoochorous dispersal, seedling emergence and fruiting success in disturbed and undisturbed successional stages of sheep-grazed inland sand ecosystems. Flora 202:3–27
Ellenberg H (1991) Indicator values of vascular plants in Europe 1. Indicator values of plants not including Rubus. Scripta Geobot 18:9–166
Gibson CWD, Brown VK (1991) The nature and rate of development of calcareous grassland in southern Britain. Biol Conserv 58:297–316
Golterman HL (1998) The calcium- and iron bound phosphate phase diagram. Hydrobiologia 159:149–151
Gønget H (1997) The Brentidae (Coleoptera) of Northern Europe. Fauna Entomol Scan 34:119–121
Graham DJ, Hutchings MJ (1988) A field investigation of germination from the seed bank of a chalk grassland ley on former arable land. J Appl Ecol 25:253–264
Hansen V (1918) Biller IV. Snudebiller. Danmarks Fauna 22
Hansson S-Å, Jeppson M (2005) Gasteromyceter i östra Skånes sandstäppsområden—en sammanfattning av elva års inventeringsarbete. Sven Mykol Tidskr 26:61–83
Hirst RA, Pywell RF, Marrs RH et al (2005) The resilience of calcareous and mesotrophic grasslands following disturbance. J Appl Ecol 42:498–506
Hobbs RJ, Huenneke LF (1992) Disturbance diversity and invasion implications for conservation. Conserv Biol 6:324–337
Janssens F, Peeters A, Tallowin JRB et al (1998) Relationship between soil chemical factors and grassland diversity. Plant Soil 202:69–78
Jeschke M, Kiehl K (2008) Restoration and management of calcareous grasslands: Is there a trade-off between species diversity of cryptogams and vascular plants? In: Paper presented at the 6th European conference on ecological restoration, Ghent, Belgium, 8–12 September 2008
Kiehl K, Pfadenhauer J (2007) Establishment and persistence of target species in newly created calcareous grasslands on former arable fields. Plant Ecol 189:31–48
Klimešovà J, Klimeš L (2006) Clo-Pla3. http://clopla.butbn.cas.cz/. Cited 5 Sept 2010
Kooijman AM, Besse M (2002) The higher availability of N and P in lime-poor than in lime-rich coastal dunes in the Netherlands. J Ecol 90:394–403
Kotanen PM (1996) Revegetation following soil disturbance in a California meadow: the role of propagule supply. Oecologia 108:652–662
Kotanen PM (2004) Revegetation following soil disturbance and invasion in a Californian meadow: a 10-year history of recovery. Biol Invasions 6:245–254
Laurence M, Jones M, Norman K et al (2010) Topsoil inversion as a restoration measure in sand dunes, early results from a UK field-trial. J Coast Conserv 14:139–151
Ljungberg H (1999) Skalbaggar och andra insekter på sandstäppslokaler i östra Skåne. Report from the County Administrative Board of Scania, Malmö, Sweden
Ljungberg H (2002a) Bete, störning och biologisk mångfald i odlingslandskapet—Hotade skalbaggar i öländska torrmarker. Report no. 2002:20. County Administrative Board of Kalmar, Kalmar, Sweden
Ljungberg H (2002b) Important habitats for red-listed ground beetles in Sweden. Entomol Tidskr 123:167–185
Ljungberg H (2007) Åtgärdsprogram för dynglevande skalbaggar 2007–2011. Report no. 5689. Swedish Environmental Protection Agency, Stockholm, Sweden
Lundberg S, Gustafsson B (1995) Catalogus Coleopterorum Sueciae. Naturhistoriska Riksmuseet, Stockholm, Sweden
Mårtensson LM, Olsson PA (2010) Soil chemistry of local vegetation gradients in sandy calcareous grasslands. Plant Ecol 206:127–138
Mattiasson G (1974) Sandstäpp: vegetation, dynamik och skötsel. Meddelanden för Avdelningen för Ekologisk Botanik 2:1–39
Mattiasson G (2009) Åtgärdsprogram för sandnörel (Minuartia viscosa) 2008–2014. Report no. 5949. Swedish Environmental Protection Agency, Stockholm, Sweden
Matus G, Tothmeresz B, Papp M (2003) Restoration prospects of abandoned species-rich sandy grassland in Hungary. Appl Veg Sci 6:169–178
Mitchley J, Xofis P (2005) Landscape structure and management regime as indicators of calcareous grassland habitat condition and species diversity. J Nat Conserv 13:171–183
Mossberg B, Stenberg L (2003) Den nya Nordiska floran, 2nd edn. Wahlström and Widstrand, Stockholm, Sweden
Niemelä J, Baur B (1998) Threatened species in a vanishing habitat: plants and invertebrates in calcareous grasslands in the Swiss Jura mountains. Biodivers Conserv 7:1407–1416
Olsson K-A (1994) Sandstäpp i Skåne—ett upprop. Lunds Botaniska Förenings Medlemsblad 2:4–13
Olsson PA, Mårtensson LM, Bruun HH (2009) Acidification of sandy grasslands–consequences for plant diversity. Appl Veg Sci 12:350–361
Olsson PA, Schnoor TK, Hanson S-Å (2010) pH preferences of red-listed gasteromycetes in calcareous sandy grasslands: Implications for conservation and restoration. Fungal Ecol 3:357–365
Pärtel M, Kalamees R, Zobel M et al (1998) Restoration of species-rich limestone grassland communities from overgrown land: the importance of propagule availability. Ecol Eng 10:275–286
Poschlod P, WallisDeVries MF (2002) The historical and socioeconomic perspective of calcareous grasslands—lessons from the distant and recent past. Biol Conserv 104:361–376
Röder D, Kiehl K (2006) Population structure and population dynamic of Pulsatilla patens (L.) Mill. in relation to vegetation characteristics. Flora 201:499–507
Roem WJ, Berendse F (2000) Soil acidity and nutrient supply ratio as possible factors determining changes in plant species diversity in grassland and heathland communities. Biol Conserv 92:151–161
Schläpfer M, Zoller H, Koerner C (1998) Influences of mowing and grazing on plant species composition in calcareous grassland. Bot Helv 108:57–67
Sjörs H (1967) Nordisk växtgeografi, 2:a upplagan. Svenska Bokförlaget, Bonniers, Stockholm
Storm C, Suss K (2008) Are low-productive plant communities responsive to nutrient addition? Evidence from sand pioneer grassland. J Veg Sci 19:343–354
Tyler G (1992) Inability to solubilize phosphate in limestone soils—key factor controlling calcifuge habit of plants. Plant Soil 145:65–70
Tyler T (2003) The state of the sand-steppe vegetation of eastern Scania (Skåne) in early spring 2003. Bot Notiser 136:1–22
Tyler T (2005) The Bryophyte flora of Scanian sand-steppe vegetation and its relation to soil pH and phosphate availability. Lindbergia 30:11–20
Tyler G (2008) The ground beetle fauna (Coleoptera:Carabidae) of abandoned fields, as related to plant cover, previous management and succession stage. Biodivers Conserv 17:155–172
Van Swaay CAM (2002) The importance of calcareous grasslands for butterflies in Europe. Biol Conserv 104:315–318
Verhagen R, Klooker J, Bakker JP et al (2001) Restoration success of low-production plant communities on former agricultural soils after top-soil removal. Appl Veg Sci 4:75–82
WallisDeVries MF, Poschlod P, Willems JH (2002) Challenges for the conservation of calcareous grasslands in northwestern Europe: Integrating the requirements of flora and fauna. Biol Conserv 104:265–273
Wassen MJ, Venterink HO, Lapshina ED et al (2005) Endangered plants persist under phosphorus limitation. Nature 437:547–550
Wenzel M, Schmitt T, Weitzel M et al (2006) The severe decline of butterflies on western German calcareous grasslands during the last 30 years: A conservation problem. Biol Conserv 128:542–552
Willems JH (2001) Problems, approaches, and results in restoration of Dutch calcareous grassland during the last 30 years. Restor Ecol 9:147–154
Woodcock BA, Pywell RF (2010) Effects of vegetation structure and floristic diversity on detrivore, herbivore and predatory invertebrates within calcareous grassland. Biodivers Conserv 19:81–95
Woodcock BA, Pywell RF, Roy DB et al (2005) Grazing management of calcareous grasslands and its implications for the conservation of beetle communities. Biol Conserv 125:193–202
Woodcock BA, Edwards AR, Lawson CS et al (2008) Contrasting success in the restoration of plant and phytophagous beetle assemblages of species-rich mesotrophic grasslands. Oecologia 154:773–783
Acknowledgements
The Swedish Environmental Protection Agency (the LONA initiative), Region Skåne, The Swedish Research Council, The Swedish Research Council for the Environment, Agricultural Sciences and Spatial Planning (Formas), and the Oskar and Lilli Lamm Memorial Foundation are gratefully acknowledged for funding this work. We would also like to thank Carina Wettemark and Sam Skällberg at the municipality of Kristianstad for help in planning the restoration experiment.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Table 2.
Rights and permissions
About this article
Cite this article
Ödman, A.M., Mårtensson, LM., Sjöholm, C. et al. Immediate responses in soil chemistry, vegetation and ground beetles to soil perturbation when implemented as a restoration measure in decalcified sandy grassland. Biodivers Conserv 20, 3039–3058 (2011). https://doi.org/10.1007/s10531-011-0108-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-011-0108-y