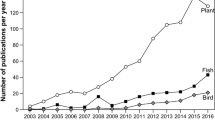
Abstract
Ecological specialisation is hypothesised to play a major role in the evolution of phenotypic diversity, especially following the colonisation of novel habitats. For example, cichlid fishes provide some of the most remarkable evidence for rapid ecological diversification. Here, we capitalised on a recently (≤ 40 years ago) introduced population of red devils (Amphilophus labiatus) in Australia to investigate adaptive phenotypic responses to a novel environment. We used stomach content analyses, morphometrics and laboratory experiments to test for covariation between diet and size of an important trophic trait, lip size. We found that, while maximum lip size in the study population was smaller than in the species’ natural range, the proportions of algae, insects and fish remains in the diet covaried with lip size. However, contrary to predictions, we found no evidence for lip development to be plastic under laboratory conditions in relation to substrate complexity or food manipulation, nor did we find any relationship between lip morphology and feeding performance in adults. Single nucleotide polymorphism data, in turn, suggested that the introduced population has reduced standing genetic variation, which potentially influences both phenotypic plasticity and diversity, in comparison to native populations. Together, the results suggest that morphological variation in a key trophic trait can respond rapidly to diet in a novel environment, despite reduced genetic diversity in the population.



Similar content being viewed by others
References
Adams CE, Huntingford FA (2004) Incipient speciation driven by phenotypic plasticity? Evidence from sympatric populations of Arctic Charr. Biol J Linn Soc 81:611–618. https://doi.org/10.1111/j.1095-8312.2004.00314.x
Agrawal N, Mittal AK (1991) Epithelium of lips and associated structures of the Indian major carp, Catla catla. Jpn J Ichthyol 37:363–373. https://doi.org/10.1007/BF02905362
Allendorf FW, Lundquist LL (2003) Introduction: Population biology, evolution, and control of invasive species. Conserv Biol 17:24–30. https://doi.org/10.1046/j.1523-1739.2003.02365.x
Arnegard ME, Snoeks J (2001) New three-spotted cichlid species with hypertrophied lips (Teleostei: Cichlidae) from the deep waters of Lake Malaŵi/Nyasa, Africa. Copeia 2001:705–717
Barlow GW, Munsey JW (1976) The red devil - Midas - arrow cichlid species complex in Nicaragua. In: Thorson TB (ed) Investigations of the ichthyofauna of Nicaraguan Lakes. School of Life Sciences, University of Nebraska-Lincoln, pp 359–369
Barluenga M, Meyer A (2004) The Midas cichlid species complex: Incipient sympatric speciation in Nicaraguan cichlid fishes? Mol Ecol 13:2061–2076. https://doi.org/10.1111/j.1365-294X.2004.02211.x
Barluenga M, Meyer A (2010) Phylogeography, colonization and population history of the Midas cichlid species complex (Amphilophus spp.) in the Nicaraguan crater lakes. BMC Evol Biol 10:326. https://doi.org/10.1186/1471-2148-10-326
Barluenga M, Stölting KN, Salzburger W, Muschick M, Meyer A (2006) Sympatric speciation in Nicaraguan crater lake cichlid fish. Nature 439:719–723. https://doi.org/10.1038/nature04325
Barrett SCH (1991) Genetic and evolutionary consequences of small population size in plants: implications for conservation. In: Falk D, Holsinger KE (eds) Genetics and conservation of rare plants. Oxford University Press, New York and Oxford, pp 3–30
Barrett RD, Schluter D (2008) Adaptation from standing genetic variation. Trends Ecol Evol 23:38–44. https://doi.org/10.1016/j.tree.2007.09.008
Baumgarten L, Machado-Schiaffino G, Henning F, Meyer A (2015) What big lips are good for: on the adaptive function of repeatedly evolved hypertrophied lips of cichlid fishes. Biol J Linn Soc 115:448–455. https://doi.org/10.1111/bij.12502
Behera N, Nanjundiah V (2004) Phenotypic plasticity can potentiate rapid evolutionary change. J Theor Biol 226:177–184. https://doi.org/10.1016/j.jtbi.2003.08.011
Bernatchez L, Wilson CC (1998) Comparative phylogeography of Nearctic and Palearctic fishes. Mol Ecol 7:431–452. https://doi.org/10.1046/j.1365-294x.1998.00319.x
Bolnick DI, Yang LH, Fordyce JA, Davis JM, Svanbäck R (2002) Measuring individual-level resource specialization. Ecology 83:2936–2941. https://doi.org/10.1890/00129658(2002)083[2936:MILRS]-2.0.CO;2
Bolnick DI, Svanbäck R, Fordyce JA, Yang LH, Davis JM, Hulsey CD, Forister ML (2003) The ecology of individuals: incidence and implications of individual specialization. Am Nat 161:1–28. https://doi.org/10.1086/343878
Bolnick DI, Snowberg LK, Hirsch PE, Lauber CL, Org E, Parks B, Lusis AJ, Knight R, Caporaso JG, Svanbäck R (2014) Individual diet has sex-dependent effects on vertebrate gut microbiota. Nat Commun 5:1–13. https://doi.org/10.1038/ncomms5500
Burress ED, Duarte A, Serra WS, Gangloff MM, Siefferman L (2013) Species-specific ontogenetic diet shifts among Neotropical Crenicichla: Using stable isotopes and tissue stoichiometry. J Fish Biol 82:1904–1915. https://doi.org/10.1111/jfb.12117
Burress ED (2015) Cichlid fishes as models of ecological diversification: Patterns, mechanisms, and consequences. Hydrobiologia 748:7–27. https://doi.org/10.1007/s10750-014-1960-z
Catchen JM, Hohenlohe PA, Bassham S, Amores A, Cresko WA (2013) Stacks: An analysis tool set for population genomics. Mol Ecol 22:3124–3140. https://doi.org/10.1111/mec.12354
Catchen JM, Amores A, Hohenlohe P, Cresko W, Postlethwait JH, Hohenlohe PA, Bassham S, Amores A, Cresko WA (2011) Stacks: Building and genotyping loci de novo from short-read sequences. G3 1:171–182. https://doi.org/10.1534/g3.111.000240
Clabaut C, Bunje PME, Salzburger W, Meyer A (2007) Geometric morphometric analyses provide evidence for the adaptive character of the Tanganyikan cichlid fish radiations. Evolution 61:560–578. https://doi.org/10.1111/j.1558-5646.2007.00045.x
Colombo M, Diepeveen ET, Muschick M, Santos ME, Indermaur A, Boileau N, Barluenga M, Salzburger W (2013) The ecological and genetic basis of convergent thick-lipped phenotypes in cichlid fishes. Mol Ecol 22:670–684. https://doi.org/10.1111/mec.12029
Corfield J, Diggles B, Jubb C, McDowall RM, Moore A, Richards A, Rowe DK (2008) Review of the impacts of introduced ornamental fish species that have established wild populations in Australia. Prepared for the Department of the Environment, Water, Heritage and the Arts, Canberra, ACT. www.environment.gov. au/biodiversity/publications/index.html
Crawley MJ (2012) Mixed-effects models. The R book. Wiley, United Kingdom, pp 681–714
Doebeli M, Dieckmann U, Metz JA, Tautz D (2005) What we have also learned: adaptive speciation is theoretically plausible. Evolution 59:691–695. https://doi.org/10.1554/04-154
Elston DA, Moss R, Boulinier T, Arrowsmith C, Lambin X (2001) Analysis of aggregation, a worked example: number of ticks on red grouse. Parasitology 122:563–569. https://doi.org/10.1017/S0031182001007740
Elmer KR, Lehtonen TK, Kautt A, Harrod C, Meyer A (2010a) Rapid sympatric ecological differentiation of crater lake cichlid fishes within historic times. BMC Biol 8:60. https://doi.org/10.1186/1741-7007-8-60
Elmer KR, Kusche H, Lehtonen TK, Meyer A (2010b) Local variation and parallel evolution: morphological and genetic diversity across a species complex of Neotropical crater lake cichlid fishes. Philos T R Soc B 365:1763–1782. https://doi.org/10.1098/rstb.2009.0271
Elmer KR, Lehtonen TK, Fan S, Meyer A (2013) Crater lake colonization by Neotropical cichlid fishes. Evolution 67:281–288. https://doi.org/10.1111/j.1558-5646.2012.01755.x
Ferry LA, Konow N, Gibb AC (2012) Are kissing gourami specialized for substrate-feeding? Prey capture kinematics of Helostoma temminckii and other Anabantoid fishes. J Exp Zool 317:571–579. https://doi.org/10.1002/jez.1749
Fierst JL (2011) A history of phenotypic plasticity accelerates adaptation to a new environment. J Evol Biol 24:1992–2001. https://doi.org/10.1111/j.1420-9101.2011.02333.x
Fryer G (1959) The trophic interrelationships and ecology of some littoral communities of Lake Nyasa with especial reference to the fishes, and a discussion of the evolution of a group of rock-frequenting Cichlidae. P Zool Soc Lond 132:153–281. https://doi.org/10.1111/j.1469-7998.1959.tb05521.x
Ghalambor CK, McKay JK, Carroll SP, Reznick DN (2007) Adaptive versus non-adaptive phenotypic plasticity and the potential for contemporary adaptation in new environments. Funct Ecol 21:394–407. https://doi.org/10.1111/j.1365-2435.2007.01283.x
Grant PR, Grant B (2008) How and why species multiply: the radiation of Darwin’s finches. Princeton University Press, Princeton
Greenwood PH (1974) The cichlid fishes of Lake Victoria, East Africa: The biology and evolution of a species flock. British Natural History Museum, London, pp 134
Hendry AP (2009) Ecological speciation! Or the lack thereof? Can J Fish Aquat Sci 66:1383–1398. https://doi.org/10.1139/F09-074
Hendry AP, Wenburg JK, Bentzen P, Volk EC, Quinn TP (2000) Rapid evolution of reproductive isolation in the wild: evidence from introduced salmon. Science 290:516–518. https://doi.org/10.1126/science.290.5491.516
Henning F, Machado-Schiaffino G, Baumgarten L, Meyer A (2017) Genetic dissection of adaptive form and function in rapidly speciating cichlid fishes. Evolution 71:1297–1312. https://doi.org/10.1111/evo.13206
Hulsey CD, Hendrickson DA, García de León FJ (2005) Trophic morphology, feeding performance and prey use in the polymorphic fish Herichthys minckleyi. Evol Ecol Res 7:303–324
Kinziger AP, Nakamoto RJ, Anderson EC, Harvey BC (2011) Small founding number and low genetic diversity in an introduced species exhibiting limited invasion success (speckled dace, Rhinichthys osculus). Ecol Evol 1:73–84. https://doi.org/10.1002/ece3.8
Klingenberg CP, Barluenga M, Meyer A (2003) Body shape variation in cichlid fishes of the Amphilophus citrinellus species complex. Biol J Linn Soc 80:397–408. https://doi.org/10.1046/j.1095-8312.2003.00246.x
Kocher TD (2004) Adaptive evolution and explosive speciation: the cichlid fish model. Nat Rev Genet 5:288–298. https://doi.org/10.1038/nrg1316
Kornfield IL, Koehn RK (1975) Genetic variation and speciation in new world cichlids. Evolution 29:427–437. https://doi.org/10.2307/2407255
Ledon-Rettig CC, Pfennig DW, Nascone-Yoder N (2008) Ancestral variation and the potential for genetic accommodation in larval amphibians: implications for the evolution of novel feeding strategies. Evol Dev 10:316–325. https://doi.org/10.1111/j.1525-142X.2008.00240.x
Lehtonen TK, Kvarnemo C (2015) Infections may select for filial cannibalism by impacting egg survival in interactions with water salinity and egg density. Oecologia 178:673–683. https://doi.org/10.1007/s00442-015-3246-1
Losos JB (2011) Convergence, adaptation, and constraint. Evolution 65:1827–1840. https://doi.org/10.1111/j.1558-5646.2011.01289.x
Lu G, Bernatchez L (1999) A study of fluctuating asymmetry in hybrids of dwarf and normal lake whitefish ecotypes (Coregonus clupeaformis) from different glacial races. Heredity 83:742–747. https://doi.org/10.1046/j.1365-2540.1999.00625.x
Machado-Schiaffino G, Henning F, Meyer A (2014) Species-specific differences in adaptive phenotypic plasticity in an ecologically relevant trophic trait: hypertrophic lips in Midas cichlid fishes. Evolution 68:2086–2091. https://doi.org/10.1111/evo.12367
Machado-Schiaffino G, Henning GF, Meyer A (2017) Incipient speciation driven by hypertrophied lips in Midas cichlids fish? Mol Ecol 26:2348–2362. https://doi.org/10.1111/mec.14029
Manousaki T, Hull PM, Kusche H, Machado-Schiaffino G, Franchini P, Harrod C, Elmer KR, Meyer A (2013) Parsing parallel evolution: ecological divergence and differential gene expression in the adaptive radiations of thick-lipped Midas cichlid fishes from Nicaragua. Mol Ecol 22:650–669. https://doi.org/10.1111/mec.12034
McKinnon JS, Rundle HD (2002) Speciation in nature: the threespine stickleback model systems. Trends Ecol Evol 17:480–488. https://doi.org/10.1016/S0169-5347(02)02579-X
Meyer A (1987) Phenotypic plasticity and heterochrony in Cichlasoma managuense (Pisces, Chichlidae) and their implications for speciation in cichlid fishes. Evolution 41:1357–1369. https://doi.org/10.2307/2409100
Muschick M, Barluenga M, Salzburger W, Meyer A (2011) Adaptive phenotypic plasticity in the Midas cichlid fish pharyngeal jaw and its relevance in adaptive radiation. BMC Evol Biol 11:116. https://doi.org/10.1186/1471-2148-11-116
Oliver MK, Arnegard ME (2010) A new genus for Melanochromis labrosus, a problematic Lake Malawi cichlid with hypertrophied lips (Teleostei: Cichlidae). Ichthyol Explor Fres 21:209–232
Pfennig DW, McGee M (2010) Resource polyphenism increases species richness: a test of the hypothesis. Philos T R Soc B 365:577–591. https://doi.org/10.1098/rstb.2009.0244
Pfennig DW, Wund MA, Snell-Rood EC, Cruickshank T, Schlichting CD, Moczek AP (2010) Phenotypic plasticity’s impacts on diversification and speciation. Trends Ecol Evol 25:459–467. https://doi.org/10.1016/j.tree.2010.05.006
Pigliucci M (2005) Evolution of phenotypic plasticity: where are we going now? Trends Ecol Evol 20:481–486. https://doi.org/10.1016/j.tree.2005.06.001
Rasband W (1997) ImageJ (version 1.50b). National Institutes of Health, Bethesda. https://www.rsb.info.nih.gov/ij
Salzburger W (2009) The interaction of sexually and naturally selected traits in the adaptive radiations of cichlid fishes. Mol Ecol 18:169–185. https://doi.org/10.1111/j.1365-294X.2008.03981.x
Salzburger W, Mack T, Verheyen E, Meyer A (2005) Out of Tanganyika: genesis, explosive speciation, key-innovations and phylogeography of the Haplochromine cichlid fishes. BMC Evol Biol 5:17. https://doi.org/10.1186/1471-2148-5-17
Savolainen V, Lexer C, Anstett M-C, Hutton I, Clarkson JJ, Norup MV, Powell MP, Springate D, Salamin N, Baker WJ (2006) Evolutionary biology: sympatric plant speciation in islands? Nature 441:210–213. https://doi.org/10.1038/nature05217
Scheiner SM (1993) Genetics and evolution of phenotypic plasticity. Annu Rev Ecol Evol S 24:35–68. https://doi.org/10.1146/annurev.es.24.110193.000343
Schlichting CD (2004) The role of phenotypic plasticity in diversification. Oxford University Press, New York and Oxford
Schluter D (2000) The ecology of adaptive radiation. Oxford University Press, New York and Oxford
Schluter D, Conte GL (2009) Genetics and ecological speciation. P Natl Acad Sci USA 106:9955–9962. https://doi.org/10.1073/pnas.0901264106
Schluter D, Nagel LM (1995) Parallel speciation by natural selection. Am Nat 146:292–301. https://doi.org/10.1086/285799
Skúlason S, Snorrason SS, Jonsson B (1999) Sympatric morphs, populations and speciation in freshwater fish with emphasis on Arctic charr. In: Magurran AE, May RM (eds) Evolution of biological diversity. Oxford University Press, New York and Oxford, pp 70–92
Smith TB, Skúlason S (1996) Evolutionary significance of resource polymorphisms in fishes, amphibians, and birds. Annu Rev Ecol Evol S 27:111–133. https://doi.org/10.1146/annurev.ecolsys.27.1.111
Sorenson MD, Sefc KM, Payne RB (2003) Speciation by host switch in brood parasitic indigobirds. Nature 424:928–931. https://doi.org/10.1038/nature01863
Stauffer JR, van Snick GE (2004) Phenotypic plasticity: Its role in trophic radiation and explosive speciation in cichlids (Teleostei: Cichlidae). Anim Biol 54:137–158. https://doi.org/10.1163/1570756041445191
Wagner CE, McIntyre PB, Buels KS, Gilbert DM, Michel E (2009) Diet predicts intestine length in Lake Tanganyika’s cichlid fishes. Funct Ecol 23:1122–1131. https://doi.org/10.1111/j.1365-2435.2009.01589.x
Wente WH, Phillips JB (2003) Fixed green and brown color morphs and a novel color-changing morph of the Pacific tree frog Hyla regilla. Am Nat 162:461–473. https://doi.org/10.1086/378253
West-Eberhard MJ (1989) Phenotypic plasticity and the origins of diversity. Annu Rev Ecol Evol S 20:249–278. https://doi.org/10.1146/annurev.es.20.110189.001341
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press, New York and Oxford
Willis TJ, Babcock RC (1998) Retention and in situ detectability of visible implant fluorescent elastomer (VIFE) tags in Pagrus auratus (Sparidae). New Zeal J Mar Fresh 32:247–254. https://doi.org/10.1080/00288330.1998.9516823
Witte F, Barel C, Hoogerhoud R (1989) Phenotypic plasticity of anatomical structures and its ecomorphological significance. Neth J Zool 40:278–298. https://doi.org/10.1163/156854289X00309
Wund MA (2012) Assessing the impacts of phenotypic plasticity on evolution. Integr Comp Biol 52:5–15. https://doi.org/10.1093/icb/ics050
Yamaoka K (1997) Trophic ecomorphology of Tanganyikan cichlids. In: Kawanabe H, Hori M, Nagoshi M (eds) Fish communities in Lake Tanganyika. Kyoto University Press, Kyoto, pp 25–26
Yeh PJ, Price TD (2004) Adaptive phenotypic plasticity and the successful colonization of a novel environment. Am Nat 164:531–542. https://doi.org/10.1086/423825
Zuur AF, Hilbe J, Ieno EN (2013) A beginner’s guide to GLM and GLMM with R: A frequentist and Bayesian perspective for ecologists. Highland Statistics Ltd., United Kingdom
Acknowledgements
We thank Carlos Garita Alvarado, Fiona Kang, Rowan Jacques-Hamilton, Nicholas Deal, Rachel Fetherston, Eeling Ng, Ruby Albury, Andrej Hohmann, Stefanie Forster, Madeleine De Jong and Matthew Simpson for logistic support and animal collection, and members of the Wong and Chapple Labs, Monash University, who assisted with animal husbandry. We also thank Bernd Egger, Christian Beisel Melinda Hofmann, Mariana Leal Cardín and Gema Aguilera García for their assistance in the laboratory, with preparation of samples for genetic analysis. We thank the staff of the DNA Database of Japan (DDBJ) for their assistance with the use of the NIG supercomputer to conduct population genomic analyses. Lastly, we thank David Chapple, Walter Salzburger, Marius Rösti, Bernd Egger and Alysha Heimberg for their advice in regard to genetic analysis.
Funding
This study was funded by a Holsworth Wildlife Endowment, and a Linnean Society of New South Wales Small Research Grant, both awarded to WS. The research also received support from the SYNTHESYS Project (https://www.synthesys.info/), which is financed by European Community Research Infrastructure Action under the FP7 "Capacities" Program (to TKL and MB).
Author information
Authors and Affiliations
Contributions
WS, TKL and BBMW formulated the idea, developed methodology, and collected the field data. MB organised, and participated in, data collection in Nicaragua. WS wrote the first draft of the manuscript, conducted laboratory experiments and prepared samples for sequencing. WS, TKL and MR performed statistical analyses. All authors contributed to writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Collection and experimental procedures were approved by the Animal Ethics Committee of Monash University, Australia (BSCI/2012/23), and complied with all relevant State and Federal laws. Collection in Nicaragua was approved by the Ministerio del Ambiente y los Recursos Naturales (MARENA), Permit Number: 013-102013.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sowersby, W., Lehtonen, T.K., Ravinet, M. et al. Resource trait specialisation in an introduced fish population with reduced genetic diversity. Biol Invasions 22, 2447–2460 (2020). https://doi.org/10.1007/s10530-020-02264-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02264-y