Abstract
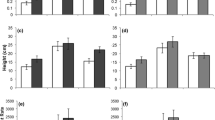
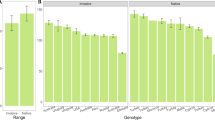
Biotic resistance to invasion arises from strong species interactions that decrease the fitness and population growth rates of potential invaders. Strong, direct interactions such as predation and competition are typically thought to drive biotic resistance, but in diverse communities, indirect interactions among species may also affect biotic resistance. Further, genetic variation in traits of the invading species that affect species interactions may allow some genotypes to overcome biotic resistance. We investigated the direct and indirect effects of a native legume (Acmispon wrangelianus) and insect herbivores on the fitness of different genotypes of an invasive legume (Medicago polymorpha) in a California grassland. Insect herbivores decreased Medicago fitness, but only in the presence of Acmispon, suggesting that indirect interactions mediated through insects and Acmispon are important for deterring Medicago invasion. Some Medicago genotypes were less affected by interactions with other species, however. This genetic variance suggests that while biotic resistance reduced the reproductive success of most genotypes, a few genotypes were able to overcome these complex interactions. However, Medicago invasion was unsuccessful in all treatments at several of our sites, suggesting that factors beyond those manipulated here also play a key role at many sites. At sites where biotic resistance is important, spatial and temporal variation in community composition and the genetic composition of the invasion pool may explain the invasion success of Medicago into this community.



Similar content being viewed by others
References
Abrams PA (1992) Predators that benefit prey and prey that harm predators: unusual effects of interacting foraging adaptation. Am Nat 140:573–600
Adams MJ, Pearl CA, Bury RB (2003) Indirect facilitation of an anuran invasion by non-native fishes. Ecol Lett 6:343–351
Barney JN, Di Tommaso A, Weston LA (2005) Differences in invisibility of two contrasting habitats and invasiveness of two mugwort Artemisia vulgaris populations. J Appl Ecol 42:567–576
Case TJ (1990) Invasion resistance arises in strongly interacting species-rich model competition communities. Proc Natl Acad Sci USA 87:9610–9614
Crawford KM, Whitney KD (2010) Population genetic diversity influences colonization success. Mol Ecol 19:1253–1263
Dawson W, Burslem DFRP, Hulme PE (2009) Factors explaining alien plant invasion success in a tropical ecosystem differ at each stage of invasion. J Ecol 97:657–665
de Haan RL, Barnes DK (1998) Inheritance of pod type, stem color, and dwarf growth habit in Medicago polymorpha. Crop Sci 38:1558–1561
Elton CS (1958) The ecology of invasions by animals and plants. Metheuan and Co., Ltd., London
Epelbaum A, Pearce CM, Barker DJ, Paulson A, Therriault TW (2009) Susceptibility of non-indigenous ascidian species in British Columbia (Canada) to invertebrate predation. Mar Biol 156:1311–1320
Fridley JD, Stachowicz JJ, Naeem S, Sax DF, Seabloom EW, Smith MD, Stohlgren TJ, Tilman D, Von Holle B (2007) The invasion paradox: reconciling patterns and process in species invasions. Ecology 88:3–17
Gordon DR, Onderdonk DA, Fox AM, Stocker RK (2008) Consistent accuracy of the Australia weed risk assessment system across varied geographies. Divers Distrib 14:234–242
Gruner DS (2005) Biotic resistance to an invasive spider conferred by generalist insectivorous birds on Hawai’I Island. Biol Invasion 7:541–546
Lau JA, Strauss SY (2005) Insect herbivores drive important indirect effects of exotic plants on native communities. Ecology 86:2990–2997
Lee CE (2002) Evolutionary genetics of invasive species. Trends Ecol Evol 17:386–391
Levine JM, Adler PB, Yelenik SG (2004) A meta-analysis of biotic resistance to exotic plant invasions. Ecol Lett 7:975–989
Maron JL, Connors PG (1996) A native nitrogen-fixing shrub facilitates weed invasion. Oecologia 105:302–312
Maron JL, Vila M (2001) When do herbivores affect plant invasion? Evidence for the natural enemies and biotic resistance hypotheses. Oikos 95:361–373
May RM, MacArthur RH (1972) Niche overlap as a function of environmental variability. Proc Natl Acad Sci USA 69:1109–1113
Menge BA (1995) Indirect effects in marine rocky intertidal interaction webs: patterns and importance. Ecol Monogr 65:21–74
Miller TE, terHorst CP (2012) Indirect effects in communities and ecosystems. Oxford Bibliographies in Ecology. In: David Gibson (ed), http://www.oxfordbibliographies.com/obo/page/ecology. Oxford University Press, New York
Moulton MP, Pimm SL (1983) The introduced Hawiian avifauna: biogeographic evidence for competition. Am Nat 121:669–690
Paini DR, Funderburk JE, Reitz SR (2008) Competitive exclusion of a worldwide invasive pest by a native: quantifying competition between two phytophagous insects on two host plant species. J Anim Ecol 77:184–190
Parker JD, Hay ME (2005) Biotic resistance to plant invasions? Native herbivores prefer non-native plants. Ecol Lett 8:959–967
Porter SS, Rice KJ (2013) Trade-offs, spatial heterogeneity, and the maintenance of microbial diversity. Evolution 67:599–608
Strauss SY (1991) Indirect effects in community ecology: their definition, study, and importance. Trends Ecol Evol 6:206–210
Suwa T, Louda SM (2012) Combined effects of plant competition and insect herbivory hinder invasiveness of an introduced thistle. Oecologia 169:467–476
terHorst CP (2010) Evolution in response to direct and indirect effects in pitcher plant inquiline communities. Am Nat 176:675–685
terHorst CP, Lau JA (2012) Direct and indirect transgenerational effects alter plant-herbivore interactions. Evol Ecol 26:1469–1480
Theoharides KA, Dukes JS (2007) Plant invasion across space and time: factors affecting nonindigenous species success during four stages of invasion. New Phytol 176:256–273
Van Kleunen M, Weber E, Fischer M (2010) A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol Lett 13:235–245
Violle C, Enquist BJ, McGill BJ, Jiang L, Albert CH, Hulshof C, Jung V, Messier J (2012) The return of the variance: intraspecific variability in community ecology. Trends Ecol Evol 332(27):244–252
Vitale M, Pupilli F, Labombarda P, Arcioni S (1998) RAPD analysis reveals a low rate of outcrossing in burr medic (Medicago polymorpha L.). Gen Res Crop Evol 45:337–342
Williamson M, Fitter A (1996) The varying success of invaders. Ecology 77:1661–1666
Wootton JT (1994) The nature and consequences of indirect effects in ecological communities. Ann Rev Ecol Syst 25:443–466
Acknowledgments
We thank C. Gomola for field assistance in work conducted at the University of California Natural Reserve System’s Donald and Sylvia McLaughlin Reserve. This manuscript was improved by comments from T. Bassett, R. Prunier, E. Schultheis, T. Suwa, K. Whitney, the Ecology Reading Group at Florida State University, and several anonymous reviewers. Funding was provided by awards from the National Science Foundation to JAL (DEB-0918963) and to CPt (DMS-132490). Data from this study are archived at Dryad (doi:10.5061/dryad.s6n57). This is contribution #1707 from the W. K. Kellogg Biological Station.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
terHorst, C.P., Lau, J.A. Genetic variation in invasive species response to direct and indirect species interactions. Biol Invasions 17, 651–659 (2015). https://doi.org/10.1007/s10530-014-0756-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-014-0756-4