Abstract
Objectives
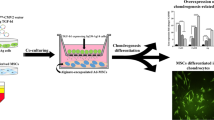
We genetically modified dedifferentiated chondrocytes (DCs) using lentiviral vectors and adenoviral vectors encoding TGF-β3 (referred to as transgenic groups below) and encapsulated these DCs in the microcavitary hydrogel and investigated the combinational effect on redifferentiation of the genetically manipulated DCs.
Results
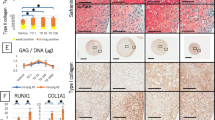
The Cell Counting Kit-8 data indicated that both transgenic groups exhibited significantly higher cell viability in the first week but inferior cell viability in the subsequent timepoints compared with those of the control group. Real-time polymerase chain reaction and western blot analysis results demonstrated that both transgenic groups had a better effect on redifferentiation to some extent, as evidenced by higher expression levels of chondrogenic genes, suggesting the validity of combination with transgenic DCs and the microcavitary hydrogel on redifferentiation. Although transgenic DCs with adenoviral vectors presented a superior extent of redifferentiation, they also expressed greater levels of the hypertrophic gene type X collagen. It is still worth further exploring how to deliver TGF-β3 more efficiently and optimizing the appropriate parameters, including concentration and duration.
Conclusions
The results demonstrated the better redifferentiation effect of DCs with the combinational use of transgenic TGF-β3 and a microcavitary alginate hydrogel and implied that DCs would be alternative seed cells for cartilage tissue engineering due to their easily achieved sufficient cell amounts through multiple passages and great potential to redifferentiate to produce cartilaginous extracellular matrix.









Similar content being viewed by others
References
Bohme K, Winterhalter KH, Bruckner P (1995) Terminal differentiation of chondrocytes in culture is a spontaneous process and is arrested by transforming growth factor-beta 2 and basic fibroblast growth factor in synergy. Exp Cell Res 216(1):191–198. https://doi.org/10.1006/excr.1995.1024
Byers BA, Mauck RL, Chiang IE, Tuan RS (2008) Transient exposure to transforming growth factor beta 3 under serum-free conditions enhances the biomechanical and biochemical maturation of tissue-engineered cartilage. Tissue Eng Pt A 14(11):1821–1834. https://doi.org/10.1089/ten.tea.2007.0222
Charlier E, Deroyer C, Ciregia F, Malaise O, Neuville S, Plener Z, Malaise M, de Seny D (2019) Chondrocyte dedifferentiation and osteoarthritis (oa). Biochem Pharmacol 165:49–65. https://doi.org/10.1016/j.bcp.2019.02.036
Chen Y, Ouyang X, Wu Y, Guo S, Xie Y, Wang G (2020) Co-culture and mechanical stimulation on mesenchymal stem cells and chondrocytes for cartilage tissue engineering. Curr Stem Cell Res Ther 15(1):54–60. https://doi.org/10.2174/1574888x14666191029104249
Dahlin RL, Ni M, Meretoja VV, Kasper FK, Mikos AG (2014) Tgf-β3-induced chondrogenesis in co-cultures of chondrocytes and mesenchymal stem cells on biodegradable scaffolds. Biomaterials 35(1):123–132. https://doi.org/10.1016/j.biomaterials.2013.09.086
Du X, Cai L, Xie J, Zhou X (2023) The role of tgf-beta3 in cartilage development and osteoarthritis. Bone Res 11(1):2. https://doi.org/10.1038/s41413-022-00239-4
Georgi N, van Blitterswijk C, Karperien M (2014) Mesenchymal stromal/stem cell-or chondrocyte-seeded microcarriers as building blocks for cartilage tissue engineering. Tissue Eng Pt A 20(17–18):2513–2523. https://doi.org/10.1089/ten.TEA.2013.0681
Glyn-Jones S, Palmer AJR, Agricola R, Price AJ, Vincent TL, Weinans H, Carr AJ (2015) Osteoarthritis. Lancet 386(9991):376–387. https://doi.org/10.1016/s0140-6736(14)60802-3
Han Y, Lian MF, Wu Q, Qiao ZG, Sun BB, Dai KR (2021) Effect of pore size on cell behavior using melt electrowritten scaffolds. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2021.629270
He A, Ye A, Song N, Liu N, Zhou G, Liu Y, Ye X (2020) Phenotypic redifferentiation of dedifferentiated microtia chondrocytes through a three-dimensional chondrogenic culture system. Am J Transl Res 12(6):2903–2915
Hou M, Bai B, Tian B, Ci Z, Liu Y, Zhou G, Cao Y (2021) Cartilage regeneration characteristics of human and goat auricular chondrocytes. Front Bioeng Biotechnol 9:766363. https://doi.org/10.3389/fbioe.2021.766363
Islam A, Tom VJ (2022) The use of viral vectors to promote repair after spinal cord injury. Exp Neurol 354:114102. https://doi.org/10.1016/j.expneurol.2022.114102
Jung H, McClellan P, Welter JF, Akkus O (2021) Chondrogenesis of mesenchymal stem cells through local release of tgf-β3 from heparinized collagen biofabric. Tissue Eng Part A 27(21–22):1434–1445. https://doi.org/10.1089/ten.TEA.2020.0383
Kim C, Linsenmeyer KD, Vlad SC, Guermazi A, Clancy MM, Niu JB, Felson DT (2014) Prevalence of radiographic and symptomatic hip osteoarthritis in an urban united states community. Arthritis Rheumatol 66(11):3013–3017. https://doi.org/10.1002/art.38795
Kim MJ, Lee JH, Kim JS, Kim HY, Lee HC, Byun JH, Lee JH, Kim NH, Oh SH (2020) Intervertebral disc regeneration using stem cell/growth factor-loaded porous particles with a leaf-stacked structure. Biomacromol 21(12):4795–4805. https://doi.org/10.1021/acs.biomac.0c00992
Ko CY, Ku KL, Yang SR, Lin TY, Peng S, Peng YS, Cheng MH, Chu IM (2016) In vitro and in vivo co-culture of chondrocytes and bone marrow stem cells in photocrosslinked pcl-peg-pcl hydrogels enhances cartilage formation. J Tissue Eng Regen Med 10(10):E485-e496. https://doi.org/10.1002/term.1846
Lee WYW, Wang B (2017) Cartilage repair by mesenchymal stem cells: Clinical trial update and perspectives. J Orthop Transl 9:76–88. https://doi.org/10.1016/j.jot.2017.03.005
Minegishi Y, Hosokawa K, Tsumaki N (2013) Time-lapse observation of the dedifferentiation process in mouse chondrocytes using chondrocyte-specific reporters. Osteoarthr Cartil 21(12):1968–1975. https://doi.org/10.1016/j.joca.2013.09.004
Rakic R, Bourdon B, Hervieu M, Branly T, Legendre F, Saulnier N, Audigié F, Maddens S, Demoor M, Galera P (2017) Rna interference and bmp-2 stimulation allows equine chondrocytes redifferentiation in 3d-hypoxia cell culture model: Application for matrix-induced autologous chondrocyte implantation. Int J Mol Sci 18(9):1842. https://doi.org/10.3390/ijms18091842
Savenkova DA, Makarova AA, Shalik IK, Yudkin DV (2022) Mirna pathway alteration in response to non-coding rna delivery in viral vector-based gene therapy. Int J Mol Sci 23(23):14954. https://doi.org/10.3390/ijms232314954
Wang X, Manner PA, Horner A, Shum L, Tuan RS, Nuckolls GH (2004) Regulation of mmp-13 expression by runx2 and fgf2 in osteoarthritic cartilage. Osteoar Cartil 12(12):963–973. https://doi.org/10.1016/j.joca.2004.08.008
Yao YC, Zeng L, Huang YY (2016) The enhancement of chondrogenesis of atdc5 cells in rgd-immobilized microcavitary alginate hydrogels. J Biomater Appl 31(1):92–101. https://doi.org/10.1177/0885328216640397
Yao Y, Huang Y, Qian D, Zhang S, Chen Y, Bai B (2017) Effect of various ratios of co-cultured atdc5 cells and chondrocytes on the expression of cartilaginous phenotype in microcavitary alginate hydrogel. J Cell Biochem 118(11):3607–3615. https://doi.org/10.1002/jcb.26218
Yao Y, Zhang T, Chen H, Zheng S, Chen Y, Zhang S (2020) Enhanced chondrogenesis in a coculture system with genetically manipulated dedifferentiated chondrocytes and atdc5 cells. Biotechnol Bioeng. https://doi.org/10.1002/bit.27482
Yao Y, Wang C (2020) Dedifferentiation: Inspiration for devising engineering strategies for regenerative medicine. NPJ Regen Med 5:14. https://doi.org/10.1038/s41536-020-00099-8
Yoo KH, Thapa N, Chwae YJ, Yoon SH, Kim BJ, Lee JO, Jang YN, Kim J (2022) Transforming growth factor-β family and stem cell-derived exosome therapeutic treatment in osteoarthritis (review). Int J Mol Med. https://doi.org/10.3892/ijmm.2022.5118
Zeng L, Chen X, Zhang Q, Yu F, Li Y, Yao Y (2015) Redifferentiation of dedifferentiated chondrocytes in a novel three-dimensional microcavitary hydrogel. J Biomed Mater Res A 103(5):1693–1702. https://doi.org/10.1002/jbm.a.35309
Zeng L, Yao YC, Wang DA, Chen XF (2014) Effect of microcavitary alginate hydrogel with different pore sizes on chondrocyte culture for cartilage tissue engineering. Mat Sci Eng C-Mater 34:168–175. https://doi.org/10.1016/j.msec.2013.09.003
Zhang F, Yao Y, Su K, Fang Y, Citra F, Wang DA (2015) Co-transduction of lentiviral and adenoviral vectors for co-delivery of growth factor and shrna genes in mesenchymal stem cells-based chondrogenic system. J Tissue Eng Regen Med 9(9):1036–1045. https://doi.org/10.1002/term.1656
Zylinska B, Silmanowicz P, Sobczynska-Rak A, Jarosz L, Szponder T (2018) Treatment of articular cartilage defects: Focus on tissue engineering. In Vivo (athens, Greece) 32(6):1289–1300. https://doi.org/10.21873/invivo.11379
Acknowledgements
This work was supported by the Science and Technology Program of Guangzhou (Dengfeng Hospital 2022) (Grant number 202201020460), Guangzhou Medical University Research Ability Enhancement Project (2024) (Grant number 2024SRP072) and Guangzhou Health Science and Technology Project (Grant number 20241A010112).
Funding
The authors have not disclosed any funding The Science and Technology Program of Guangzhou (Dengfeng Hospital 2022),202201020460,Yongchang Yao,Guangzhou Medical University Research Ability Enhancement Project,2024,Yongchang Yao,Guangzhou Health Science and Technology Project,Grant number 20241A010112.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yao, Y., Chen, K., Pan, Q. et al. Redifferentiation of genetically modified dedifferentiated chondrocytes in a microcavitary hydrogel. Biotechnol Lett 46, 483–495 (2024). https://doi.org/10.1007/s10529-024-03475-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-024-03475-2