Abstract
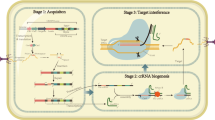
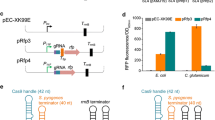
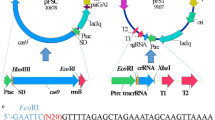
Clustered regularly interspaced short palindromic repeats-associated protein 9 (CRISPR/Cas9) system has been widely used in gene editing of various organisms. However, food-grade gene editing systems in lactic acid bacteria are still preliminary. Red/ET-dependent homologous recombination or CRISPR-based systems have been developed to gene editing in Lactococcus lactis, but these methods are overall inefficient. In the present study, a recombinant system based on CRISPR/Cas9 technology combined with Red/ET was developed using the plasmid pMG36e derived from Lactococcus lactis. Then, the developed recombinant system was applied to Lactococcus lactis. Knockout efficiency was significantly higher using the developed system (91%). In addition, this system showed the potential to be used as a high-throughput method for hierarchical screening. Finally, a gene-edited strain was obtained, and no antibiotics or exogenous genes were introduced using the developed gene editing system. Thus, the efficient system in lactic acid bacteria was constructed and optimized.




Similar content being viewed by others
References
Alvarez-Sieiro P, Montalbán-López M, Mu D, Kuipers OP (2016) Bacteriocins of lactic acid bacteria, extending the family. Appl Microbiol Biotechnol 100:2939–2951. https://doi.org/10.1007/s00253-016-7343-9
Aparicio T, Jensen SI, Nielsen AT, de Lorenzo V, Martínez-García E (2016) The Ssr protein (T1E_1405) from Pseudomonas putida DOT-T1E enables oligonucleotide-based recombineering in platform strain P. putida EM42. Biotechnol J 11:1309–1319. https://doi.org/10.1002/biot.201600317
Barrangou R, Fremaux C, Deveau H, Richards M, Boyaval P, Moineau S, Romero DA, Horvath P (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315:1709–1712. https://doi.org/10.1126/science.1138140
Bleckwedel J, Mohamed F, Mozzi F, Raya RR (2020) Major role of lactate dehydrogenase D-LDH1 for the synthesis of lactic acid in Fructobacillus tropaeoli CRL 2034. Appl Microbiol Biotechnol 104:7409–7426. https://doi.org/10.1007/s00253-020-10776-9
Boubakri H (2023) Recent progress in CRISPR/Cas9-based genome editing for enhancing plant disease resistance. Gene 866:147334. https://doi.org/10.1016/j.gene.2023.147334
Burnette WN (1981) “Western blotting”, electrophoretic transfer of proteins from sodium dodecyl sulfate–polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem 112:195–203. https://doi.org/10.1016/0003-2697(81)90281-5
Guo T, Kong J, Zhang L, Zhang C, Hu S (2012) Fine tuning of the lactate and diacetyl production through promoter engineering in Lactococcus lactis. PLoS ONE 7:e36296. https://doi.org/10.1371/journal.pone.0036296
Guo T, Xin Y, Zhang Y, Gu X, Kong J (2019) A rapid and versatile tool for genomic engineering in Lactococcus lactis. Microb Cell Fact 18:22. https://doi.org/10.1186/s12934-019-1075-3
Heigwer F, Kerr G, Boutros M (2014) E-CRISP, fast CRISPR target site identification. Nat Methods 11:122–123. https://doi.org/10.1038/nmeth.2812
Huang H, Song X, Yang S (2019) Development of a RecE/T-Assisted CRISPR-Cas9 Toolbox for Lactobacillus. Biotechnol J 14:e1800690. https://doi.org/10.1002/biot.201800690
Kachroo AH, Jayaram M, Rowley PA (2009) Metabolic engineering without plasmids. Nat Biotechnol 27:729–731. https://doi.org/10.1038/nbt0809-729
Kong LH, Xiong ZQ, Song X, Xia YJ, Ai LZ (2022) CRISPR/dCas9-based metabolic pathway engineering for the systematic optimization of exopolysaccharide biosynthesis in Streptococcus thermophilus. J Dairy Sci 105(8):6499–6512. https://doi.org/10.3168/jds.2021-21409
Leenay RT, Vento JM, Shah M, Martino ME, Leulier F, Beisel CL (2019) Genome editing with CRISPR-Cas9 in Lactobacillus plantarum revealed that editing outcomes can vary across strains and between methods. Biotechnol J 14:e1700583. https://doi.org/10.1002/biot.201700583
Li C, Du ZH, Qi SQ, Zhang X, Wang MS, Zhou YP, Lu HQ, Gu XX, Tian HT (2020) Food-grade expression of nattokinase in Lactobacillus delbrueckii subsp. bulgaricus and its thrombolytic activity in vitro. Biotechnol Lett 42(11):2179–2187. https://doi.org/10.1007/s10529-020-02974-2
Li Y, Lian D, Wang J, Zhao Y, Li Y, Liu G, Wu S, Deng S, Du X, Lian Z (2023) MDM2 antagonists promote CRISPR/Cas9-mediated precise genome editing in sheep primary cells. Mol Ther Nucleic Acids 31:309–323. https://doi.org/10.1016/j.omtn.2022.12.020
Linares DM, Kok J, Poolman B (2020) Genome sequences of Lactococcus lactis MG1363 (revised) and NZ9000 and comparative physiological studies. J Bacteriol 192:5806–5812. https://doi.org/10.1128/JB.00533-10
Mitsui R, Yamada R, Matsumoto T, Yoshihara S, Tokumoto H, Ogino H (2020) Construction of lactic acid-tolerant Saccharomyces cerevisiae by using CRISPR-Cas-mediated genome evolution for efficient D-lactic acid production. Appl Microbiol Biotechnol 104:9147–9158. https://doi.org/10.1007/s00253-020-10906-3
Miyoshi A, Poquet I, Azevedo V, Commissaire J, Bermudez-Humaran L, Domakova E, Le Loir Y, Oliveira SC, Gruss A, Langella P (2020) Controlled production of stable heterologous proteins in Lactococcus lactis. Appl Environ Microbiol 68:3141–3146. https://doi.org/10.1128/AEM.68.6.3141-3146.2002
Moritz CP (2020) 40 years Western blotting: a scientific birthday toast. J Proteomics 212:103575. https://doi.org/10.1016/j.jprot.2019.103575
Naz M, Benavides-Mendoza A, Tariq M, Zhou J, Wang J, Qi S, Dai Z, Du D (2023) CRISPR/Cas9 technology as an innovative approach to enhancing the phytoremediation: concepts and implications. J Environ Manage 323:116296. https://doi.org/10.1016/j.jenvman.2022.116296
Pšeničnik A, Reberšek R, Slemc L, Godec T, Kranjc L, Petković H (2022) Simple and reliable in situ CRISPR-Cas9 nuclease visualization tool is ensuring efficient editing in Streptomyces species. J Microbiol Methods 200:106545. https://doi.org/10.1016/j.mimet.2022.106545
Qi W, Li XX, Guo YH, Bao YZ, Wang N, Luo XG, Yu CD, Zhang TC (2020) Integrated metabonomic-proteomic analysis reveals the effect of glucose stress on metabolic adaptation of Lactococcus lactis ssp. lactis CICC23200. J Dairy Sci 103:7834–7850. https://doi.org/10.3168/jds.2019-17810
Roberts A, Barrangou R (2020) Applications of CRISPR-Cas systems in lactic acid bacteria. FEMS Microbiol Rev 44:523–537. https://doi.org/10.1093/femsre/fuaa016
Song X, Huang H, Xiong ZQ, Ai LZ, Yang S (2017) CRISPR-Cas 9(D10A) nickase-assisted genome editing in Lactobacillus casei. Appl Environ Microbiol 83:e01259-e1317. https://doi.org/10.1128/AEM.01259-17
Song X, Zhang XY, Xiong ZQ, Liu XX, Xia YJ, Wang SJ, Ai LZ (2020) CRISPR-Cas-mediated gene editing in lactic acid bacteria. Mol Biol Rep 47:8133–8144. https://doi.org/10.1007/s11033-020-05820-w
Song X, Liu L, Liu XX, Xiong ZQ, Xie CL, Wang SJ, Ai LZ (2021) Single-plasmid systems based on CRISPR-Cas9 for gene editing in Lactococcus lactis. J Dairy Sci 104:10576–10585. https://doi.org/10.3168/jds.2020-19901
Sun Z, Deng A, Hu T, Wu J, Sun Q, Bai H, Zhang G, Wen T (2015) A high-efficiency recombineering system with PCR-based ssDNA in Bacillus subtilis mediated by the native phage recombinase GP35. Appl Microbiol Biotechnol 99:5151–5162. https://doi.org/10.1007/s00253-015-6485-5
Szewczyk E, Nayak T, Oakley CE, Edgerton H, Xiong Y, Taheri-Talesh N, Osmani SA, Oakley BR (2006) Fusion PCR and gene targeting in Aspergillus nidulans. Nat Protoc 1:3111–3120. https://doi.org/10.1038/nprot.2006.405
van Kessel JC, Hatfull GF (2007) Recombineering in Mycobacterium tuberculosis. Nat Methods 4:147–152. https://doi.org/10.1038/nmeth996
Wang Q, Zhao Q, Liu Q, He X, Zhong Y, Qin Y, Gao L, Liu G, Qu Y (2021) CRISPR/Cas9-mediated genome editing in Penicillium oxalicum and Trichoderma reesei using 5S rRNA promoter-driven guide RNAs. Biotechnol Lett 43:1–8. https://doi.org/10.1007/s10529-020-03024-7
Xiong ZQ, Wei YY, Kong LH, Song X, Yi HX, Ai LZ (2020) Short communication: An inducible CRISPR/dCas9 gene repression system in Lactococcus lactis. J Dairy Sci 103:161–165. https://doi.org/10.3168/jds.2019-17346
Zhou D, Jiang ZN, Pang QX, Zhu Y, Wang Q, Qi QS (2019) CRISPR/Cas9-Assisted Seamless Genome Editing in Lactobacillus plantarum and its application in N-Acetylglucosamine production. Appl Environ Microbiol 85(21):e01367-e1419. https://doi.org/10.1128/AEM.01367-19
Acknowledgements
This research was funded by Opening Project of Key Laboratory of Safety Assessment of Genetically Modified Organism (Food Safety) from Ministry of Agriculture and Rural Affairs of the People's Republic of China (SAGM-Y-2017006), the Key Research and Development Program of Hebei Province (Grant No. 19227134D), the Science and Technology Research Projects of Colleges and Universities in Hebei province (Grant No. ZD2021059), and the Food Processing Discipline Group of Hebei Agricultural University (Grant No. 2021-08).
Funding
Fnding was provided by Opening Project of Key Laboratory of Safety Assessment of Genetically Modified Organism (Food Safety) from Ministry of Agriculture and Rural Affairs of the People's Republic of China (Grant No. SAGM-Y-2017006), the Key Research and Development Program of Hebei Province (Grant No. 19227134D), the Science and Technology Research Projects of Colleges and Universities in Hebei province (Grant No. ZD2021059), and the Food Processing Discipline Group of Hebei Agricultural University (Grant No. 2021-08).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, Y., Song, F., Yang, H. et al. Construction of a food-grade gene editing system based on CRISPR-Cas9 and its application in Lactococcus lactis NZ9000. Biotechnol Lett 45, 955–966 (2023). https://doi.org/10.1007/s10529-023-03398-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-023-03398-4