Abstract
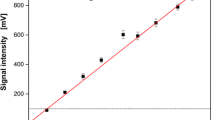
Accurate monitoring of dissolved oxygen (DO) is vital for aerobic fermentation process control. This work presents an autoclavable Micro-Dissolved oxygen Sensor (MDS) that can monitor real time DO. The proposed sensor is much cheaper to be manufactured (< $35) and can be adapted to varying measurement environments. An ultra-micropore matrix was created using femtosecond laser processing technology to reduce flow dependency of probe signals. The validity of the proposed DO sensor was verified by testing it under different DO levels. The result revealed consistency between the new designed sensor and a commercial DO sensor. The obtained sensitivity was− 7.93 μA·L·mg−1 (MDS with ultra-micropore matrix). Moreover, the MDS can function without an oxygen-permeable membrane and a solid electrolyte was used which reduced the response time (4.6 s). For real-time monitoring, the stability of the MDS was validated during a yeast batch fermentation carried out until 18 h.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- \(k_{DO}\) :
-
First order rate constant for DO probe model (s−1)
- \(c_{{l,O_{2} }}\) :
-
Oxygen concentration near the surface of DO sensor
- \(c_{{s,O_{2} }}\) :
-
Oxygen concentration inside the sensor
- \(c_{DO}\) :
-
Output of the DO sensor
- \(\tau_{e}\) :
-
Response time
- \(I_{relative}\) :
-
Relative value of current to characterize flow dependence
- \(Current_{{rpm_{i} }}\) :
-
The measured current of sensors under different rotation speeds
- \(Current_{{max \in rpm_{i} }}\) :
-
The maximum current of \(Current_{{rpm_{i} }}\)
- \(X_{{CO_{2} ,out}}\) :
-
Volume fraction of CO2 in off-gas
- \(X_{{CO_{2} ,in}}\) :
-
Volume fraction of CO2 in off-gas
- \(X_{{O_{2} ,in}}\) :
-
Volume fraction of O2 in air
- \(X_{{O_{2} ,out}}\) :
-
Volume fraction of O2 in air
- \(F_{in} \;{\text{and}}\;F_{out}\) :
-
Volumetric flow rate of inlet air and outlet off-gas under standard condition
- \(V_{broth}\) :
-
Volume of the broth in bioreactor
References
Andrijana B, Hilde L et al (2016) A flexible well-mixed milliliter-scale reactor with high oxygen transfer rate for microbial cultivations. Chem Eng J 303:655–666. https://doi.org/10.1016/j.cej.2016.05.117
Anthony KA, Wilson H et al (2016) 3D-printed microfluidics. Angew Chem Int Ed 55(12):3862–3881. https://doi.org/10.1002/anie.201504382
Buchenauer A, Hofmann MC et al (2009) Micro-bioreactors for fed-batch fermentations with integrated online monitoring and microfluidic devices. Biosens Bioelectron 24(5):1411–1416. https://doi.org/10.1016/j.bios.2008.08.043
de Jongea LP, Heijnena JJ, van Guilk WM (2014) Reconstruction of the oxygen uptake and carbon dioxide evolution rates of microbial cultures at near-neutral pH during highly dynamic conditions. Biochem Eng J 84:42–54
Diana V, Gertan A. van Z et al (2004) Analysis of in vivo kinetics of glycolysis in aerobic Saccharomyces cerevisiae by application of glucose and ethanol pulses. Biotechnol Bioeng 88(2):157–167. https://doi.org/10.1002/bit.20235
Diano A, Bekker-Jensen S et al (2006) Polyol synthesis in Aspergillus niger: influence of oxygen availability, carbon and nitrogen sources on the metabolism. Biotechnol Bioeng 94(5):899–908. https://doi.org/10.1002/bit.20915
Eric EK, van Leeuwen M et al (2008) A integrated electrochemical sensor array for on-line monitoring of yeast fermentations. Biotechnol Bioeng 99:884–892
Gabi G, Ralf H et al (2011) A new microfluidic concept for parallel operated milliliter-scale stirred tank bioreactors. Biotechnol Prog 27(3):684–690. https://doi.org/10.1002/btpr.570
Gregor W, Aleksandr O, Perdo F C (2019) Functional 3D printing for microfluidic chips. Adv Mater Technol 4(10):1900275. https://doi.org/10.1002/admt.201900275
Haiyuan G, Michael S et al (2020) Applications of off-gas mass spectrometry in fed-batch mammalian cell culture. Bioprocess Biosyst Eng 43(3):483–493. https://doi.org/10.1007/s00449-019-02242-2
Hyunjin L, Hyoungmin K et al (2017) Fabrication and characterization of micro dissolved oxygen sensor activated on demand using electrolysis. Sens Actuators B Chem 241:923–930. https://doi.org/10.1016/j.snb.2016.10.145
Jia Q, Wenbo L et al (2020) A microfluidic mixer of high throughput fabricated in glass using femtosecond laser micromachining combined with glass bonding. Micromachines 11(2):213
Jianye X, Benjamin JS et al (2022) Proteome allocations change linearly with the specific growth rate of Saccharomyces cerevisiae under glucose limitation. Nat Commun 13(1):2819. https://doi.org/10.1038/s41467-022-30513-2
Justin MS, Luke AB et al (2020) Electrochemical dissolved oxygen sensor-integrated platform for wireless in situ bioprocess monitoring. Sen Actuators b: Chem. https://doi.org/10.1016/j.snb.2020.128381
Jungil P, Jong-hyeon C et al (2007) Microfabirated clark-type sensor for measuing dissolved oxygen. IEEE. https://doi.org/10.1109/ICSENS.2007.4388677
Kazuyoshi I, Wataru W et al (2006) Ultrafast processes for bulk modification of transparent materials. MRS Bull 31(8):620–625. https://doi.org/10.1557/mrs2006.159
Koji S, Ya C (2014) Ultrafast lasers—reliable tools for advanced materials processing. Light Sci Appl 3(4):e149–e149. https://doi.org/10.1038/lsa.2014.30
Lee JH, Kim TJ (2004) A study on oxygen permeability of polypropylene membranes and their temperature dependency using medical oxygen sensor. Ksbb J 19(1):62–66
Mário APN, Pedro CBF, Maria HLR (2013) Microtiter plates versus stirred mini-bioreactors in biocatalysis: a scalable approach. Bioresour Technol 136:30–40. https://doi.org/10.1016/j.biortech.2013.02.057
Michel VL, Eric EK et al (2010) Aerobic batch cultivation in micro bioreactor with integrated electrochemical sensor array. Biotechnol Prog 26(1):293–300. https://doi.org/10.1002/btpr.315
Milica R, Jos Malda et al (2006) Oxygen gradients correlate with cell density and cell viability in engineered cardiac tissue. Biotechnol Bioeng 93(2):332–343
Nulee J, Mungyu L et al (2020) Behavior of CO-water mass transfer coefficient in membrane sparger-integrated bubble column for synthesis gas fermentation. Bioresour Technol 311:123594. https://doi.org/10.1016/j.biortech.2020.123594
Peng L, Shuai W et al (2021) Dynamic response of Aspergillus niger to periodical glucose pulse stimuli in chemostat cultures. Biotechnol Bioeng 118(6):2265–2282. https://doi.org/10.1002/bit.27739
Rafael RG, Eric M (2008) Femtosecond laser micromachining in transparent materials. Nat Photonics 2(4):219–225. https://doi.org/10.1038/nphoton.2008.47
Schmiderder A, Severien T et al (2015) A novel milliliter-scale chemostat system for parallel cultivation of microorganisms in stirred-tank bioreactors. J Biotechnol 210:19–24. https://doi.org/10.1016/j.jbiotec.2015.06.402
Seyed Ali MS, Fabio de F et al (2016) A microfluidic optical platform for real-time monitoring of pH and oxygen in microfluidic bioreactors and organ-on-chip devices. Biomicrofluidics 10(4):044111. https://doi.org/10.1063/1.4955155
Tobias K, Konstantin S, Elmar H (2013) A system of miniaturized stirred bioreactors for parallel continuous cultivation of yeast with online measurement of dissolved oxygen and off-gas. Biotechnol Bioeng 110(2):535–542. https://doi.org/10.1002/bit.24633
Tom F, Bland F (2008) Oxygen and the spatial structure of microbial communities. Biol Rev Camb Philos Soc 83(4):553–569. https://doi.org/10.1111/j.1469-185X.2008.00054.x
Wei C, Peng W et al (2019) High-resolution femtosecond laser 3D micromachining using low-NA focusing systems (Vol. 10908): SPIE
Xiao L, Cong H et al (2019) High cell density culture of baker’s yeast FX-2 based on pH-stat coupling with respiratory quotient. Biotechnol Appl Biochem 66(3):389–397. https://doi.org/10.1002/bab.1735
Xiaolong L, Jian X et al (2019) Polarization-insensitive space-selective etching in fused silica induced by picosecond laser irradiation. Appl Surface Sci 485:188–193. https://doi.org/10.1016/j.apsusc.2019.04.211
Yang L, Jiangxin S et al (2012) Rapid prototyping of three-dimensional microfluidic mixers in glass by femtosecond laser direct writing. Lab Chip 12(4):746–749. https://doi.org/10.1039/c2lc21015k
Zhen G, Huixin L et al (2018) A thumb-size electrochemical system for portable sensors. Analyst 143(12):2760–2764. https://doi.org/10.1039/c8an00645h
Zhiqiang X, Meijin G et al (2008) Real-time viable-cell mass monitoring in high-cell-density fed-batch glutathione fermentation by Saccharomyces cerevisiae T65 in industrial complex medium. J Biosci Bioeng 105(4):409–413. https://doi.org/10.1263/jbb.105.409
Zijie L, Jian X et al (2020) Freeform microfluidic networks encapsulated in laser-printed 3D macroscale glass objects. Adv Mater Technol 5(2):1900989. https://doi.org/10.1002/admt.201900989
Supporting Information
Supplementary Figure 1—Validation of the Nafion MDS under (a) 2.5-10 % Citric acid; (b) 2.5-10 % Lactic acid; (c) 2.5-10 % Malic acid; (d) 2.5-10 % Ethanol; (e) pH 0-14; (f) 15-55 °C.
Supplementary Figure 2—Linear characterizations of sensors every 5 times autoclaves for a total of 50 times autoclaves.
Funding
This work was financially supported by the National Key Research and Development Program of China (Grant No. 2021YFC2101100).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Specific contributions are as follows: Conceptualization and writing—review and editing, XJ and ZY; methodology, FM and CW; writing—original draft preparation, FM; software and hardware, GZ and WH. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fan, M., Gu, Z., Chen, W. et al. Micro-electrochemical DO sensor with ultra-micropore matrix fabricated with femtosecond laser processing successfully applied in on-line DO monitoring for yeast culture. Biotechnol Lett 45, 449–461 (2023). https://doi.org/10.1007/s10529-023-03348-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-023-03348-0