Abstract
Objective
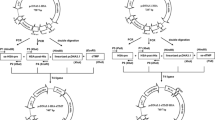
Thrombopoietin mimetic peptide (TMP), an analog of natural thrombopoietin, can be used to treat primary immune thrombocytopenia. However, the short half-life of TMP limits its application in clinics. The present study aimed to improve the stability and biological activity of TMP in vivo via genetic fusion to the albumin-binding protein domain (ABD).
Results
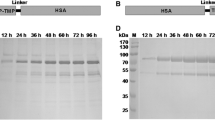
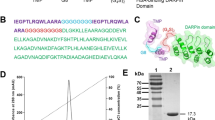
TMP dimer was genetically fused to the N-terminal or C-terminal of ABD, denoted as TMP-TMP-ABD and ABD-TMP-TMP. A Trx-tag was used to improve the fusion proteins’ expression levels effectively. ABD-fusion TMP proteins were produced in Escherichia coli and purified by Ni2+-NTA and SP ion exchange column. Albumin binding studies in vitro showed that the fusion proteins could effectively bind to serum albumin to extend their half-lives. The fusion proteins effectively induced platelet proliferation in healthy mice, and the platelet count was increased by more than 2.3-fold compared with the control group. The increased platelet count induced by the fusion proteins lasted 12 days compared with the control group. The increasing trend was maintained for 6 days before a decline occurred after the last injection in the fusion-protein-treated mice group.
Conclusions
ABD can effectively improve the stability and pharmacological activity of TMP by binding to serum albumin, and the ABD-fusion TMP protein can promote platelet formation in vivo.




Similar content being viewed by others
References
Arai R, Ueda H, Kitayama A et al (2001) Design of the linkers which effectively separate domains of a bifunctional fusion protein. Protein Eng 14:529–532
Chen X, Zaro JL, Shen WC (2013) Fusion protein linkers: property, design and functionality. Adv Drug Deliv Rev 65:1357–1369
Chen YZ, Tan SM, Yang F et al (2017) Soluble expression and purification of a functional harpin protein in Escherichia coli. Process Biochem 57:200–206
Cwirla SE, Balasubramanian P, Duffin DJ et al (1997) Peptide agonist of the thrombopoietin receptor as potent as the natural cytokine. Science 276:1696–1699
Feese MD, Tamada T, Kato Y et al (2004) Structure of the receptor-binding domain of human thrombopoietin determined by complexation with a neutralizing antibody fragment. Proc Natl Acad Sci U S A 101:1816–1821
Frederickson S, Renshaw MW, Lin B et al (2006) A rationally designed agonist antibody fragment that functionally mimics thrombopoietin. Proc Natl Acad Sci U S A 103:14307–14312
Gapizov SS, Petrovskaya LE, Shingarova LN et al (2019) Fusion with an albumin-binding domain improves pharmacokinetics of an αvβ3-integrin binding fibronectin scaffold protein. Biotechnol Appl Biochem 66:617–625
Ghavim M, Abnous K, Arasteh F et al (2017) High level expression of recombinant human growth hormone in Escherichia coli: crucial role of translation initiation region. Res Pharm Sci 12:168–175
Kratz F (2008) Albumin as a drug carrier: Design of prodrugs, drug conjugates and nanoparticles. J Control Release 132:171–183
Kuter DJ (2013) The biology of thrombopoietin and thrombopoietin receptor agonists. Int J Hematol 98:10–23
Lamiable A, Thevenet P, Rey J et al (2016) PEP-FOLD3: faster de novo structure prediction for linear peptides in solution and in complex. Nucleic Acids Res 44:W449-454
Lok S, Kaushansky K, Holly RD et al (1994) cloning and expression of murine thrombopoietin cdna and stimulation of platelet production in-vivo. Nature 369:565–568
Marti-Renom MA, Stuart AC, Fiser A et al (2000) Comparative protein structure modeling of genes and genomes. Annu Rev Biophys Biomol Struct 29:291–325
Molineux G (2011) The development of romiplostim for patients with immune thrombocytopenia. Ann N Y Acad Sci 1222:55–63
Mutalik VK, Guimaraes JC, Cambray G et al (2013) Precise and reliable gene expression via standard transcription and translation initiation elements. Nat Methods 10(4):354–360
Neunert C, Noroozi N, Norman G et al (2015) Severe bleeding events in adults and children with primary immune thrombocytopenia: a systematic review. J Thromb Haemost 13:457–464
Nilvebrant J, Hober S (2013) The albumin-binding domain as a scaffold for protein engineering. Comput Struct Biotechnol J 6:e201303009–e201303009
Nomura S (2016) Advances in diagnosis and treatments for immune thrombocytopenia. Clin Med Insights Blood Disord 9:15–22
Parret AHA, Besir H, Meijers R (2016) Critical reflections on synthetic gene design for recombinant protein expression. Curr Opin Struct Biol 38:155–162
Pollaro L, Heinis C (2010) Strategies to prolong the plasma residence time of peptide drugs. Medchemcomm 1:319–324
Portielje JE, Westendorp RG, Kluin-Nelemans HC, Brand A (2001) Morbidity and mortality in adults with idiopathic thrombocytopenic purpura. Blood 97:2549–2554
Provan D, Newland AC (2015) Current management of primary immune thrombocytopenia. Adv Ther 32:875–887
Ru Y, Zhi D, Guo D et al (2016) Expression and bioactivity of recombinant human serum albumin and dTMP fusion proteins in CHO cells. Appl Microbiol Biotechnol 100:7565–7575
de Serres M, Yeager RL, Dillberger JE et al (1999) Pharmacokinetics and hematological effects of the PEGylated thrombopoietin peptide mimetic GW395058 in rats and monkeys after intravenous or subcutaneous administration. Stem Cells 17:316–326
Sockolosky JT, Szoka FC (2015) The neonatal Fc receptor, FcRn, as a target for drug delivery and therapy. Adv Drug Deliv Rev 91:109–124
Tan H, Su W, Zhang W et al (2021) Albumin-binding domain extends half-life of glucagon-like peptide-1. Eur J Pharmacol 890:173650
Wang L, Gao Z, Chen XP et al (2016a) Efficacy and safety of thrombopoietin receptor agonists in patients with primary immune thrombocytopenia: a systematic review and meta-analysis. Scientific Rep. https://doi.org/10.1038/srep39003
Wang MZ, Zhi DJ, Xian J et al (2016b) Functional expression of human serum albumin-tandem thrombopoietin mimetic peptide fusion protein as a novel thrombopoietin analog in Pichia pastoris. Biotech Lett 38:779–785
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Funding
This work was supported by Key Research Foundation of Hwa Mei Hospital, University of Chinese Academy of Sciences, China (Grant No. 2019HMKY17); Key Medical Subjects of Joint Construction Between Provinces and Cites (Infectious Diseases), China (Grant No. 2016017); Key Laboratory of Diagnosis and Treatment of Digestive System Tumors of Zhejiang Province (Grant No. 2019E10020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, D., Gao, G., Zhu, B. et al. Improving pharmacological activities of thrombopoietin mimetic peptide by genetic fusion to albumin-binding domain. Biotechnol Lett 45, 439–448 (2023). https://doi.org/10.1007/s10529-023-03345-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-023-03345-3