Abstract
Objectives
Ovarian cancer is one of the most fatal gynecological malignancies. It is emergently needed to select a novel molecular fragment as a targeting element for the future development of molecular imaging diagnosis and targeting chemotherapy to ovarian cancer.
Results
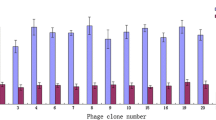
After five rounds of biopanning, a total of 44 positive phage clones were selected from final phage displayed peptide library. Nine consensus sequences were found based on the assay of sequencing results, then one clone of each consensus group was characterized and identified further by immunofluorescence assay. The result showed the phage clone R20 presents best targeting capacity. Then we synthesized peptide (OSP2) clone R20 displayed, it was characterized with high specificity and sensitivity binding to human ovarian cancer by a tissue chip assay. The target of OSP2 was predicted and docked as human carbonic anhydrase XII (CA12), an important protein usually deregulated in cancer.
Conclusions
Taken together, OSP2 and its target indicate a novel investigation way in future to develop novel agent or drug delivery formulation for molecular imaging diagnosis and targeting chemotherapy of ovarian cancer.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Allemani C, Weir HK, Carreira H, Harewood R, Spika D, Wang XS (2015) Global surveillance of cancer survival 1995–2009: analysis of individual data for 25,676,887 patients from 279 population based registries in 67 countries (CONCORD-2). Lancet 385(9972):977–1010. https://doi.org/10.1016/S0140-6736(14)62038-9
Bozkurt MF, Virgolini I, Balogova S, Beheshti M, Rubello D, Decristoforo C, Ambrosini V, Kjaer A, Delgado-Bolton R, Kunikowska J, Oyen WJG, Chiti A, Giammarile F, Sundin A, Fanti S (2017) Guideline for PET/CT imaging of neuroendocrine neoplasms with 68Ga-DOTA-conjugated somatostatin receptor targeting peptides and 18F-DOPA. Eur J Nucl Med Mol Imaging 44(9):1588–1601. https://doi.org/10.1007/s00259-017-3728-y
Franke CM, Gu VW, Grimm BG, Cassady VC, White JR, Weigel RJ, Kulak MV (2020) TFAP2C regulates carbonic anhydrase XII in human breast cancer. Oncogene 39(6):1290–1301. https://doi.org/10.1038/s41388-019-1062-5
Gu G, Gao X, Hu Q, Kang T, Liu Z, Jiang M, Miao D, Song Q, Yao L, Tu Y, Pang Z, Chen H, Jiang X, Chen J (2013) The influence of the penetrating peptide iRGD on the effect of paclitaxel-loaded MT1-AF7p-conjugated nanoparticles on glioma cells. Biomaterials 34(21):5138–5148. https://doi.org/10.1016/j.biomaterials.2013.03.036
Herrera FG, Irving M, Kandalaft LE, Coukos G (2019) Rational combinations of immunotherapy with radiotherapy in ovarian cancer. Lancet Oncol 20(8):e417–e433. https://doi.org/10.1016/S1470-2045(19)30401-2
Hilchie AL, Doucette CD, Pinto DM, Patrzykat A, Douglas S, Hoskin DW (2011) Pleurocidin-family cationic antimicrobial peptides are cytolytic for breast carcinoma cells and prevent growth of tumor xenografts. Breast Cancer Res 13(5):R102. https://doi.org/10.1186/bcr3043
Hou L, Zhu D, Liang Y, Tian X, Li L, Wang P, Zhu L, Weng X, Wang Y, Li Y, Wu T, Wang J, Meng X (2018) Identification of a specific peptide binding to colon cancer cells from a phage-displayed peptide library. Br J Cancer 118(1):79–87. https://doi.org/10.1038/bjc.2017.366
Lheureux S, Gourley C, Vergote I, Oza AM (2019) Epithelial ovarian cancer. Lancet 393(10177):1240–1253. https://doi.org/10.1016/S0140-6736(18)32552-2
Li G, Chen TW, Nickel AC, Muhammad S, Steiger HJ, Tzaridis T, Hänggi D, Zeidler R, Zhang W, Kahlert UD (2021) Carbonic anhydrase XII is a clinically significant, molecular tumor-subtype specific therapeutic target in glioma with the potential to combat invasion of brain tumor cells. Onco Targets Ther 14:1707–1718. https://doi.org/10.2147/OTT.S300623
Lomelino C, McKenna R (2016) Carbonic anhydrase inhibitors: a review on the progress of patent literature (2011–2016). Expert Opin Ther Pat 26(8):947–956. https://doi.org/10.1080/13543776.2016.1203904
Ma Z, Qin H, Chen H, Yang H, Xu J, Yang S, Hu J, Xing D (2017) Phage display-derived oligopeptide-functionalized probes for in vivo specific photoacoustic imaging of osteosarcoma. Nanomedicine 13(1):111–121. https://doi.org/10.1016/j.nano.2016.09.002
Menon U, Karpinskyj C, Gentry-Maharaj A (2018) Ovarian cancer prevention and screening. Obstet Gynecol 131(3):909–927. https://doi.org/10.1097/AOG.0000000000002580
Matsuo AL, Tanaka AS, Juliano MA, Rodrigues EG, Travassos LR (2010) A novel melanoma-targeting peptide screened by phage display exhibits antitumor activity. J Mol Med (berl) 88(12):1255–1264. https://doi.org/10.1007/s00109-010-0671-9
Nebgen DR, Lu KH, Bast RC Jr (2019) Novel approaches to ovarian cancer screening. Curr Oncol Rep 21(8):75. https://doi.org/10.1007/s11912-019-0816-0
Peres LC, Cushing-Haugen KL, Köbel M, Harris HR, Berchuck A, Rossing MA, Schildkraut JM, Doherty JA (2019) Invasive epithelial ovarian cancer survival by histotype and disease stage. J Natl Cancer Inst 111(1):60–68. https://doi.org/10.1093/jnci/djy071
Pham TC, Jayasinghe MK, Pham TT, Yang Y, Wei L, Usman WM, Chen H, Pirisinu M, Gong J, Kim S, Peng B, Wang W, Chan C, Ma V, Nguyen NTH, Kappei D, Nguyen XH, Cho WC, Shi J, Le MTN (2021) Covalent conjugation of extracellular vesicles with peptides and nanobodies for targeted therapeutic delivery. J Extracell Vesicles 10(4):e12057. https://doi.org/10.1002/jev2.12057
Seward SM, Winer I (2015) Primary debulking surgery and neoadjuvant chemotherapy in the treatment of advanced epithelial ovarian carcinoma. Cancer Metastasis Rev 34(1):5–10. https://doi.org/10.1007/s10555-014-9536-y
Supuran CT (2018) Carbonic anhydrase inhibitors as emerging agents for the treatment and imaging of hypoxic tumors. Expert Opin Investig Drugs 27(12):963–970. https://doi.org/10.1080/13543784.2018.1548608
Tian R, Zhu L, Qin Z, Wang G, Wang J, Zhang H (2019) Glypican-3 (GPC3) targeted Fe3O4 core/Au shell nanocomplex for fluorescence/MRI/photoacoustic imaging-guided tumor photothermal therapy. Biomater Sci 7(12):5258–5269. https://doi.org/10.1039/c9bm01248f
Torre LA, Trabert B, DeSantis CE, Miller KD, Samimi G, Runowicz CD, Gaudet MM, Jemal A, Siegel RL (2018) Ovarian cancer statistics, 2018. CA Cancer J Clin 68(4):284–296. https://doi.org/10.3322/caac.21456
Vázquez-Ríos AJ, Molina-Crespo Á, Bouzo BL, López-López R, Moreno-Bueno G, de la Fuente M (2019) Exosome-mimetic nanoplatforms for targeted cancer drug delivery. J Nanobiotechnol 17(1):85. https://doi.org/10.1186/s12951-019-0517-8
Vergote I, Harter P, Chiva L (2019) Hyperthermic intraperitoneal chemotherapy does not improve survival in advanced ovarian cancer. Cancer 125(24):4594–4597. https://doi.org/10.1002/cncr.32496
Waheed A, Sly WS (2017) Carbonic anhydrase XII functions in health and disease. Gene 623:33–40. https://doi.org/10.1016/j.gene.2017.04.027
Wang S, Blois A, El Rayes T, Liu JF, Hirsch MS, Gravdal K, Palakurthi S, Bielenberg DR, Akslen LA, Drapkin R, Mittal V, Watnick RS (2016) Development of a prosaposin-derived therapeutic cyclic peptide that targets ovarian cancer via the tumor microenvironment. Sci Transl Med 8(329):329–334. https://doi.org/10.1126/scitranslmed.aad5653
Xiao L, Hou Y, He H, Cheng S, Hou Y, Jin H, Song X, Nie G, Hou Y (2020) A novel targeted delivery system for drug-resistant hepatocellular carcinoma therapy. Nanoscale 12(32):17029–17044. https://doi.org/10.1039/d0nr01908a
Zhai W, Zhou X, Zhai M, Li W, Ran Y, Sun Y, Du J, Zhao W, Xing L, Qi Y, Gao Y (2021) Blocking of the PD-1/PD-L1 interaction by a novel cyclic peptide inhibitor for cancer immunotherapy. Sci China Life Sci 64(4):548–562. https://doi.org/10.1007/s11427-020-1740-8
Funding
This work was supported by the Shaanxi Province Natural Science Foundation (2022JQ-219) and Guangxi Innovation Driven Development Major Project (Guike AA20302013).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Ethical approval
This article does not contain any study with human participant or tissue from hospital.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Q., Chen, L., Jia, C. et al. Selection and identification of a specific peptide binding to ovarian cancer cells from a phage-displayed peptide library. Biotechnol Lett 44, 951–960 (2022). https://doi.org/10.1007/s10529-022-03263-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-022-03263-w