Abstract
Objectives
The bioaccumulation of keratinous wastes from poultry and dairy industries poses a danger of instability to the biosphere due to resistance to common proteolysis and as such, microbial- and enzyme-mediated biodegradation are discussed.
Results
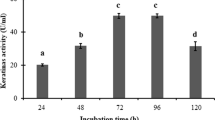
In submerged fermentation medium, Proteus vulgaris EMB-14 utilized and efficiently degraded feather, fur and scales by secreting exogenous keratinase. The keratinase was purified 14-fold as a monomeric 49 kDa by DEAE-Sephadex A-50 anion exchange and Sephadex G-100 size-exclusion chromatography. It exhibited optimum activity at pH 9.0 and 60 °C and was alkaline thermostable (pH 7.0–11.0), retaining 87% of initial activity after 1 h pre-incubation at 60 °C. The Km and Vmax of the keratinase with keratin azure were respectively 0.283 mg/mL and 0.241 U/mL/min. Activity of P. vulgaris keratinase was stimulated by Ca2+, Mg2+, Zn2+, Na+ and maintained in the presence of some denaturing agents, except β-mercaptoethanol while Cu2+ and Pb2+ showed competitive and non-competitive inhibition with Ki 6.5 mM and 17.5 mM, respectively.
Conclusion
This purified P. vulgaris keratinase could be surveyed for the biotechnological transformation of bioorganic keratinous wastes into valuable products such as soluble peptides, cosmetics and biodegradable thermoplastics.







Similar content being viewed by others
References
Akhter M, Marzan LW, Akter Y, Shimizu K (2020) Microbial bioremediation of feather waste for keratinase production: an outstanding solution for leather dehairing in tanneries. Microbiol Insights 13:1–12
Allure N, Madhusudhan DN, Agsar D (2015) Detection of keratinolytic Actinobacteria and evaluation of bioprocess for production of alkaline keratinase. Int J Curr Microbiol Appl Sci 4(7):907–918
Anwar A, Saleemuddin M (2000) Alkaline protease from Spilosoma obliqua: potential application in bioformulations. Biotechnol App Biochem 31:85–89
Ayodeji AO, Bamidele OS, Kolawole AO, Ajele JO (2017) Physicochemical and kinetic properties of a high salt tolerant Aspergillus flavus glucoamylase. Biocatal Agric Biotechnol 9:35–40
Bhange K, Chaturvedi V, Bhatt R (2016) Simultaneous production of detergent stable keratinolytic protease, amylase and biosurfactant by Bacillus subtilis PF1 using agro industrial waste. Biotechnol Rep 10:94–104
Bouacem K, Bouanane-Darenfed A, Jaouadi NZ, Joseph M, Hacene H, Ollivier B, Fardeau ML, Bejar S, Jaouadi B (2016) Novel serine keratinase from Caldicoprobacter algeriensis exhibiting outstanding hide dehairing abilities. Int J Biol Macromol 86:321–328
de Oliveira CT, Pellenz L, Pereira JQ, Brandelli A, Daroit DJ (2016) Screening of bacteria for protease production and feather degradation. Waste Biomass Valoriz 7:447–453
de Paiva DP, de Oliveira SS, Mazotto AM, Vermelho AB, de Oliveira SS (2018) Keratinolytic activity of Bacillus subtilis LFB-FIOCRUZ 1266 enhanced by whole-cell mutagenesis. 3 Biotech 9(1):2. https://doi.org/10.1007/s13205-018-1527-1
Dudhgara PR, Sunil B, Anjana G (2015) Hide dehairing and laundry detergent compatibility testing of thermostable and solvents tolerant alkaline protease from hot spring isolate Bacillus cohnii U3. Online J Biol Sci 15(3):152–161. https://doi.org/10.3844/ojbsci.2015.152.161
Fakhfakh N, Kanoun S, Manni L, Nasri M (2009) Production and biochemical and molecular characterization of a keratinolytic serine protease from chicken feather-degrading Bacillus licheniformis RPk. Can J Microbiol 55:427–436
Fellahi S, Chibani A, Feuk-Lagerstedt E, Taherzadeh MJ (2016) Identification of two new keratinolytic proteases from a Bacillus pumilus strain using protein analysis and gene sequencing. AMB Exp 6:42. https://doi.org/10.1186/s13568-016-0213-0
Ghosh A, Chakrabarti K, Chattopadhyay D (2008) Degradation of a raw feather by a novel high molecular weight extracellular protease from newly isolated Bacillus cereus DCUW. J Ind Microbiol Biotechnol 35(8):825–834. https://doi.org/10.1007/s10295-008-0354-5
Gupta R, Ramnani P (2006) Microbial keratinases and their prospective applications: an overview. Appl Microbiol Biotechnol 70:21–33. https://doi.org/10.1007/s00253-005-0239-8
Gupta S, Nigam A, Singh R (2015) Purification and characterization of a Bacillus subtilis keratinase and its prospective application in feed industry. Acta Biol Szeged 59(2):197–204
Haddar A, Kamoun AS, Zouari NF, Hmidet N, Nasri M (2010) Characterization of detergent stable and feather degrading serine proteases from Bacillus mojavensis A21. Biochem Eng J 51:53–63. https://doi.org/10.1016/j.bej.2010.05.002
Hadj-Ali NE, Agrebi R, Ghorbel-Frikha B, Sellami-Kamoun A, Kanoun S, Nasri M (2007) Biochemical and molecular characterization of a detergent stable alkaline serine-protease from a newly isolated Bacillus licheniformis NH1. Enzyme Microb Technol 40:515–523. https://doi.org/10.1016/j.enzmictec.2006.05.007
Han HY, Zou HC, Jeon JY, Wang YJ, Xu WA, Yang JM, Park YD (2007) The inhibition kinetics and thermodynamic changes of Tyrosinase via the zinc ion. Biochim Biophys Acta 1774(7):822–827. https://doi.org/10.1016/j.bbapap.2007.04.011
Hutadilok-Towatana N, Painupong A, Suntinanalert P (1999) Purification and characterization of an extracellular protease from alkaliphilic and thermophilic Bacillus sp PS719. J Biosci Bioeng 87(5):581–587. https://doi.org/10.1016/s1389-1723(99)80118-2
Jadhav RS, Karad DD, Kulakrni SW (2016) Isolation, identification and characterization of keratinolytic Streptomyces coelicoflavus. Int J Curr Microbiol Appl Sci 5(7):153–163
Jaouadi B, Abdelmalek B, Fodil D, Ferradji FZ, Rekik H, Zarai N, Bejar S (2010) Purification and characterization of a thermostable keratinolytic serine alkaline proteinase from Streptomyces sp. strain AB1 with high stability in organic solvent. Bioresour Technol 101:8361–8369
Jin HS, Park SY, Kim K, Lee YJ, Nam GW, Kang NJ, Lee DW (2017) Development of a keratinase activity assay using recombinant chicken feather keratin substrates. PLoS ONE 12(2):e0172712. https://doi.org/10.1371/journal.pone.0172712
Kobayashi T, Hakamada Y, Hitomi J, Koike K, Ito S (1996) Purification of alkaline proteases from a Bacillus strain and their possible interrelationship. Appl Microbiol Biotechnol 45:63–71
Laemmli UK (1970) Cleavage of structural proteins during assembly of head of bacteriophage T4. Nature 227:680–685
Lange L, Huang Y, Busk PK (2016) Microbial decomposition of keratin in nature-a new hypothesis of industrial relevance. Appl Microbiol Biotechnol 100(5):2083–2096. https://doi.org/10.1007/s00253-015-7262-1
Lee YJ, Kim JH, Kim HK, Lee JS (2004) Production and characterization of keratinase from Paracoccuc sp. WJ-98. Biotechnol Bioprocess Eng 9:17–22
Li J, Shi PJ, Han XY, Meng K, Yang PL, Wang YR, Luo HY, Wu NF, Yao B, Fan YL (2007) Functional expression of the keratinolytic serine protease gene sfp2 from Streptomyces fradiae var. k11 in Pichia pastoris. Protein Exp Purif 54:79–86
Lin X, Lee SW, Bae HD, Shelford JA, Cheng KJ (2001) Comparison of two feather-degrading Bacillus licheniformis strains. Asian-Australas J Ani Sci 14(12):1769–1774
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56(3):658–666. https://doi.org/10.1021/ja01318a036
Patil U, Mokashe N, Chaudhari A (2016) Detergent-compatible, organic solvent-tolerant alkaline protease from Bacillus circulans MTCC 7942: purification and characterization. Prep Biochem Biotechnol 46(1):56–64
Pawar VA, Prajapati AS, Akhani RC, Patel DH, Subramanian RB (2018) Molecular and biochemical characterization of a thermostable keratinase from Bacillus altitudinis RBDV1. 3 Biotech 8:107. https://doi.org/10.1007/s13205-018-1130-5
Prakasham RS, Rao CS, Sarma PN (2006) Green gram husk-an inexpensive substrate for alkaline protease production by Bacillus sp. in solid-state fermentation. Bioresour Technol 97(13):1449–1454
Radha S, Gunasekaran P (2009) Purification and characterization of keratinase from recombinant Pichia and Bacillus strains. Protein Expr Purif 64(1):24–31
Rao MB, Tanksale AM, Ghatge MS, Deshpande VV (1998) Molecular and biotechnological aspects of microbial proteases. Microbiol Mol Biol 62(3):597–635
Riffel A, Lucas F, Heeb P, Brandelli A (2003) Characterization of new keratinolytic bacterium that completely degrades native feather keratin. Arch Microbiol 179(4):258–265
Riffel A, Brandelli A, Bellato CM, Souza GHMF, Eberlin MN, Tavares FCA (2007) Purification and characterization of a keratinolytic metalloprotease from Chryseobacterium sp. kr6. J Biotechnol 128(3):693–703
Saibabu V, Niyonzima FN, More SS (2013) Isolation, partial purification and characterization of keratinase from Bacillus megaterium. Int Res J Biol Sci 2(2):13–20
Sana B, Ghosh D, Saha M, Mukherjee J (2006) Purification and characterization of a salt, solvent, detergent and bleach tolerant protease from a new gamma-Proteobacterium isolated from the marine environment of the Sundarbans. Process Biochem 41(1):208–215
Sharma R, Devi S (2018) Versatility and commercial status of microbial keratinases: a review. Rev Environ Sci Biotechnol 17(1):19–45. https://doi.org/10.1007/s11157-017-9454-x
Silveira ST, Gemelli S, Segalin J, Brandellli A (2012) Immobilization of keratinolytic metalloprotease from Chryseobacterium sp. strain kr6 on glutaraldehyde-activated chitosan. J Microbiol Biotechnol 22:818–825
Sousa F, Jus S, Erbel A, Kokol V, Cavaco-Paulo A, Gubitz GM (2007) A novel metalloprotease from Bacillus cereus for protein fibre processing. Enzyme Microb Technol 40(7):1772–1781
Subba Rao C, Sathish T, Ravichandra P, Prakasham RS (2009) Characterization of thermo and detergent stable serine-protease from isolated Bacillus circulans and evaluation of ecofriendly applications. Process Biochem 44:262–268
Syed DG, Lee JC, Li W, Kim C, Agasar D (2009) Production, characterization and application of keratinase from Streptomyces gulbargensis. Bioresour Technol 100:1868–1871
Tiwary E, Gupta R (2010) Medium optimization for a novel 58 kDa dimeric keratinase from Bacillus licheniformis ER-15: biochemical characterization and application in feather degradation and dehairing of hides. Bioresour Technol 101:6103–6110
Uttatree S, Charoenpanich J (2018) Purification and characterization of a harsh conditions-resistant protease from a new strain of Staphylococcus saprophyticus. Agric Nat Resour 52:16–23
Walsh R, Martin E, Darvesh S (2007) A versatile equation to describe reversible enzyme inhibition and activation kinetics: modelling 2-galactosidasse and butyrylcholinesterase. Biochim Biophys Acta 1770(5):733–746
Wang X, Parsons CM (1997) Effect of processing systems on protein quality of feather meals and hog hair meals. Poult Sci 76(3):491–496. https://doi.org/10.1093/ps/76.3.491
Wang P, Wang Q, Cui L, Gao M, Fan X (2011) The combined use of cutinase, keratinase and protease treatments for wool bio-antifelting. Fibers Polym 12:760–764
Yamarama S, Morita Y, Hasan Q, Rao SR, Murakami Y, Yokoyama K, Tamiya E (2002) Characterization of a new keratin-degrading bacterium isolated from deer fur. J Biosci Bioeng 93:595–600
Yusuf I, Ahmad SA, Phang LY, Yasid NA, Shukor MY (2019) Effective production of keratinase by gellan gum-immobilised Alcaligenes sp. AQ05–001 using heavy metal-free and polluted feather wastes as substrates. 3 Biotech 9:32. https://doi.org/10.1007/s13205-018-1555-x
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of experiments; MOB and AOA performed the experiments; OSB and AOA analysed the data; OSB and JOA contributed to writing the manuscript and all authors approved the final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest as touching the publication of this research article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Babalola, M.O., Ayodeji, A.O., Bamidele, O.S. et al. Biochemical characterization of a surfactant-stable keratinase purified from Proteus vulgaris EMB-14 grown on low-cost feather meal. Biotechnol Lett 42, 2673–2683 (2020). https://doi.org/10.1007/s10529-020-02976-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02976-0