Abstract
Objective
To investigate the functions of eIF3b in chronic myelogenous leukemia (CML).
Methods
The expression of eIF3b was inhibited by transfecting aspecifically designed shRNA into the CML cell lines of TK-6 and K562. The CCK8 assay was conducted to determine cell viability, and flow cytometry was used to examine the change in the cell cycle and cell apoptosis. RNAsequencing was applied to screen the candidate targets of eIF3b to identify the underlying mechanisms of eIF3b.An in vivo tumour xenograft mouse model was established by injecting shRNA transfected cells into the NCG mice. The tumour size and body weight of mice were monitored every other day. The mice were sacrificed 2 weeks after the tumour cell injection. The expression of eIF3b and target genes in the tumour tissues were determined by immunohistochemical staining and Western blotting.
Results
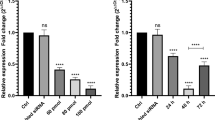
The group with inhibited expression of eIF3b led to about 50% lower cell viability compared with that of the control group (P < 0.05). Flow cytometry suggested that the percentage of increase in apoptotic cells was eight times higher than those in control group for TK-6 and K562 cells (P < 0.05). However, the difference between the cell amounts in the S phase for the experiment and control groups was not significant. After RNAsequencing and further validation via qPCR, C3G was screened as the potential target of eIF3b involved in the cell proliferation and apoptosis of CML cell lines. Subsequent in vivo analysis proved that the inhibition of eIF3b suppressed tumour formation and decreased C3G expression, thereby indicating that C3G was the potential target of eIF3b.
Conclusion
eIF3b is correlated with the cell proliferation and cell apoptosis of CML. Moreover, eIF3b regulation most probably occurs via regulating the expression of C3G.






Similar content being viewed by others
References
Bhat M, Robichaud N, Hulea L, Sonenberg N, Pelletier J, Topisirovic I (2015) Targeting the translation machinery in cancer. Nat Rev Drug Discov 14:261
Burke BA, Carroll M (2010) BCR–ABL: a multi-faceted promoter of DNA mutation in chronic myelogeneous leukemia. Leukemia 24:1105–1112
Choi YJ, Lee YS, Lee HW, Shim DM, Seo SW (2017) Silencing of translation initiation factor eIF3b promotes apoptosis in osteosarcoma cells. Bone Jt Res 6:186–193
Ghosh D, Dey SK, Saha C (2014) Protective effect of black tea extract during chemotherapeutic drug induced oxidative damage on normal lymphocytes in comparison with cancerous K562 cells. Int J Sci Eng Res 5:437–447
Gregor T, Bosakova MK, Nita A, Abraham SP, Fafilek B, Cernohorsky NH, Rynes J, Foldynova-Trantirkova S, Zackova D, Mayer J, Trantirek L, Krejci P (2019) Elucidation of protein interactions necessary for the maintenance of the BCR-ABL signaling complex. Cell Mol Life Sci. https://doi.org/10.1007/s00018-019-03397-7
Gu Y, Chen T, Meng Z, Gan Y, Xu X, Lou G, Li H, Gan X, Zhou H, Tang J, Xu G, Huang L, Zhang X, Fang Y, Wang K, Zheng S, Huang W, Xu R (2012) CaMKII gamma, a critical regulator of CML stem/progenitor cells, is a target of the natural product berbamine. Blood 120:4829–4839
Jackson RJ, Hellen CU, Pestova TV (2010) The mechanism of eukaryotic translation initiation and principles of its regulation. Nat Rev Mol Cell Biol 11:113
Jiang X, Tang X, Zhang P, Liu G, Guo H (2014) Cyanidin-3-O-beta-glucoside protects primary mouse hepatocytes against high glucose-induced apoptosis by modulating mitochondrial dysfunction and the PI3K/Akt pathway. Biochem Pharmacol 90:135–144
Massaro F, Colafigli G, Molica M, Breccia M (2018) Novel tyrosine-kinase inhibitors for the treatment of chronic myeloid leukemia: safety and efficacy. Expert Rev Hematol 11:301–306
Mi S, Lee X, Hu Y, Ji B, Shao Z, Yang W, Huang G, Walus L, Rhodes K, Gong BJ (2011) Death receptor 6 negatively regulates oligodendrocyte survival, maturation and myelination. Nat Med 17:816–821
Nikolaev A, McLaughlin T, O’Leary DDM, Tessier-Lavigne M (2009) APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature 457:981–989
Pan G, Bauer JH, Haridas V, Wang S, Liu D, Yu G, Vincenz C, Aggarwal BB, Ni J, Dixit VM (1998) Identification and functional characterization of DR6, a novel death domain-containing TNF receptor. FEBS Lett 431:351–356
Pettersson F, Del Rincon SV, Miller W Jr (2014) Eukaryotic translation initiation factor 4E as a novel therapeutic target in hematological malignancies and beyond. Expert Opin Ther Targets 18:1035–1048
Priego N, Arechederra M, Sequera C, Bragado P, Vazquez-Carballo A, Gutierrez-Uzquiza A, Martin-Granado V, Ventura JJ, Kazanietz MG, Guerrero C, Porras A (2016) C3G knock-down enhances migration and invasion by increasing Rap1-mediated p38alpha activation, while it impairs tumor growth through p38alpha-independent mechanisms. Oncotarget 7:45060–45078
Sequera C, Manzano S, Guerrero C, Porras A (2018) How Rap and its GEFs control liver physiology and cancer development C3G alterations in human hepatocarcinoma. Hepat Oncol 5:HEP05
Silvera D, Formenti SC, Schneider RJ (2010) Translational control in cancer. Nat Rev Cancer 10:254–266
Sridharan S, Robeson M, Bastihalli-Tukaramrao D, Howard CM, Subramaniyan B, Tilley AMC, Tiwari AK, Raman D (2019) Targeting of the eukaryotic translation initiation factor 4A against breast cancer stemness. Front Oncol 9:1311
Tian Y, Zhao K, Yuan L, Li J, Feng S, Feng Y, Fang Z, Li H, Deng R (2018) EIF3B correlates with advanced disease stages and poor prognosis, and it promotes proliferation and inhibits apoptosis in non-small cell lung cancer. Cancer Biomark 23:291–300
Voss AK, Krebs DL, Thomas T (2006) C3G regulates the size of the cerebral cortex neural precursor population. EMBO J 25:3652–3663
Wang H, Ru Y, Sanchez-Carbayo M, Wang X, Kieft JS, Theodorescu D (2013) Translation initiation factor eIF3b expression in human cancer and its role in tumor growth and lung colonization. Clin Cancer Res 19:2850–2860
Wang L, Wen X, Luan F, Fu T, Gao C, Du H, Guo T, Han J, Huangfu L, Cheng X, Ji J (2019) EIF3B is associated with poor outcomes in gastric cancer patients and promotes cancer progression via the PI3K/AKT/mTOR signaling pathway. Cancer Manag Res 11:7877–7891
Xu F, Zhang S, Liu Z, Gu J, Li Y, Wang L, Mao W, Zhu Q, Shou H, Ge D, Lu C (2019) TEX9 and eIF3b functionally synergize to promote the progression of esophageal squamous cell carcinoma. BMC Cancer 19:875
Zeng L, Li T, Xu DC, Liu J, Mao G, Cui MZ, Fu X, Xu X (2012) Death receptor 6 induces apoptosis not through type I or type II pathways, but via a unique mitochondria-dependent pathway by interacting with Bax protein. J Biol Chem 287:29125–29133
Zheng X, Gao L, Wang BT, Shen P, Yuan XF, Zhang LQ, Yang L, Zhang DP, Zhang Q, Wang XM (2019) Overexpression of EIF5A2 is associated with poor survival and aggressive tumor biology in gallbladder cancer. Histol Histopathol. https://doi.org/10.14670/HH-18-186
Funding
This study was supported by Natural Science Research Project of Colleges and Universities in Anhui Province (KJ2017A263).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that that there are no conflict of interest.
Research involving human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, L., Wei, Z., Chang, X. et al. eIF3b regulates the cell proliferation and apoptosis processes in chronic myelogenous leukemia cell lines via regulating the expression of C3G. Biotechnol Lett 42, 1275–1286 (2020). https://doi.org/10.1007/s10529-020-02878-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02878-1