Abstract
Objective
To evaluate the induction of monoterpenoid indole alkaloids (MIA) and phenolic compound production by yeast extract (YE) and its relationship with defense responses in Uncaria tomentosa (Rubiaceae) root cultures.
Results
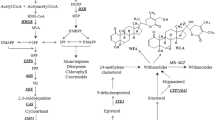
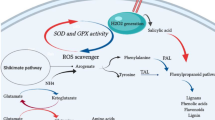
Root cultures were elicited by YE at three concentrations. The 0.5 mg YE ml−1 treatment did not affect cell viability but increased the hydrogen peroxide concentration by 5.7 times; guaiacol peroxidase activity by twofold; and the glucoindole alkaloid 3α-dihydrocadambine (DHC) content by 2.6 times (to 825.3 ± 27.3 μg g−1). This treatment did not affect the contents of monoterpenoid oxindole alkaloids or chlorogenic acids. In response to 0.5 mg YE ml−1 treatment, the transcript levels of MIA biosynthetic genes, TDC and LAMT, increased 5.4 and 1.9-fold, respectively, that of SGD decreased by 32%, and that of STR did not change. The transcript levels of genes related to phenolic compounds, PAL, CHS and HQT, increased by 1.7, 7.7, and 1.2-fold, respectively. Notably, the transcript levels of Prx1 and Prx encoding class III peroxidases increased by 1.4 and 2.5-fold.
Conclusion
The YE elicitor induced an antioxidant defense response, increased the transcript levels of genes encoding enzymes related to strictosidine biosynthesis precursors and class III peroxidases, and decreased the transcript level of SGD. Thus, YE could stimulate antifungal DHC production in root cultures of U. tomentosa.





Similar content being viewed by others
References
Almagro L, Gómez Ros LV, Belchi-Navarro S, Bru R, Ros Barceló A, Pedreño MA (2009) Class III peroxidases in plant defence reactions. J Exp Bot 60:377–390. https://doi.org/10.1093/jxb/ern277
Brown RT, Duckworth DM, Santos KAM (1991) Biogenetically patterned synthesis of cadambine. Tetrahedron Lett 32:1987–1991
Costa MM, Hilliou F, Duarte P, Pereira LG, Almeida I et al (2008) Molecular cloning and characterization of a vacuolar class III peroxidase involved in the metabolism of anticancer alkaloids in Catharanthus roseus. Plant Physiol 146:403–417. https://doi.org/10.1104/pp.107.107060
Demidchik V (2015) Mechanisms of oxidative stress in plants: from classical chemistry to cell biology. Environ Exp Bot 109:212–228. https://doi.org/10.1016/j.envexpbot.2014.06.021
Dwight Z, Palais R, Wittwer CT (2011) uMELT: prediction of high-resolution melting curves and dynamic melting profiles of PCR products in a rich web application. Bioinformatics 27:1019–1020. https://doi.org/10.1093/bioinformatics/btr065
Endo K, Oshima Y, Kikuchi H, Koshihara Y, Hikino H (1983) Hypotensive principles of Uncaria hooks. Planta Med 49:188–190. https://doi.org/10.1055/s-2007-969846
Ferreres F, Figueiredo R, Bettencourt S, Carqueijeiro I, Oliveira J, Gil-Izquierdo A et al (2011) Identification of phenolic compounds in isolated vacuoles of the medicinal plant Catharanthus roseus and their interaction with vacuolar class III peroxidase: an H2O2 affair? J Exp Bot 62:2841–2854. https://doi.org/10.1093/jxb/erq458
Guirimand G, Courdavault V, Lanoue A et al (2010) Strictosidine activation in Apocynaceae: towards a “nuclear time bomb”? BMC Plant Biol 10:182. https://doi.org/10.1186/1471-2229-10-182
Guo Q, Ma X, Wei S, Qui D, Wilson IW et al (2014) De novo transcriptome sequencing and digital gene expression analysis predict biosynthetic pathway of rhynchophylline and isorhynchophylline from Uncaria rhynchophylla, a non-model plant with potent anti-alzheimer’s properties. BMC Genomics 15:1–16. https://doi.org/10.1186/1471-2164-15-676
Hanafy MS, Matter MA, Rady MR (2017) Production of indole alkaloids in Catharanthus roseus L. hairy root cultures. In: Malik S (ed) Production of plant derived natural compounds through hairy hoot culture. Springer, Cham, pp 89–116. https://doi.org/10.1007/978-3-319-69769-7_5
Huerta-Heredia AA, Marín-López R, Ponce-Noyola T, Cerda-García-Rojas CM, Trejo-Tapia G, Ramos-Valdivia AC (2009) Oxidative stress induces alkaloid production in Uncaria tomentosa root and cell cultures in bioreactors. Eng Life Sci 3:211–221. https://doi.org/10.1002/elsc.200800118
Jaggi M, Kumar S, Sinha AK (2011) Overexpression of an apoplastic peroxidase gene CrPrx in transgenic hairy root lines of Catharanthus roseus. Appl Microbiol Biotechnol 90:1005–1016. https://doi.org/10.1007/s00253-011-3131-8
Kärkönen A, Kuchitsu K (2015) Reactive oxygen species in cell wall metabolism and development in plants. Phytochemistry 112:22–32. https://doi.org/10.1016/j.phytochem.2014.09.016
Kohda H, Namera A, Koyama A, Yamasaki K, Tani T (1996) Indole alkaloid production in callus cultures of Uncaria rhynchophylla (Miq.) Miquel. Chem Pharm Bull 44:352–357
Kumar S, Dutta A, Sinha AK, Sen J (2007) Cloning, characterization and localization of a novel basic peroxidase gene from Catharanthus roseus. FEBS J 274:1290–1303. https://doi.org/10.1111/j.1742-4658.2007.05677.x
Kumar S, Jaggi M, Sinha AK (2012) Ectopic overexpression of vacuolar and apoplastic Catharanthus roseus peroxidases confers differential tolerance to salt and dehydration stress in transgenic tobacco. Protoplasma 249:423–432. https://doi.org/10.1007/s00709-011-0294-1
Liu J, Cai J, Wang R, Yang S (2017) Transcriptional regulation and transport of terpenoid indole alkaloid in Catharanthus roseus: exploration of new research directions. Int J Mol Sci 18:53. https://doi.org/10.3390/ijms18010053
Luna-Palencia G, Huerta-Heredia A, Cerda-García-Rojas CM, Ramos-Valdivia AC (2013) Differential alkaloid profile in Uncaria tomentosa micropropagated plantlets and root cultures. Biotechnol Lett 35:791–797. https://doi.org/10.1007/s10529-012-1128-8
MacRae E (2007) Extraction of plant RNA BT. In: Hilario E, Mackay J (eds) Protocols for nucleic acid analysis by nonradioactive probes. Humana Press, Totowa, pp 15–24. https://doi.org/10.1385/1-59745-229-7:15
Mondolot L, La Fisca P, Buatois B, Talansier E et al (2006) Evolution in caffeoylquinic acid content and histolocalization during Coffea canephora leaf development. Ann Bot 98:33–40. https://doi.org/10.1093/aob/mcl080
Naoumkina MA, He X, Dixon RA (2008) Elicitor-induced transcription factors for metabolic reprogramming of secondary metabolism in Medicago truncatula. BMC Plant Biol 8:132–146. https://doi.org/10.1186/1471-2229-8-132
Pan Q, Wang Q, Yuan F, Xing S, Zhao J et al (2012) Overexpression of ORCA3 and G10H in Catharanthus roseus plants regulated alkaloid biosynthesis and metabolism revealed by NMR-metabolomics. PLoS ONE 7(8):e43038. https://doi.org/10.1371/journal.pone.0043038
Patel DA, Darji VC, Bariya AH, Patel KR, Sonpal RN (2011) Evaluation of antifungal activity of Neolamarckia cadamba (roxb.) bosser leaf and bark extract. Int Res J Pharm 2:192–193
Pauw B, Duijin B, Kjine JW, Memelink J (2004) Activation of the oxidative burst by yeast elicitor in Catharanthus roseus cells occurs independently of the activation of genes involved in alkaloid biosynthesis. Plant Mol Biol 55:797–805. https://doi.org/10.1007/s11103-004-1968-2
Pavei C, Kaiser S, Borré GL, Ortega GG (2010) Validation of a LC method for polyphenols assay in Cat´s claw (Uncaria tomentosa). J Liq Chromatogr Relat Technol 33:1551–1561. https://doi.org/10.1080/10826076.2010.503753
Putter J (1974) Peroxidases. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Verlag Chemie, Weinhan, pp 685–690
Ramos-Valdivia AC, Huerta-Heredia AA, Trejo-Tapia G, Cerda- García-Rojas CM (2012) Secondary metabolites as non-enzymatic plant protectors from oxidative stress. In: Anjum NA, Umar S, Ahmad A (eds) Oxidative stress in plants: causes, consequences and tolerance. International publishing house, New Delhi, pp 413–441
Sergeiv I, Aleixieva V, Karanov E (1997) Effect of spermine, atrazine and combination between them on some endogenous protective systems and stress markers in plants. Proc Bulg Acad Sci 51:121–124
Sottomayor M, Duarte P, Figueiredo R, Ros Barceló A (2008) A vacuolar class III peroxidase and the metabolism of anticancer indole alkaloids in Catharanthus roseus. Can peroxidases, secondary metabolites and arabinogalactan proteins be partners in microcompartmentation of cellular reactions? Plant Signal Behav 3:899–901. https://doi.org/10.4161/psb.3.10.6576
Sun J, Peebles CAM (2016) Engineering overexpression of ORCA3 and strictosidine glucosidase in Catharanthus roseus hairy roots increases alkaloid production. Protoplasma 253:1255–1264. https://doi.org/10.1007/s00709-015-0881-7
Suttipanta N, Pattanaik S, Kulshrestha M, Patra B, Singh SK, Yuan L (2011) The transcription factor CrWRKY1 positively regulates the terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Plant Physiol 157:2081–2093. https://doi.org/10.1104/pp.111.181834
Szabó LF (2008) Rigorous biogenetic network for a group of indole alkaloids derived from strictosidine. Molecules 13:1875–1896. https://doi.org/10.3390/molecules13081875
Takahama U (2004) Oxidation of vacuolar and apoplastic phenolic substrate by peroxidase: physiological significance of the oxidation reactions. Phytochem Rev 3:207–219. https://doi.org/10.1023/B:PHYT.0000047805.08470.e3
Vera-Reyes I, Huerta-Heredia AA, Ponce-Noyola T, Flores-Sánchez IJ, Esparza-García F, Cerda-García-Rojas CM, Trejo-Tapia G, Ramos-Valdivia AC (2013) Strictosidine-related enzymes involved in the alkaloid biosynthesis of Uncaria tomentosa root cultures grown under oxidative stress. Biotechnol Prog 29:621–630. https://doi.org/10.1002/btpr.1723
Vera-Reyes I, Huerta-Heredia AA, Ponce-Noyola T, Cerda-García-Rojas C, Trejo-Tapia G, Ramos-Valdivia AC (2015) Monoterpenoid indole alkaloids and phenols are required antioxidants in glutathione depleted Uncaria tomentosa root cultures. Front Environ Sci 3:1–11. https://doi.org/10.3389/fenvs.2015.00027
Wungsintaweekul J, Choo-Malee J, Charoonratana T, Keawpradub N (2012) Methyl jasmonate and yeast extract stimulate mitragynine production in Mitragyna speciosa (Roxb.) Korth. shoot culture. Biotechnol Lett 34:1945–1950. https://doi.org/10.1007/s10529-012-0968-6
Yue L, Twell D, Kuang Y, Liao J, Zhou X (2017) Transcriptome analysis of Hamelia patens (Rubiaceae) anthers reveals candidate genes for tapetum and pollen wall development. Front Plant Sci. 7:1–20. https://doi.org/10.3389/fpls.2016.01991
Zhang Q, Zhao JJ, Xu J, Feng F, Qu W (2015) Medicinal uses, phytochemistry and pharmacology of the genus Uncaria. J Ethnopharmacol 173:48–80. https://doi.org/10.1016/j.jep.2015.06.011
Zhao S, Fernald RD (2005) Comprehensive algorithm for quantitative real-time polymerase chain reaction. J Comput Biol 12:1047–1064. https://doi.org/10.1089/cmb.2005.12.1047
Acknowledgements
This work was supported by CONACYT (National Council for Science and Technology, Mexico, Grant No. 222097, doctoral fellowship 423774 to L.J.C.H., and postdoctoral fellowship to E.B.S.G.) and SIP-IPN (National Polytechnic Institute, Grant No. 20181377). The authors thank V. Medina-Pérez for assistance with chromatographic analysis and C. Fontaine for technical support. L.J.C.H. acknowledge to Unitrópico for allowing its academic leave.
Supporting information
Supplementary Fig. 1—Viability of Uncaria tomentosa roots after 96 h of elicitation with yeast extract at different concentrations.
Supplementary Fig. 2—HPLC chromatogram λ = 244 nm for the alkaloids present in Uncaria tomentosa root culture, after 96 h of elicitation with yeast extract (0.5 mg ml−1).
Supplementary Fig. 3—HPLC chromatogram λ = 325 nm for phenolic compounds present in Uncaria tomentosa root culture, after 96 h of elicitation with yeast extract (0.5 mg ml−1).
Supplementary Figs. 4–12—Sequences selected for primer design of biosynthetic genes.
Supplementary Table 1—Primers used in RT-qPCR.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Correa-Higuera, L.J., Sepúlveda-García, E.B., Ponce-Noyola, T. et al. Glucoindole alkaloid accumulation induced by yeast extract in Uncaria tomentosa root cultures is involved in defense response. Biotechnol Lett 41, 1233–1244 (2019). https://doi.org/10.1007/s10529-019-02714-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02714-1