Abstract
Background
Previously we reported that natural nut lipids were necessary for sensitization and that natural killer T cells (NKTs) must play a critical role in the development of food allergic responses. A major bottleneck in further understanding the interaction of nut lipids with the cells of the human immune system is the lack of well-characterized lipid responsive human cell lines.
Objective
In the present study, we engineered human T cell receptor (TCR) sequences TRAV10 and TRBV25 responsive to α-GalCer into a stable murine iNKT hybridoma and surrogate human T cell lines.
Results
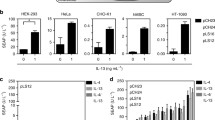
The murine hybridoma system has shown to be problematic. To overcome this limitation, the expression of human TCR α/β sequences has been achieved driven by a bidirectional promoter on a plasmids or a lentivirus system, employing stable DC cell lines as lipid presenting cells, and a stable T cell line as a surrogate system. Further, a commercial human Jurkat T cell line containing an inducible secreted luciferase reporter construct was shown to be functional and can be used for a transient expression of human TCRs in a lipid screening program. The transfection efficiencies were improved using the lentivirus polycistronic constructs containing the P2A sequence in a TCR αβ/γδ null cell line (Jurkat 76).
Conclusions
The results suggest that the mis-pairing of the endogenous α/β TCR during ER folding in the presence of the new human TCR sequences could be impairing the functionality of the TCR lipid receptors. The surrogate systems presented here are important first steps in the establishment of human cell-specific lipid responsive libraries for the study of natural lipid substances.





Similar content being viewed by others
References
Ahmadi M, King JW, Xue SA et al (2011) CD3 limits the efficacy of TCR gene therapy in vivo. Blood 118:3528–3537. https://doi.org/10.1182/blood-2011-04-346338
Bai L, Constantinides MG, Thomas SY et al (2012) Distinct APCs explain the cytokine bias of α-galactosylceramide variants in vivo. J Immunol 188:3053–3061. https://doi.org/10.4049/jimmunol.1102414
Brigl M, van den Elzen P, Chen X et al (2006) Conserved and heterogeneous lipid antigen specificities of CD1d-restricted NKT cell receptors. J Immunol 162:161–167. https://doi.org/10.4049/jimmunol.176.6.3625
de Jong A (2015) Activation of human T cells by CD1 and self-lipids. Immunol Rev 267:16–29. https://doi.org/10.1111/imr.12322
Dearman RJ, Alcocer MJC, Kimber I (2007) Influence of plant lipids on immune responses in mice to the major Brazil nut allergen Ber e 1. Clin Exp Allergy 37:582–591. https://doi.org/10.1111/j.1365-2222.2007.02689.x
Dowds CM, Kornell S-C, Blumberg RS, Zeissig S (2014) Lipid antigens in immunity. J Biol Chem 395:61–81. https://doi.org/10.1515/hsz-2013-0220.Lipid
Ghumra A, Alcocer M (2017) The use of a semi-automated system to measure mouse natural killer T (NKT) cell activation by lipid-loaded dendritic cells. Methods in molecular biology. Human Press, Clifton, pp 249–262
Godfrey DI, Uldrich AP, Mccluskey J et al (2015) The burgeoning family of unconventional T cells. Nat Immunol 16:1114–1123. https://doi.org/10.1038/ni.3298
Guo XJ, Dash P, Calverley M et al (2016) Rapid cloning, expression, and functional characterization of paired αβ and γδ T-cell receptor chains from single-cell analysis. Mol Ther 3:15054. https://doi.org/10.1038/mtm.2015.54
Heemskerk MHM, Hoogeboom M, De Paus RA et al (2003) Redirection of antileukemic reactivity of peripheral T lymphocytes using genetransfer of minor histocompatibility antigen HA-2-specific T-cell receptor complexes expressing a conserved alpha joining region. Blood 102:3530–3540. https://doi.org/10.1182/blood-2003-05-1524
Jordan-Williams KL, Poston S, Taparowsky EJ (2013) BATF regulates the development and function of IL-17 producing iNKT cells. BMC Immunol 14:16. https://doi.org/10.1186/1471-2172-14-16
Kean DE, Goodridge HS, McGuinness S et al (2006) Differential polarization of immune responses by plant 2S seed albumins, Ber e 1, and SFA8. J Immunol 177:1561–1566. https://doi.org/10.4049/jimmunol.177.3.1561
Kim PJ, Pai S-Y, Brigl M et al (2006) GATA-3 Regulates the development and function of invariant NKT cells. J Immunol 177:6650–6659. https://doi.org/10.4049/jimmunol.177.10.6650
Kim HS, Kim HS, Lee CW, Chung DH (2010) T cell Ig domain and mucin domain 1 engagement on invariant NKT cells in the presence of TCR stimulation enhances IL-4 production but inhibits IFN-γ production. J Immunol 184:4095–4106. https://doi.org/10.4049/jimmunol.0901991
Knies D, Klobuch S, Xue S-A et al (2016) An optimized single chain TCR scaffold relying on the assembly with the native CD3-complex prevents residual mispairing with endogenous TCRs in human T-cells. Oncotarget 7:21199–21221. https://doi.org/10.18632/oncotarget.8385
Lantz O, Bendelac A (1994) An invariant T cell receptor α chain is used by a unique subset of major histocompatibility complex class I-specific CD4 + and CD4-8- T cells in mice and humans. J Exp Med 180:1097. https://doi.org/10.1084/jem.180.3.1097
Liu Z, Chen O, Wall JBJ et al (2017) Systematic comparison of 2A peptides for cloning multi-genes in a polycistronic vector. Sci Rep 7:2193. https://doi.org/10.1038/s41598-017-02460-2
Masterson AJ, Sombroek CC, De Gruijl TD et al (2002) MUTZ-3, a human cell line model for the cytokine-induced differentiation of dendritic cells from CD34 + precursors. Blood 100:701–703. https://doi.org/10.1182/blood.V100.2.701
Mirotti L, Florsheim E, Rundqvist L et al (2013) Lipids are required for the development of Brazil nut allergy: the role of mouse and human iNKT cells. Allergy Eur J Allergy Clin Immunol 68:74–83. https://doi.org/10.1111/all.12057
Ning J, Morgan D, Pamphilon D (2011) A rapid culture technique produces functional dendritic-like cells from human acute myeloid leukemia cell lines. J Biomed Biotechnol 2011:172965. https://doi.org/10.1155/2011/172965
O’Konek JJ, Terabe M, Berzofsky JA (2012) The role of NKT cells in the immune regulation of neoplastic disease. Innate immune regulation and cancer immunotherapy. Springer, New York, pp 7–21
Roth H, Magg V, Uch F et al (2017) Flavivirus infection uncouples translation suppression from cellular stress responses. MBio. https://doi.org/10.1128/mBio.02150-16
Rundqvist L, Tengel T, Zdunek J et al (2012) Solution structure, copper binding and backbone dynamics of recombinant Ber e 1-the major allergen from Brazil Nut. PLoS ONE 7:46435. https://doi.org/10.1371/journal.pone.0046435
Sommermeyer D, Neudorfer J, Weinhold M et al (2006) Designer T cells by T cell receptor replacement. Eur J Immunol 36:3052–3059. https://doi.org/10.1002/eji.200636539
Taniguchi M, Seino KI, Nakayama T (2003) The NKT cell system: bridging innate and acquired immunity. Nat Immunol 4:1164–1165. https://doi.org/10.1038/ni1203-1164
Thomas S, Stauss HJ, Morris EC (2010) Molecular immunology lessons from therapeutic T-cell receptor gene transfer. Immunology 129:170–177. https://doi.org/10.1111/j.1365-2567.2009.03227.x
Zhong S, Malecek K, Perez-Garcia A, Krogsgaard M (2010) Retroviral transduction of T-cell receptors in mouse T-cells. J Vis Exp 44:2307. https://doi.org/10.3791/2307
Zhou Q, Buchholz CJ (2013) Cell type specific gene delivery by lentiviral vectors: new options in immunotherapy. Oncoimmunology 2:22566. https://doi.org/10.4161/onci.22566
Acknowledgements
We would like also to thank Dr Cinzia Allegrucci, School of Veterinary, University of Nottingham for support on the lentivirus work. A.F. has been financially supported by a Research Grant (534231) from Unilever UK Central resources, hence the execution of the work has been partially funded and the final article for publication reviewed by Unilever.
Funding
This study was partially funded by a Research Grant (534231) from Unilever UK Central resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Author A. Ghumra received research grant from Unilever UK Central resources, hence the execution of the work has been partially funded and the final article for publication reviewed by Unilever.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, R., Pscheid, R., Ghumra, A. et al. Towards a surrogate system to express human lipid binding TCRs. Biotechnol Lett 41, 1095–1104 (2019). https://doi.org/10.1007/s10529-019-02713-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02713-2