Abstract
Objective
The objective of this work was to isolate bacteria from Red Sea invertebrates, determine their antimicrobial activity, and screen for the biosynthetic gene clusters [polyketides (PKs) and nonribosomal peptides (NRPs)] which could be involved in the production of bioactive secondary metabolites.
Result
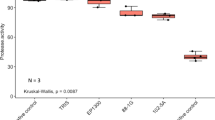
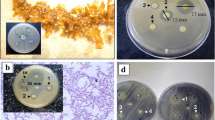
Eleven different samples of marine invertebrates’ were collected from Egypt’s Red Sea (El-Tor-Sharm El-Sheikh and Hurghada) by scuba diving, and a total 80 isolates of the associated microorganisms were obtained from the cultivation on six different cultural medium. Seven isolates of them showed an antimicrobial activity against five pathogenic reference strains, while the most active antimicrobial agent was isolate number HFF-8 which was 99% identical to Bacillus amyloliquefaciens. HFF-8’s extract showed positive results against Gram negative bacteria, Gram positive bacteria and yeast. Moreover, the isolates gave positive bands when screened for the presence of PK synthase (PKS) I and II and NRP synthetase (NRPS) I and II biosynthetic genes, those biosynthetic fragments when cloned and sequenced were primitively predicted as biosynthetic fragments for kirromycin and leinamycin production by NaPDoS program with 56 and 55%, respectively.
Conclusion
The Red Sea can provide a sustainable solution to combat bacterial resistance. The contribution of this work is that B. amyloliquefaciens was isolated from Heteroxenia fuscescens, Red Sea, Egypt. Moreover, the bacterial extract showed a broad spectrum with a potent antimicrobial activity.




Similar content being viewed by others
References
Abdelmohsen UR, Balasubramanian S, Oelschlaeger TA, Grkovic T, Pham NB, Quinn RJ, Hentschel U (2017) Potential of marine natural products against drug-resistant fungal, viral, and parasitic infections. Lancet Infect Dis 17:e30–e41
Abou-Elela GM, Abd-Elnaby H, Ibrahim HA, Okbah M (2009) Marine natural products and their potential applications as anti-infective agents. World Appl Sci J 7:872–880
Aboul-Ela HM, Shreadah MA, Abdel-Monem NM, Yakout GA, van Soest RW (2012) Isolation, cytotoxic activity and phylogenetic analysis of Bacillus sp. bacteria associated with the Red Sea sponge Amphimedon ochracea. Adv Biosci Biotechnol 3:815
Al-Amoudi S et al (2016) Bioprospecting Red Sea coastal ecosystems for culturable microorganisms and their antimicrobial potential. Mar Drugs 14:165
Amos GC et al (2015) Designing and implementing an assay for the detection of rare and divergent NRPS and PKS clones in European, Antarctic and Cuban soils. PLoS ONE 10:e0138327
Ayuso A, Clark D, González I, Salazar O, Anderson A, Genilloud O (2005) A novel actinomycete strain de-replication approach based on the diversity of polyketide synthase and nonribosomal peptide synthetase biosynthetic pathways. Appl Microbiol Biotechnol 67:795–806
Blosser SJ et al (2016) Multi-center MALDI-TOF MS study for the identification of clinically-relevant Nocardia spp. J Clin Microbiol. https://doi.org/10.1128/JCM.02942-15
Boottanun P, Potisap C, Hurdle JG, Sermswan RW (2017) Secondary metabolites from Bacillus amyloliquefaciens isolated from soil can kill Burkholderia pseudomallei. AMB Express 7:16
Chen Y-H et al (2012) Isolation of marine bacteria with antimicrobial activities from cultured and field-collected soft corals. World J Microbiol Biotechnol 28:3269–3279
Chen JN et al (2016) Extracts containing CLPs of Bacillus amyloliquefaciens JN68 isolated from chicken intestines exert antimicrobial effects, particularly on methicillin-resistant Staphylococcus aureus and Listeria monocytogenes. Mol Med Rep 14:5155–5163
Jiang S et al (2007) Diversity of culturable actinobacteria isolated from marine sponge Haliclona sp. Antonie Van Leeuwenhoek 92:405–416
Kara M, Saitoh Y, Nakano H (1990) DNA strand scission by the novel antitumor antibiotic leinamycin. Biochemistry 29:5676–5681
Kennedy J et al (2009) Isolation and analysis of bacteria with antimicrobial activities from the marine sponge Haliclona simulans collected from Irish waters. Mar Biotechnol 11:384–396
Khameneh B, Diab R, Ghazvini K, Bazzaz BSF (2016) Breakthroughs in bacterial resistance mechanisms and the potential ways to combat them. Microb Pathog 95:32–42
Kleigrewe K, Gerwick L, Sherman DH, Gerwick WH (2016) Unique marine derived cyanobacterial biosynthetic genes for chemical diversity. Nat Prod Rep 33:348–364
Machado H, Sonnenschein EC, Melchiorsen J, Gram L (2015) Genome mining reveals unlocked bioactive potential of marine Gram-negative bacteria. BMC Genomics 16:158
Mani P, Dineshkumar G, Jayaseelan T, Deepalakshmi K, Kumar CG, Balan SS (2016) Antimicrobial activities of a promising glycolipid biosurfactant from a novel marine Staphylococcus saprophyticus SBPS 15. 3 Biotech 6:163
McFadden CS, Sánchez JA, France SC (2010) Molecular phylogenetic insights into the evolution of Octocorallia: a review. Integr Comp Biol. https://doi.org/10.1093/icb/icq056
Mincer TJ, Jensen PR, Kauffman CA, Fenical W (2002) Widespread and persistent populations of a major new marine actinomycete taxon in ocean sediments. Appl Environ Microbiol 68:5005–5011
Mukherjee A, Das R, Pal A, Paul A (2017) Antibacterial activity of endophytic Bacillus safensis isolated from Ophioglossum reticulatum L. Microbiol Res J Int 18:1–12
Ndlovu T, Rautenbach M, Vosloo JA, Khan S, Khan W (2017) Characterisation and antimicrobial activity of biosurfactant extracts produced by Bacillus amyloliquefaciens and Pseudomonas aeruginosa isolated from a wastewater treatment plant. AMB Express 7:108
Ngugi DK, Antunes A, Brune A, Stingl U (2012) Biogeography of pelagic bacterioplankton across an antagonistic temperature–salinity gradient in the Red Sea. Mol Ecol 21:388–405
Ponnappan N, Budagavi DP, Yadav BK, Chugh A (2015) Membrane-active peptides from marine organisms—antimicrobials, cell-penetrating peptides and peptide toxins: applications and prospects. Probiotics Antimicrob Proteins 7:75–89
Reysenbach A-L, Giver LJ, Wickham GS, Pace NR (1992) Differential amplification of rRNA genes by polymerase chain reaction. Appl Environ Microbiol 58:3417–3418
Temraz TA, Houssen WE, Jaspars M, Woolley DR, Wease KN, Davies SN, Scott RH (2006) A pyridinium derivative from Red Sea soft corals inhibited voltage-activated potassium conductances and increased excitability of rat cultured sensory neurones. BMC Pharmacol 6:10
Weber T et al (2008) Molecular analysis of the kirromycin biosynthetic gene cluster revealed beta-alanine as precursor of the pyridone moiety. Chem Biol 15:175–188. https://doi.org/10.1016/j.chembiol.2007.12.009
Webster NS, Wilson KJ, Blackall LL, Hill RT (2001) Phylogenetic diversity of bacteria associated with the marine sponge Rhopaloeides odorabile. Appl Environ Microbiol 67:434–444
World Health Organization (2014) WHO’s first global report on antibiotic resistance reveals serious, worldwide threat to public health. In: Antimicrobial resistance—global surveillance report. Virtual press conference
Yan Y et al (2016) Complete genome sequence of Bacillus amyloliquefaciens B15 isolated from grape skin, a strain of strong inhibitory activity against fungi. J Biotechnol 228:28–29
Acknowledgements
We would like to thank Dr. Hossamuldin Elfeqy for his help in collecting marine invertebrates samples from Eltor- Sharm El sheikh.
Supporting information
Supplementary Table 1—Identification score criteria by Bruker Daltonics.
Supplementary Table 2—The primers used in screening or biosynthetic genes PKS I, II and NRPS I, II genes.
Supplementary Table 3—The amplification protocol of the primers used in screening for biosynthetic PKS I, II and NRPS I, II genes.
Supplementary Table 4—Classification of the isolates according to the type of the invertebrate and the type of the medium.
Supplementary Table 5—Identification of characteristic isolates using MALDI-TOF MS.
Supplementary Table 6—Screening of the isolates for biosynthetic gene fragments PKS I&II and NRPS I&II.
Supplementary Figure 1—Screening of the isolates for biosynthetic gene fragments NRPS I (primer 1) and NRPS II (primer 2).
Supplementary Figure 2—Screening of the isolates for biosynthetic gene fragments PKS I (primer 3) and PKS I (primer 4).
Supplementary Figure 3—Screening of the isolates for biosynthetic gene fragments PKS II (primer 5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Accession numbers: for 16S sequences: MG757672.1–MG757678.1; for PKS and NRPS sequences: MG975081–MG975083.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Helal, H.S., Hanora, A., Khattab, R.A. et al. Mining of Egypt’s Red Sea invertebrates for potential bioactive producers. Biotechnol Lett 40, 1519–1530 (2018). https://doi.org/10.1007/s10529-018-2600-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-018-2600-x