Abstract
Objective
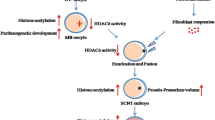
To examine the effect of PCI-24781 (abexinostat) on the blastocyst formation rate in pig somatic cell nuclear transferred (SCNT) embryos and acetylation levels of the histone H3 lysine 9 and histone H4 lysine 12.
Results
Treatment with 0.5 nM PCI-24781 for 6 h significantly improved the development of cloned embryos, in comparison to the control group (25.3 vs. 10.5 %, P < 0.05). Furthermore, PCI-24781 treatment led to elevated acetylation of H3K9 and H4K12. TUNEL assay and Hoechst 33342 staining revealed that the percentage of apoptotic cells in blastocysts was significantly lower in PCI-24781-treated SCNT embryos than in untreated embryos. Also, PCI-24781-treated embryos were transferred into three surrogate sows, one of whom became pregnant and two fetuses developed.
Conclusion
PCI-24781 improves nuclear reprogramming and the developmental potential of pig SCNT embryos.




Similar content being viewed by others
References
Adimoolam S, Sirisawad M, Chen J et al (2007) HDAC inhibitor PCI-24781 decreases RAD51 expression and inhibits homologous recombination. Proc Natl Acad Sci USA 104:19482–19487
Benedetti R, Conte M, Altucci L (2015) Targeting histone deacetylases in diseases: where are we? Antioxid Redox Signal 23:99–126
Buggy JJ, Cao ZA, Bass KE et al (2006) CRA-024781: a novel synthetic inhibitor of histone deacetylase enzymes with antitumor activity in vitro and in vivo. Mol Cancer Ther 5:1309–1317
Bui HT, Wakayama S, Kishigami S et al (2010) Effect of trichostatin A on chromatin remodeling, histone modifications, DNA replication, and transcriptional activity in cloned mouse embryos. Biol Reprod 83:454–463
Campbell KH, Fisher P, Chen WC et al (2007) Somatic cell nuclear transfer: past, present and future perspectives. Theriogenology 68(Suppl 1):S214–S231
Cibelli J (2007) Developmental biology. A decade of cloning mystique. Science 316:990–992
Glass CK, Rosenfeld MG (2000) The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev 14:121–141
Hardy K (1997) Cell death in the mammalian blastocyst. Mol Hum Reprod 3:919–925
Hochedlinger K, Jaenisch R (2006) Nuclear reprogramming and pluripotency. Nature 441:1061–1067
Jin JX, Li S, Gao QS et al (2013) Significant improvement of pig cloning efficiency by treatment with LBH589 after somatic cell nuclear transfer. Theriogenology 80:630–635
Jin JX, Li S, Hong Y et al (2014) CUDC-101, a histone deacetylase inhibitor, improves the in vitro and in vivo developmental competence of somatic cell nuclear transfer pig embryos. Theriogenology 81:572–578
Jin JX, Kang JD, Li S et al (2015) PXD101 significantly improves nuclear reprogramming and the in vitro developmental competence of porcine SCNT embryos. Biochem Biophys Res Commun 456:156–161
Kang JD, Li S, Lu Y et al (2013) Valproic acid improved in vitro development of pig cloning embryos but did not improve survival of cloned pigs to adulthood. Theriogenology 79(306–311):e301
Kishigami S, Mizutani E, Ohta H et al (2006) Significant improvement of mouse cloning technique by treatment with trichostatin A after somatic nuclear transfer. Biochem Biophys Res Commun 340:183–189
Lai L, Kang JX, Li R et al (2006) Generation of cloned transgenic pigs rich in omega-3 fatty acids. Nat Biotechnol 24:435–436
Lee DY, Hayes JJ, Pruss D et al (1993) A positive role for histone acetylation in transcription factor access to nucleosomal DNA. Cell 72:73–84
Liang S, Zhao MH, Choi JW et al (2015) Scriptaid treatment decreases dna methyltransferase 1 expression by induction of MicroRNA-152 expression in porcine somatic cell nuclear transfer embryos. PLoS One 10:e0134567
Liu L, Liu Y, Gao F et al (2012) Embryonic development and gene expression of porcine SCNT embryos treated with sodium butyrate. J Exp Zool B 318:224–234
Luo B, Ju S, Muneri CW et al (2015) Effects of histone acetylation status on the early development of in vitro porcine transgenic cloned embryos. Cell Reprogr 17:41–48
Mao J, Zhao MT, Whitworth KM et al (2015) Oxamflatin treatment enhances cloned porcine embryo development and nuclear reprogramming. Cell Reprogr 17:28–40
Polejaeva IA, Chen SH, Vaught TD et al (2000) Cloned pigs produced by nuclear transfer from adult somatic cells. Nature 407(2000):86–90
Pratt SL, Sherrer ES, Reeves DE et al (2006) Factors influencing the commercialization of cloning in the pork industry. Soc Reprod Fertil Suppl 62:303–315
Qian L, Tang M, Yang J et al (2015) Targeted mutations in myostatin by zinc-finger nucleases result in double-muscled phenotype in Meishan pigs. Sci Rep 5:14435
Rideout WM 3rd, Eggan K, Jaenisch R (2001) Nuclear cloning and epigenetic reprogramming of the genome. Science 293:1093–1098
Rybouchkin A, Kato Y, Tsunoda Y (2006) Role of histone acetylation in reprogramming of somatic nuclei following nuclear transfer. Biol Reprod 74:1083–1089
Salvador MA, Wicinski J, Cabaud O et al (2013) The histone deacetylase inhibitor abexinostat induces cancer stem cells differentiation in breast cancer with low Xist expression. Clin Cancer Res 19:6520–6531
Song Y, Hai T, Wang Y et al (2014) Epigenetic reprogramming, gene expression and in vitro development of porcine SCNT embryos are significantly improved by a histone deacetylase inhibitor-m-carboxycinnamic acid bishydroxamide (CBHA). Protein Cell 5:382–393
Su J, Wang Y, Li Y et al (2011) Oxamflatin significantly improves nuclear reprogramming, blastocyst quality, and in vitro development of bovine SCNT embryos. PLoS One 6:e23805
Turner BM (1998) Histone acetylation as an epigenetic determinant of long-term transcriptional competence. Cell Mol Life Sci 54:21–31
Turner BM (2002) Cellular memory and the histone code. Cell 111:285–291
Wang F, Kou Z, Zhang Y et al (2007) Dynamic reprogramming of histone acetylation and methylation in the first cell cycle of cloned mouse embryos. Biol Reprod 77:1007–1016
Wang Y, Su J, Wang L et al (2011) The effects of 5-aza-2′-deoxycytidine and trichostatin A on gene expression and DNA methylation status in cloned bovine blastocysts. Cell Reprogr 13:297–306
Whitworth KM, Zhao J, Spate LD et al (2011) Scriptaid corrects gene expression of a few aberrantly reprogrammed transcripts in nuclear transfer pig blastocyst stage embryos. Cell Reprogr 13:191–204
Whitworth KM, Zhao J, Lee K et al (2015) Transcriptome analysis of pig in vivo, in vitro-fertilized, and nuclear transfer blastocyst-stage embryos treated with histone deacetylase inhibitors postfusion and activation reveals changes in the lysosomal pathway. Cell Reprogr 17:243–258
Yang X, Smith SL, Tian XC et al (2007) Nuclear reprogramming of cloned embryos and its implications for therapeutic cloning. Nat Genet 39:295–302
Yang C, Choy E, Hornicek FJ et al (2011) Histone deacetylase inhibitor PCI-24781 enhances chemotherapy-induced apoptosis in multidrug-resistant sarcoma cell lines. Anticancer Res 31:1115–1123
Yin XJ, Tani T, Yonemura I et al (2002) Production of cloned pigs from adult somatic cells by chemically assisted removal of maternal chromosomes. Biol Reprod 67:442–446
Yoshioka K, Suzuki C, Tanaka A et al (2002) Birth of piglets derived from porcine zygotes cultured in a chemically defined medium. Biol Reprod 66:112–119
Yu Y, Ding C, Wang E et al (2007) Piezo-assisted nuclear transfer affects cloning efficiency and may cause apoptosis. Reproduction 133:947–954
Zhan Q, Tsai S, Lu Y et al (2013) RuvBL2 is involved in histone deacetylase inhibitor PCI-24781-induced cell death in SK-N-DZ neuroblastoma cells. PLoS One 8:e71663
Acknowledgments
This work was supported by the State Key Development Program for Basic Research of China (Grant No. 20150622005JC) and the institute for Basic Science (Grant No. IBS-R021-D1-2015-a02).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Long Jin and Hai-Ying Zhu have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Jin, L., Zhu, HY., Guo, Q. et al. PCI-24781 can improve in vitro and in vivo developmental capacity of pig somatic cell nuclear transfer embryos. Biotechnol Lett 38, 1433–1441 (2016). https://doi.org/10.1007/s10529-016-2141-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2141-0