Abstract
Objectives
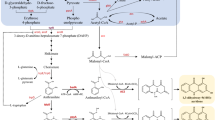
To produce rosmarinic acid analogues in the recombinant Escherichia coli BLRA1, harboring a 4-coumarate: CoA ligase from Arabidopsis thaliana (At4CL) and a rosmarinic acid synthase from Coleus blumei (CbRAS).
Results
Incubation of the recombinant E. coli strain BLRA1 with exogenously supplied phenyllactic acid (PL) and analogues as acceptor substrates, and coumaric acid and analogues as donor substrates led to production of 18 compounds, including 13 unnatural RA analogues.
Conclusion
This work demonstrates the viability of synthesizing a broad range of rosmarinic acid analogues in E. coli, and sheds new light on the substrate specificity of CbRAS.




Similar content being viewed by others
References
Ajikumar PK, Xiao WH, Tyo KEJ, Wang Y, Simeon F, Leonard E, Mucha O, Phon TH, Pfeifer B, Stephanopoulos G (2010) Isoprenoid pathway optimization for taxol precursor overproduction in Escherichia coli. Science 330:70–74
Bai YF, Bi HP, Zhuang YB, Liu C, Cai T, Liu XN, Zhang XL, Liu T, Ma YH (2014) Production of salidroside in metabolically engineered Escherichia coli. Sci Rep 4:6640
Berger A, Meinhard J, Petersen M (2006) Rosmarinic acid synthase is a new member of the superfamily of BAHD acyltransferases. Planta 224:1503–1510
Bloch SE, Schmidt-Dannert C (2014) Construction of a chimeric biosynthetic pathway for the de novo biosynthesis of rosmarinic acid in Escherichia coli. ChemBioChem 15:2393–2401
Bulgakov VP, Inyushkina YV, Fedoreyev SA (2012) Rosmarinic acid and its derivatives: biotechnology and applications. Crit Rev Biotechnol 32:203–217
Cane DE, Kudo F, Kinoshita K, Khosla C (2002) Precursor-directed biosynthesis: biochemical basis of the remarkable selectivity of the erythromycin polyketide synthase toward unsaturated triketides. Chem Biol 9:131–142
Di P, Zhang L, Chen J, Tan H, Xiao Y, Dong X, Zhou X, Chen W (2013) C-13 Tracer reveals phenolic acids biosynthesis in hairy root cultures of Salvia miltiorrhiza. ACS Chem Biol 8:1537–1548
Jiang JJ, Bi HP, Zhuang YB, Liu SW, Liu T, Ma YH (2015) Engineered synthesis of rosmarinic acid in Escherichia coli resulting production of a new analogue caffeoyl-phenyllactate. Biotechnol Lett. doi:10.1007/s10529-015-1945-7
Katsuyama Y, Funa N, Miyahisa I, Horinouchi S (2007) Synthesis of unnatural flavonoids and stilbenes by exploiting the plant biosynthetic pathway in Escherichia coli. Chem Biol 14:613–621
Khojasteh A, Mirjalili MH, Hidalgo D, Corchete P, Palazon J (2014) New trends in biotechnological production of rosmarinic acid. Biotechnol Lett 36:2393–2406
Kim GD, Park YS, Jin YH, Park CS (2015) Production and applications of rosmarinic acid and structurally related compounds. Appl Microbiol Biot 99:2083–2092
Landmann C, Hucherig S, Fink B, Hoffmann T, Dittlein D, Coiner HA, Schwab W (2011) Substrate promiscuity of a rosmarinic acid synthase from lavender (Lavandula angustifolia L.). Planta 234:305–320
Mora-Pale M, Sanchez-Rodriguez SP, Linhardt RJ, Dordick JS, Koffas MA (2013) Metabolic engineering and in vitro biosynthesis of phytochemicals and non-natural analogues. Plant Sci 210:10–24
Petersen M (2013) Rosmarinic acid: new aspects. Phytochem Rev 12:207–227
Sander M, Petersen M (2011) Distinct substrate specificities and unusual substrate flexibilities of two hydroxycinnamoyltransferases, rosmarinic acid synthase and hydroxycinnamoyl-CoA:shikimate hydroxycinnamoyl-transferase, from Coleus blumei Benth. Planta 233:1157–1171
Zhang PX, Tang YP, Li NG, Zhu Y, Duan JA (2014) Bioactivity and chemical synthesis of caffeic acid phenethyl ester and its derivatives. Molecules 19:16458–16476
Zhao SJ, Jones JA, Lachance DM, Bhan N, Khalidi O, Venkataraman S, Wang ZT, Koffas MAG (2015) Improvement of catechin production in Escherichia coli through combinatorial metabolic engineering. Metab Eng 28:43–53
Acknowledgments
This work was supported by grants from the 973 Program of China (no. 2012CB721100), the 863 Program of China (no. 2012AA02A704), the 100 Talents Program of the Chinese Academy of Sciences and the National Natural Science Foundation of China (nos. 21302214, 31300040, and 31400026).
Supporting information
Supplementary NMR data for compounds 3–15.
Supplementary Fig. 1—HPLC–MS analysis of metabolite profile of the recombinant E. coli strain BLRA1 supplied with ferulic acid and other substrates.
Supplementary Fig. 2—HPLC–MS analysis of metabolite profile of the recombinant E. coli strain BLRA1 supplied with p-hydroxyphenyl propionic acid (HPPA) and other substrates.
Supplementary Fig. 3—HPLC–MS analysis of metabolites profile of recombinant E. coli strain BLRA1 supplied with mandelic acid and other substrates.
Supplementary Fig. 4—HPLC–MS analysis of fermentation broth of recombinant E. coli strain BLRA1 fed with tyrosol and other substrates.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yibin Zhuang, Jingjie Jiang, and Huiping Bi have contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhuang, Y., Jiang, J., Bi, H. et al. Synthesis of rosmarinic acid analogues in Escherichia coli . Biotechnol Lett 38, 619–627 (2016). https://doi.org/10.1007/s10529-015-2011-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-015-2011-1