Abstract
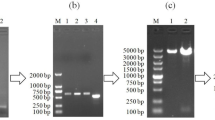
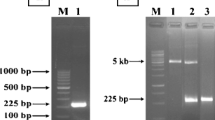
A novel production method in Escherichia coli for an antimicrobial peptide of 21 amino acids, buforin IIb, which is a synthetic analog of buforin II, has been developed. The buforin IIb gene was cloned into the vector pET32a to construct an expression vector pET32a–buforin IIb. The fusion protein Trx-buforin IIb, purified by nickel nitrilo-triacetic acid (Ni-NTA) resin chromatography, was cleaved by hydroxylamine hydrochloride to release recombinant buforin IIb. Purification of recombinant buforin IIb was achieved by HPLC: about 3.1 mg/l active recombinant buforin IIb with purity >99% was obtained. The recombinant buforin IIb showed antimicrobial activities that were similar to the synthetic one.




Similar content being viewed by others
References
Cao W, Zhou Y, Ma Y, Luo Q, Wei D (2005) Expression and purification of antimicrobial peptide adenoregulin with C-amidated terminus in Escherichia coli. Protein Expr Purif 40:404–410
Hara S, Yamakawa M (1995) Moricin, a novel type of antibacterial peptide isolated from the silkworm, Bombyx mori. J Biol Chem 70:29923–29927
Iijima R, Kisugi J, Yamazaki Y (2003) A novel antimicrobial peptide from the sea hare Dolabella auricularia. Dev Comp Immunol 27:305–311
Kamysz W, Okroj M, Lukasiak J (2003) Novel properties of antimicrobial peptides. Acta Biochim Pol 50:461–469
Lee DG, Park Y, Kim HN, Kim HK, Kim PI, Choi BH, Hahm KS (2002) Antifungal mechanism of an antimicrobial peptide, HP(2–20), derived from N-terminus of Helicobacter pylori ribosomal protein L1 against Candida albicans. Biochem Biophys Res Commun 291:1006–1013
Lee HS, Park CB, Kim JM, Jang SA, Park IY, Kim MS, Cho JH, Kim SC (2008) Mechanism of anticancer activity of buforin IIb, a histone H2A-derived peptide. Cancer Lett 271:47–55
Loll PJ (2003) Membrane protein structural biology: the high throughput challenge. J Struct Biol 142:144–153
Merrifield RB (1963) Solid phase peptide synthesis. I. the synthesis of a tetrapeptide. J Am Chem Soc 85:2149–2154
Moks T, Abrahmsen L, Holmgren E, Bilich M, Olsson A, Uhlen M, Pohl G, Sterky C, Hultberg H, Josephson S (1987) Expression of human insulin-like growth factor I in bacteria: use of optimized gene fusion vectors to facilitate protein purification. Biochemistry 26:5239–5244
Park CB, Kim MS, Kim SC (1996) A novel antimicrobial peptide from Bufo bufo gargarizans. Biochem Biophys Res Commun 218:408–413
Park CB, Kim HS, Kim SC (1998) Mechanism of action of the antimicrobial peptide buforin II: buforin II kills microorganisms by penetrating the cell membrane and inhibiting cellular functions. Biochem Biophys Res Commun 244:253–257
Park CB, Yi KS, Matsuzaki K, Kim MS, Kim SC (2000) Structure-activity analysis of buforin II, a histone H2A-derived antimicrobial peptide: the proline hinge is responsible for the cell-penetrating ability of buforin II. Proc Natl Acad Sci USA 97:8245–8250
Pyo SH, Lee JH, Park HB, Cho JS, Kim HR, Han BH, Park YS (2004) Expression and purification of a recombinant buforin derivative from Escherichia coli. Proc Biochem 39:1731–1736
Schägger H (2006) Tricine–SDS-PAGE. Nat Protoc 1:16–22
Wu YF, Fan YM, Xue B, Luo L, Shen JY, Zhang SQ, Jiang Y, Yin ZM (2006) Human glutathione S-transferase P1-1 interacts with TRAF2 and regulates TRAF2-ASK1 signals. Oncogene 25:5787–5800
Zasloff M (1987) Magainins, a class of antimicrobial peptides from Xenopus skin: isolation, characterization of two active forms, and partial cDNA sequence of a precursor. Proc Natl Acad Sci USA 84:5449–5453
Zuo X, Li S, Hall J, Mattern MR, Tran H, Shoo J, Tan R, Weiss SR, Butt TR (2005) Enhanced expression and purification of membrane proteins by SUMO fusion in Escherichia coli. J Struct Funct Genomics 6:103–111
Acknowledgments
This work was supported by the special funding from NJNU for talent faculty and Jiangsu superior discipline.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, Q., Zhu, F., Xin, Y. et al. Expression and purification of antimicrobial peptide buforin IIb in Escherichia coli . Biotechnol Lett 33, 2121–2126 (2011). https://doi.org/10.1007/s10529-011-0687-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-011-0687-4