Abstract
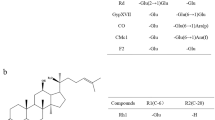
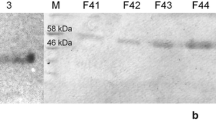
Thiohydroximates comprise a diverse class of compounds important in both biological and industrial chemistry. Their syntheses are generally limited to simple alkyl and aryl compounds with few stereocenters and a narrow range of functional groups. We hypothesized that sequential action of two recombinant enzymes, a sulfatase from Helix pomatia and a β-O-glucosidase from Caldicellulosiruptor saccharolyticus, on glucosinolates would allow synthesis of thiohydroximates from a structurally broad array of abundant precursors. We report successful synthesis of thiohydroximates of varied chemical classes, including from homochiral compounds of demonstrated biological activity. The chemoenzymatic synthetic route reported here should allow access to many, if not all, of the thiohydroximate core structures of the ~200 known naturally occurring glucosinolates. The enrichment of this group for compounds with possible pharmacological potential is discussed.




Similar content being viewed by others
References
Bauer W, Kuhlein K (1985) Methoden der organischen chemie. Georg Thieme Verlag, Stuttgart
Chimiak A, Przychodzen W, Rachon J (2002) The thiohydroxamate system. Heteroatom Chem 13:169–194
Clarke DB (2010) Glucosinolates, structures and analysis in food. Anal Meth 2:310–325
Gross HB, Dalebout T, Grubb CD, Abel S (2000) Functional detection of chemopreventive glucosinolates in Arabidopsis thaliana. Plant Sci 159:265–272
Grubb CD, Abel S (2006) Glucosinolate metabolism and its control. Trends Plant Sci 11:89–100
Grubb CD, Zipp BJ, Ludwig-Müller J, Masuno MN, Molinski TF, Abel S (2004) Arabidopsis glucosyltransferase UGT74B1 functions in glucosinolate biosynthesis and auxin homeostasis. Plant J 40:893–908
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333
Love DR, Fisher R, Bergquist PL (1988) Sequence structure and expression of a cloned beta-glucosidase gene from an extreme thermophile. Mol Gen Genet 213:84–92
Nagata K, Mizukami S (1967) Studies on thiohydroxamic acids and their metal chelates. 4. Reaction of thiohydroxamic acids with metal ions. Chem Pharma Bull 15:61–69
Olbe L, Carlsson E, Lindberg P (2003) A proton-pump inhibitor expedition: the case histories of omeprazole and esomeprazole. Nat Rev Drug Disc 2:132–139
Pedras MS, Okinyo DP (2008) Remarkable incorporation of the first sulfur containing indole derivative: another piece in the biosynthetic puzzle of crucifer phytoalexins. Org Biomol Chem 6:51–54
Walter W, Schauman E (1971) Chemistry of thiohydroxamic acids. Synthesis 111–130
Wang Q, Grubb CD, Abel S (2002) Direct analysis of single leaf disks for chemopreventive glucosinolates. Phytochem Anal 13:152–157
Wathelet JP, Iori R, Leoni O, Rollin P, Mabon N, Marlier M, Palmieri S (2001) A recombinant beta-O-glucosidase from Caldocellum saccharolyticum to hydrolyse desulfo-glucosinolates. Biotechnol Lett 23:443–446
Winkelmann G, van der Helm D, Neilands JB (1987) Iron transport in microbes plants and animals. VCH, Weinheim, New York
Acknowledgments
The authors wish to thank Drs. Soledade Pedras and Bernhard Westermann for helpful criticism of the manuscript, and Drs. Peter Bergquist and Moreland Gibbs for providing CSG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kopycki, J., Schmidt, J., Abel, S. et al. Chemoenzymatic synthesis of diverse thiohydroximates from glucosinolate-utilizing enzymes from Helix pomatia and Caldicellulosiruptor saccharolyticus . Biotechnol Lett 33, 1039–1046 (2011). https://doi.org/10.1007/s10529-011-0530-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-011-0530-y