Abstract
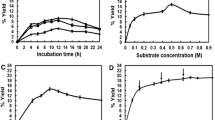
Acylation of 0.5 g sucrose with 1.2 ml acetic anhydride was carried out in 2 ml two-solvent medium of anhydrous pyridine/n-hexane (1:1, v/v) using 0.2 g crude protease from Serratia sp. Sucrose octaacetate was the sole product and more than 90% sucrose was converted in 24 h at 30°C. The purity of sucrose octaacetate reached 100%, via a simple purification method of alcohol/water washing and centrifugation.



Similar content being viewed by others
References
Harder DB, Capeless CG, Maggio JC et al (1992) Intermediate sucrose octa-acetate sensitivity suggests a third allele at mouse bitter taste locus Soa and Soa-Rua identity. Chem Senses 17:391–401
Kumar CG, Takagi H (1999) Microbial alkaline proteases: from a bioindustrial viewpoint. Biotechnol Adv 17:561–594
Mao D, Wang Y, Hu L et al (2006) Progress in application of enzyme-catalyzed regioselective acylation in synthesis of sucrose esters. China Surfact Deterg Cosmet 36:369–373
Pedersen NR, Halling PJ, Pedersen LH et al (2002) Efficient transesterification of sucrose catalysed by the metalloprotease thermolysin in dimethylsulfoxide. FEBS Lett 519:181–184
Plou FJ, Cruces MA, Ferrer M et al (2002) Enzymatic acylation of di- and trisaccharides with fatty acids: choosing the appropriate enzyme, support and solvent. J Biotechnol 96:55–66
Polat T, Linhardt RJ (2001) Syntheses and applications of sucrose-based esters. J Surfactants Deterg 4:415–421
Yoo IS, Park SJ, Yoon HH (2007) Enzymatic synthesis of sugar fatty acid esters. J Ind Eng Chem 13:1–6
Acknowledgments
This work was financially supported by the National High Technology and Development Program of China (863 Program, Grant No. 2010AA101501).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, G., Cai, Y., Liao, X. et al. Enzymatic synthesis of sucrose octaacetate using a novel alkaline protease. Biotechnol Lett 33, 607–610 (2011). https://doi.org/10.1007/s10529-010-0468-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-010-0468-5