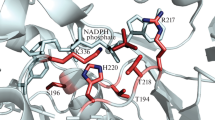
Abstract
A major problem when xylose is used for ethanol production is the intercellular redox imbalance arising from different coenzyme specificities of xylose reductase (XR) and xylitol dehydrogenase. The residue Lys21 in XR from Pichia stipitis was subjected to site-directed mutagenesis to alter its coenzyme specificity. The N272D mutant exhibited improved catalytic efficiency when NADH was the coenzyme. Both K21A and K21A/N272D preferred NADH to NADPH, their catalytic efficiencies for NADPH were almost zero. The catalytic efficiency of K21A/N272D for NADH was almost 9-fold and 2-fold that of K21A and the wild-type enzyme, respectively. Complete reversal of coenzyme specificity toward NADH and improved catalytic efficiency were achieved.

Similar content being viewed by others
References
Chu BCH, Lee H (2006) Investigation of the role of a conserved glycine motif in the Saccharomyces cerevisiae xylose reductase. Curr Microbiol 53:118–123
Hahn-Hagerdal B, Karhumaa K, Fonseca C et al (2007) Towards industrial pentose-fermenting yeast strains. Appl Microbiol Biotechnol 74:937–953
Jeong EY, Sopher C, Kim IS et al (2001) Mutational study of the role of tyrosine-49 in the Saccharomyces cerevisiae xylose reductase. Yeast 18:1081–1089
Jeppsson M, Traff K, Johansson B et al (2003) Effect of enhanced xylose reductase activity on xylose consumption and product distribution in xylose-fermenting recombinant Saccharomyces cerevisiae. FEMS Yeast Res 3:167–175
Jeppsson M, Bengtsson O, Franke K et al (2006) The expression of a Pichia stipitis xylose reductase mutant with higher Km for NADPH increases ethanol production from xylose in recombinant Saccharomyces cerevisiae. Biotechnol Bioeng 93:665–673
Katahira S, Mizuike A, Fukuda H et al (2006) Ethanol fermentation from lignocellulosic hydrolysate by a recombinant xylose- and cellooligosaccharide-assimilating yeast strain. Appl Microbiol Biotechnol 72:1136–1143
Klimacek M, Szekely M, Griessler R et al (2001) Exploring the active site of yeast xylose reductase by site-directed mutagenesis of sequence motifs characteristic of two dehydrogenase/reductase family types. FEBS Lett 500:149–152
Lin Y, Tanaka S (2006) Ethanol fermentation from biomass resources: current state and prospects. Appl Microbiol Biotechnol 69:627–642
Nigam JN (2001) Ethanol production from wheat straw hemicellulose hydrolysate by Pichia stipitis. J Biotech 87:17–27
Petschacher B, Leitgeb S, Kavanagh KL et al (2005) The coenzyme specificity of Candida tenuis xylose reductase (AKR2B5) explored by site-directed mutagenesis and X-ray crystallography. Biochem J 385:75–83
Wang JF, Wei DQ, Lin Y et al (2007) Insights from modeling the 3D structure of NAD(P)H-dependent D-xylose reductase of Pichia stipitis and its binding interactions with NAD and NADP. Biochem Biophys Res Commun 359:323–329
Watanabe S, Kodaki T, Makino K (2005) Complete reversal of coenzyme specificity of xylitol dehydrogenase and increase of thermostability by the introduction of structural zinc. J Biol Chem 280:10340–10349
Watanabe S, Abu Saleh A, Pack SP et al (2007a) Ethanol production from xylose by recombinant Saccharomyces cerevisiae expressing protein engineered NADP(+)-dependent xylitol dehydrogenase. J Biotech 130:316–319
Watanabe S, Pack SP, Abu Saleh A et al (2007b) The positive effect of the decreased NADPH-preferring activity of xylose reductase from Pichia stipitis on ethanol production using xylose-fermenting recombinant Saccharomyces cerevisiae. Biosci Biotechnol Biochem 71:1365–1369
Zeng QK, Du HL, Zhai ZC et al (2008) Mutational research on the role of lysine 21 in the Pichia stipitis xylose reductase. Chin J Biotech 24:1108–1111
Acknowledgements
This study was supported by grants from the National 863 Bioinformatics projects under contract no. 2007AA02Z333, the Chinese National Science Foundation under contract nos. 20773085 and 30870476, and the Guangdong Natural Science Foundation under contract no. 06300199. It was also supported by the Virtual Laboratory for Computational Chemistry of CNIC and the Supercomputing Center of CNIC, Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Qi-Kai Zeng, Hong-Li Du, Jing-Fang Wang have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zeng, QK., Du, HL., Wang, JF. et al. Reversal of coenzyme specificity and improvement of catalytic efficiency of Pichia stipitis xylose reductase by rational site-directed mutagenesis. Biotechnol Lett 31, 1025–1029 (2009). https://doi.org/10.1007/s10529-009-9980-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-009-9980-x