Abstract
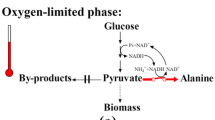
dl-Alanine was produced from glucose in an Escherichia coli pfl pps poxB ldhA aceEF pTrc99A-alaD strain which lacked pyruvate-formate lyase, phosphoenolpyruvate (PEP) synthase, pyruvate oxidase, lactate dehydogenase, components of the pyruvate dehydogenase complex and over-produced alanine dehydrogenase (ALD). A two-phase process was developed with cell growth under aerobic conditions followed by alanine production under anaerobic conditions. Using the batch mode, cells grew to 5.3 g/l in 9 h with the accumulation of 6–10 g acetate/l, and under subsequent anaerobic conditions achieved 34 g alanine/l in 13 h with a yield of 0.86 g/g glucose. Using the fed-batch mode at μ = 0.15 h−1, only about 1 g acetate/l formed in the 25 h required for the cells to reach 5.6 g/l, and 88 g alanine/l accumulated during the subsequent 23 h. This fed-batch process attained an alanine volumetric productivity of 4 g/lh during the production phase, and a yield that was essentially 1 g/g.
Similar content being viewed by others
References
Åkesson M, Hagander P, Axelsson JP (2001) Avoiding acetate accumulation in Escherichia coli cultures using feedback control of glucose feeding. Biotechnol Bioeng 73(3):223–230
Bunch PK, Mat-Jan F, Lee N, Clark DP (1997) The ldhA gene encoding the fermentative lactate dehydrogenase of Escherichia coli. Microbiol 142:187–195
Chang YY, Cronan Jr JE (1983) Genetic and biochemical analyses of Escherichia coli strains having a mutation in the structural gene (poxB) for pyruvate oxidase. J Bacteriol 154(2):756–762
Chibata TI, Kakimoto T, Kato J (1969) Process for producing l-alanine. US Patent 3,458,400
Eiteman MA, Chastain MJ (1997) Optimization of the ion-exchange analysis of organic acids from fermentation. Anal Chim Acta 338:69–75
Hashimoto S, Katsumata R (1998) l-alanine fermentation by an alanine racemase-deficient mutant of the dl-alanine hyperproducing bacterium Arthrobacter oxydans HAP-1. J Ferment Bioeng 86:385–390
Katsumata R, Hashimoto S (1996) Process for producing alanine. US Patent 5,559,016
Kleman GL, Strohl WR (1994) Acetate metabolism in Escherichia coli in high-cell-density fermentation. Appl Environ Microbiol 60(11):3952–3958
Kleman GL, Chalmers JJ, Luli GW, Strohl WR (1991) A predictive and feedback control algorithm maintains a constant glucose concentration in fed-batch fermentations. Appl Environ Microbiol 57(4):910–917
Lee M, Smith GM, Eiteman MA, Altman E (2004) Aerobic production of alanine by Escherichia coli aceF ldhA mutants expressing the Bacillus sphaericus alaD gene. Appl Microbiol Biotechnol 65:56–60
Luli GW, Strohl WR (1990) Comparison of growth, acetate production, and acetate inhibition of Escherichia coli strains in batch and fed-batch fermentations. Appl Environ Microbiol 56(4):1004–1011
Mat-Jan F, Alam KY, Clark DP (1989) Mutants of Escherichia coli deficient in the fermentative lactate deydrogenase. J Bacteriol 171(1):342–348
Ohashima T, Soda K (1979) Purification and properties of alanine dehydrogenase from Bacillus sphaericus. Eur J Biochem 100:29–39
Uhlenbusch I, Sahm H, Sprenger GA (1991) Expression of an l-alanine dehydrogenase gene in Zymomonas mobilis and excretion of l-alanine. Appl Environ Microbiol 57:1360–1366
Vemuri GN, Altman E, Sangudekar DP, Khodursky AB, Eiteman MA (2006) Overflow metabolism in Escherichia coli during steady-state growth: transcriptional regulation and effect of the redox ratio. Appl Environ Microbiol 72(5):3653–3661
Acknowledgements
Financial support from the U.S. Dept. of Energy Biobased Products Industry Education Program (DE-FG36-01ID14007), the USDA-NRI Program (2003-35504-13666) and the Georgia Experiment Station is gratefully acknowledged. We also acknowledge J. E. Cronan, Jr. for providing us with strain YYC202.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smith, G.M., Lee, S.A., Reilly, K.C. et al. Fed-batch two-phase production of alanine by a metabolically engineered Escherichia coli . Biotechnol Lett 28, 1695–1700 (2006). https://doi.org/10.1007/s10529-006-9142-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-006-9142-3