Abstract
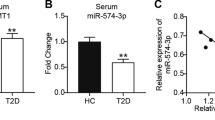
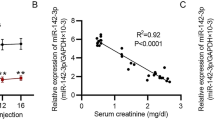
Diabetes mellitus (DM), a familiar disease, is characterized by high blood glucose levels owing to insulin deficiency. Researches have suggested that the incidence rate of diabetes is increasing and it has become an important global epidemic. The type 2 diabetes mellitus (T2DM) is featured with pancreatic β-cell loss and lack of insulin release. Nevertheless, the therapeutic methods that was helpful to improve pancreatic β-cell damage still unclear. Previous report have revealed that tensin homolog deleted on chromosome ten (PTEN) was remarkably enhanced in serum of patients with T2DM, and the lack of PTEN may prevent function deficiency of pancreatic β-cells in DM. However, the underlying mechanisms are rarely illustrated. Our purpose in this report was to illustrated the roles and potential mechanism of microRNA-296-3p (miR-296-3p) in uric acid (UA)-induced pancreatic β-cell injury. The direct target of miR-296-3p was predicted and verified by dual-luciferase reporter system and TargetScan assay. Moreover, Min6 cells were induced by 5 mg/dl UA and the cell proliferation, apoptosis, and insulin release were evaluated using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, flow cytometry and glucose-stimulated insulin secretion (GSIS), respectively. Quantitative reverse transcription PCR (qRT-PCR) and western blot assay were adopted to analyze the levels of miR-296-3p, PTEN and apoptosis-related proteins. TargetScan and Dual-luciferase reporter system confirmed that PTEN directly target miR-296-3p. MiR-296-3p was downregulated in UA-induced Min6 cells and the serum of type 2 diabetes patients, while PTEN was upregulated in UA-induced Min6 cells. Upregulation of miR-296-3p by mimic dramatically promoted miR-296-3p level and decreased PTEN level. Besides, PTEN was over-expressed after PTEN-plasmid transfection. UA treatment prominently decreased cell viability, promoted apoptotic cells, enhanced Bax levels, declined Bcl-2 level as well as decreased insulin release in Min6 cells. MiR-296-3p mimic significantly alleviated UA-induced pancreatic β-cells dysfunction, and PTEN-plasmid eliminated the protective effect of miR-296-3p on insulin release, cell viability, and apoptosis of pancreatic β-cells in UA-stimulated Min6 cells. In summary, our findings revealed that upregulation of miR-296-3p protected pancreatic β-cells functions against UA-induced dysfunction by targeting PTEN, which provides a novel agent for type 2 diabetes treatment.






Similar content being viewed by others
References
Alotaibi A, Perry L, Gholizadeh L, Al-Ganmi A (2017) Incidence and prevalence rates of diabetes mellitus in Saudi Arabia: An overview. J Epidemiol Glob Health 7:211–218
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136:215–233
Birnbaum Y, Nanhwan MK, Ling S, Perez-Polo JR, Ye Y, Bajaj M (2014) PTEN upregulation may explain the development of insulin resistance and type 2 diabetes with high dose statins. Cardiovasc Drugs Ther 28:447–457
Boraie MB, Elnaggar YA, Ahmed MO, Mahmoud AM (2019) Effect of direct acting antiviral therapy of chronic Hepatitis C virus on insulin resistance and Type2 DM in Egyptian patients (prospective study). Diabetes Metab Syndr 13:2641–2646
Bordbar M, Darvishzadeh R, Pazhouhandeh M, Kahrizi D (2020) An overview of genome editing methods based on endonucleases. Mod Genet J 15:75–92
Chakraborty C, Doss CG, Bandyopadhyay S, Agoramoorthy G (2014) Influence of miRNA in insulin signaling pathway and insulin resistance: micro-molecules with a major role in type-2 diabetes. Wiley Interdiscip Rev RNA 5:697–712
Dassano A, Loretelli C, Fiorina P (2019) Idebenone and T2D: A new insulin-sensitizing drug for personalized therapy. Pharmacol Res 139:469–470
David RB, Almeida ED, Cruz LV et al (2014) Diabetes mellitus and glucose as predictors of mortality in primary coronary percutaneous intervention. Arq Bras Cardiol 103:323–330
De Cosmo S, Viazzi F, Pacilli A et al (2015) Serum uric acid and risk of CKD in type 2 diabetes. Clin J Am Soc Nephrol 10:1921–1929
Dihingia A, Ozah D, Ghosh S et al (2018) Vitamin K1 inversely correlates with glycemia and insulin resistance in patients with type 2 diabetes (T2D) and positively regulates SIRT1/AMPK pathway of glucose metabolism in liver of T2D mice and hepatocytes cultured in high glucose. J Nutr Biochem 52:103–114
Elizabeth MM, Alarcon-Aguilar JF, Clara OC, Escobar-Villanueva DCM (2017) Pancreatic beta-Cells and Type 2 Diabetes Development. Curr Diabetes Rev 13:108–121
Ge T, Wu HC, Zhou YY, Shen SM, Zhu LG, You GX (2019) MiR-296-3p may affect the proliferation and migration of non-small cell lung cancer cells via regulating RABL3. Eur Rev Med Pharmacol Sci 23:5823–5830
Gong Z, Yang J, Bai S, Wei S (2020) MicroRNAs regulate granulosa cells apoptosis and follicular development: a review. Asian-Australas J Anim Sci 33:1714–1724
Howard-Thompson A, Khan M, Jones M, George CM (2018) Type 2 diabetes mellitus: outpatient insulin management. Am Fam Physician 97:29–37
Hu Y, Zhao H, Lu J, Xie D, Wang Q, Huang T, Xin H, Hisatome I, Yamamoto T, Wang W, Cheng J (2021) High uric acid promotes dysfunction in pancreatic beta cells by blocking IRS2/AKT signalling. Mol Cell Endocrinol 520:111070
Inoue-Narita T, Hamada K, Sasaki T et al (2008) Pten deficiency in melanocytes results in resistance to hair graying and susceptibility to carcinogen-induced melanomagenesis. Cancer Res 68:5760–5768
Jiang J, Gao Q, Gong Y et al (2018) MiR-486 promotes proliferation and suppresses apoptosis in myeloid cells by targeting Cebpa in vitro. Cancer Med 7:4627–4638
Jung JY, Choi Y, Suh CH, Yoon D, Kim HA (2018) Effect of fenofibrate on uric acid level in patients with gout. Sci Rep 8:16767
Kazemi E, Zargooshi J, Kaboudi M et al (2020) A genome-wide association study to identify candidate genes for erectile dysfunction. Brief Bioinform. https://doi.org/10.1093/bib/bbaa338
Kim MJ, Hopfer H (2016) Mayr M [Uric acid, kidney disease and nephrolithiasis]. Ther Umsch 73:159–165
Kim EM, Jung CH, Song JY, Park JK, Um HD (2018) Pro-apoptotic Bax promotes mesenchymal-epithelial transition by binding to respiratory complex-I and antagonizing the malignant actions of pro-survival Bcl-2 proteins. Cancer Lett 424:127–135
Li H, Wu J, Zhu L et al (2018) Insulin degrading enzyme contributes to the pathology in a mixed model of Type 2 diabetes and Alzheimer’s disease: possible mechanisms of IDE in T2D and AD. Biosci Rep. https://doi.org/10.1042/BSR20170862
Liu B, Li J, Cairns MJ (2014) Identifying miRNAs, targets and functions. Brief Bioinform 15:1–19
Lu TX, Rothenberg ME (2018) MicroRNA. J Allergy Clin Immunol 141:1202–1217
Lytvyn Y, Perkins BA, Cherney DZ (2015) Uric acid as a biomarker and a therapeutic target in diabetes. Can J Diabetes 39:239–246
Pordzik J, Jakubik D, Jarosz-Popek J, Wicik Z, Eyileten C, De Rosa S, Indolfi C, Siller-Matula JM, Czajka P, Postula M (2019) Significance of circulating microRNAs in diabetes mellitus type 2 and platelet reactivity: bioinformatic analysis and review. Cardiovasc Diabetol 18:113
Samad A, James A, Wong J et al (2014) Insulin protects pancreatic acinar cells from palmitoleic acid-induced cellular injury. J Biol Chem 289:23582–23595
Segerstrom AB, Glans F, Eriksson KF, Holmback AM, Groop L, Thorsson O, Wollmer P (2010) Impact of exercise intensity and duration on insulin sensitivity in women with T2D. Eur J Intern Med 21:404–408
Shirjang S, Mansoori B, Asghari S, Duijf P, Mohammadi A, Gjerstorff M, Baradaran B (2019) MicroRNAs in cancer cell death pathways: Apoptosis and necroptosis. Free Radic Biol Med 139:1–15
Thong EP, Codner E, Laven J, Teede H (2020) Diabetes: a metabolic and reproductive disorder in women. Lancet Diabetes Endocrinol 8:134–149
Titov SE, Ivanov MK, Demenkov PS et al (2019) Combined quantitation of HMGA2 mRNA, microRNAs, and mitochondrial-DNA content enables the identification and typing of thyroid tumors in fine-needle aspiration smears. BMC Cancer 19:1010
Viazzi F, Piscitelli P, Giorda C et al (2017) Metabolic syndrome, serum uric acid and renal risk in patients with T2D. PLoS ONE 12:e0176058
Vilchis-Flores LH, Barajas-Medina GA, Villa-Martinez AK et al (2019) Pancreatic β-cell dysfunction in normoglycemic patients and risk factors. Acta Diabetol 56:1305–1314
Wang L, Liu Y, Yan LS, Nguyen KT, Schroer SA, Suzuki A, Mak TW, Gaisano H, Woo M (2010) Deletion of Pten in pancreatic β-cells protects against deficient β-cell mass and function in mouse models of type 2 diabetes. Diabetes 59:3117–3126
Wang Y, Hai B, Niu X, Ai L, Cao Y, Li R, Li Y (2017) Chronic intermittent hypoxia disturbs insulin secretion and causes pancreatic injury via the MAPK signaling pathway. Biochem Cell Biol 95:415–420
Wang L, Chen R, Zhang Y (2019) miR-296-3p targets APEX1 to suppress cell migration and invasion of non-small-cell lung cancer. Oncol Lett 18:2612–2618
Wu X, Yu T, Ji N et al (2019) IL6R inhibits viability and apoptosis of pancreatic β-cells in type 2 diabetes mellitus via regulation by miR-22 of the JAK/STAT signaling pathway. Diabetes Metab Syndr Obes 12:1645–1657
Xin Y, Zhang H, Jia Z, Ding X, Sun Y, Wang Q, Xu T (2018a) Resveratrol improves uric acid-induced pancreatic beta-cells injury and dysfunction through regulation of miR-126. Biomed Pharmacother 102:1120–1126
Xin Y, Wang K, Jia Z, Xu T, Xu Q, Zhang C, Liu J, Chen R, Du Z, Sun J (2018b) Zurampic protects pancreatic beta-Cells from high uric acid induced-damage by inhibiting URAT1 and inactivating the ROS/AMPK/ERK pathways. Cell Physiol Biochem 47:1074–1083
Yang F, Chen Y, Xue Z, Lv Y, Shen L, Li K, Zheng P, Pan P, Feng T, Jin L, Yao Y (2020) High-throughput sequencing and exploration of the lncRNA-circRNA-miRNA-mRNA network in type 2 diabetes mellitus. Biomed Res Int 2020:8162524
Zhang T, Qiu F (2020) Icariin Protects Mouse Insulinoma Min6 Cell Function by Activating the PI3K/AKT Pathway. Med Sci Monit 26:e924453
Funding
This project was supported by the Science and Technology Innovation Fund Project of Longhua District, Shenzhen; Chinese character of Shenzhen Longhua science and technology (2017) NO.2 (Grant No. 20160831A1030200).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cheng, M., Guo, Y., Zhong, W. et al. Abnormal Expression of microRNA-296-3p in Type 2 Diabetes Patients and its Role in Pancreatic β-Cells Function by Targeting Tensin Homolog Deleted on Chromosome Ten. Biochem Genet 60, 39–53 (2022). https://doi.org/10.1007/s10528-021-10083-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-021-10083-6