Abstract
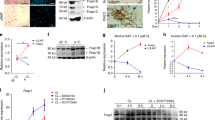
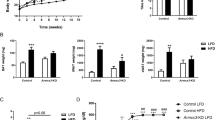
The tissue distribution of activin receptor-like kinase 7 (Alk7) expression, the signaling ability of Alk7 variants, and Alk7 expression in response to β3-adrenergic receptor activation were examined. Expression levels of Alk7 varied greatly among tissues but were highest in white adipose tissue and brown adipose tissue. In addition to full-length Alk7 (Alk7-v1), Alk7-v3, an Alk7 variant, was expressed in adipose tissues, brain, and ovary. Nodal transmits signals via Alk7 in cooperation with its coreceptor, Cripto. Evaluation of the ability of Alk7 variants to confer Nodal signaling using luciferase-based reporter assays showed that Alk7-v3 does not transmit Nodal-Cripto-mediated signals. Expression of Alk7 was down-regulated in brown but not in white adipose tissue treated with CL316,243, a β3-adrenergic receptor agonist. These results suggest involvement of Alk7 in modulation of metabolism in the adipose tissues in response to β3-adrenergic receptor activation.




Similar content being viewed by others
References
Andersson O, Reissmann E, Jörnvall H, Ibáñez CF (2006) Synergistic interaction between Gdf1 and Nodal during anterior axis development. Dev Biol 293:370–381
Andersson O, Korach-Andre M, Reissmann E, Ibáñez CF, Bertolino P (2008) Growth/differentiation factor 3 signals through ALK7 and regulates accumulation of adipose tissue and diet-induced obesity. Proc Natl Acad Sci USA 105:7252–7256
Barbatelli G, Murano I, Madsen L, Hao Q, Jimenez M, Kristiansen K, Giacobino JP, De Matteis R, Cinti S (2010) The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am J Physiol Endocrinol Metab 298:E1244–E1253
Bertolino P, Holmberg R, Reissmann E, Andersson O, Berggren PO, Ibáñez CF (2008) Activin B receptor ALK7 is a negative regulator of pancreatic β-cell function. Proc Natl Acad Sci USA 105:7246–7251
Brennan J, Lu CC, Norris DP, Rodriguez TA, Beddington RS, Robertson EJ (2001) Nodal signalling in the epiblast patterns the early mouse embryo. Nature 411:965–969
Brennan J, Norris DP, Robertson EJ (2002) Nodal activity in the node governs left-right asymmetry. Genes Dev 16:2339–2344
Cannon B, Nedergaard J (2004) Brown adipose tissue: function and physiological significance. Physiol Rev 84:277–359
Carlsson LM, Jacobson P, Walley A, Froguel P, Sjöström L, Svensson PA, Sjöholm K (2009) ALK7 expression is specific for adipose tissue, reduced in obesity and correlates to factors implicated in metabolic disease. Biochem Biophys Res Commun 382:309–314
Cinti S, Cancello R, Zingaretti MC, Ceresi E, De Matteis R, Giordano A, Himms-Hagen J, Ricquier D (2002) CL316,243 and cold stress induce heterogeneous expression of UCP1 mRNA and protein in rodent brown adipocytes. J Histochem Cytochem 50:21–31
Dennler S, Itoh S, Vivien D, ten Dijke P, Huet S, Gauthier JM (1998) Direct binding of Smad3 and Smad4 to critical TGFβ-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J 17:3091–3100
Feng XH, Derynck R (2005) Specificity and versatility in TGF-b signaling through Smads. Annu Rev Cell Dev Biol 21:659–693
Inohara N, Koseki T, Chen S, Wu X, Núñez G (1998) CIDE, a novel family of cell death activators with homology to the 45 kDa subunit of the DNA fragmentation factor. EMBO J 17:2526–2533
Jörnvall H, Reissmann E, Andersson O, Mehrkash M, Ibáñez CF (2004) ALK7, a receptor for nodal, is dispensable for embryogenesis and left–right patterning in the mouse. Mol Cell Biol 24:9383–9389
Kang Y, Reddi AH (1996) Identification and cloning of a novel type I serine/threonine kinase receptor of the TGF-β/BMP superfamily in rat prostate. Biochem Mol Biol Int 40:993–1001
Kubota N, Yano W, Kubota T, Yamauchi T, Itoh S, Kumagai H, Kozono H, Takamoto I, Okamoto S, Shiuchi T, Suzuki R, Satoh H, Tsuchida A, Moroi M, Sugi K, Noda T, Ebinuma H, Ueta Y, Kondo T, Araki E, Ezaki O, Nagai R, Tobe K, Terauchi Y, Ueki K, Minokoshi Y, Kadowaki T (2007) Adiponectin stimulates AMP-activated protein kinase in the hypothalamus and increases food intake. Cell Metab 6:55–68
Lorentzon M, Hoffer B, Ebendal T, Olson L, Tomac A (1996) Habrec1, a novel serine/threonine kinase TGF-b type I-like receptor, has a specific cellular expression suggesting function in the developing organism and adult brain. Exp Neurol 142:351–360
Miyazono K, Kamiya Y, Morikawa M (2010) Bone morphogenetic protein receptors and signal transduction. J Biochem 147:35–51
Murakami M, Iwata Y, Funaba M (2007) Expression and transcriptional activity of alternative splice variants of Mitf exon 6. Mol Cell Biochem 303:251–257
Murakami M, Kawachi H, Ogawa K, Nishino Y, Funaba M (2009) Receptor expression modulates the specificity of transforming growth factor-b signaling pathways. Genes Cells 14:469–482
Ooishi R, Shirai M, Funaba M, Murakami M (2012) Microphthalmia-associated transcription factor is required for mature myotube formation. Biochim Biophys Acta 1820(2):76–83
Rasband WS (2012) ImageJ, 1997–2012. U.S. National Institutes of Health, Bethesda. http://imagej.nih.gov/ij/. Accessed June 2012
Reissmann E, Jörnvall H, Blokzijl A, Andersson O, Chang C, Minchiotti G, Persico MG, Ibáñez CF, Brivanlou AH (2001) The orphan receptor ALK7 and the Activin receptor ALK4 mediate signaling by Nodal proteins during vertebrate development. Genes Dev 15:2010–2022
Roberts HJ, Hu S, Qiu Q, Leung PC, Caniggia I, Gruslin A, Tsang B, Peng C (2003) Identification of novel isoforms of activin receptor-like kinase 7 (ALK7) generated by alternative splicing and expression of ALK7 and its ligand, Nodal, in human placenta. Biol Reprod 68:1719–1726
Rydén M, Imamura T, Jörnvall H, Belluardo N, Neveu I, Trupp M, Okadome T, ten Dijke P, Ibáñez CF (1996) A novel type I receptor serine-threonine kinase predominantly expressed in the adult central nervous system. J Biol Chem 271:30603–30609
Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, Ishibashi J, Cohen P, Cinti S, Spiegelman BM (2011) Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Invest 121:96–105
Shimomura I, Hammer RE, Ikemoto S, Brown MS, Goldstein JL (1999) Leptin reverses insulin resistance and diabetes mellitus in mice with congenital lipodystrophy. Nature 401:73–76
Tojo M, Hamashima Y, Hanyu A, Kajimoto T, Saitoh M, Miyazono K, Node M, Imamura T (2005) The ALK-5 inhibitor A-83-01 inhibits Smad signaling and epithelial-to-mesenchymal transition by transforming growth factor-β. Cancer Sci 96:791–800
Tsuchida K, Sawchenko PE, Nishikawa S, Vale WW (1996) Molecular cloning of a novel type I receptor serine/threonine kinase for the TGF-β superfamily from rat brain. Mol Cell Neurosci 7:467–478
Tsuchida K, Nakatani M, Yamakawa N, Hashimoto O, Hasegawa Y, Sugino H (2004) Activin isoforms signal through type I receptor serine/threonine kinase ALK7. Mol Cell Endocrinol 220:59–65
Watanabe R, Yamada Y, Ihara Y, Someya Y, Kubota A, Kagimoto S, Kuroe A, Iwakura T, Shen ZP, Inada A, Adachi T, Ban N, Miyawaki K, Sunaga Y, Tsuda K, Seino Y (1999) The MH1 domains of smad2 and smad3 are involved in the regulation of the ALK7 signals. Biochem Biophys Res Commun 254:707–712
Whitman M (2001) Nodal signaling in early vertebrate embryos: themes and variations. Dev Cell 1(5):605–617
Yoshida T, Sakane N, Wakabayashi Y, Umekawa T, Kondo M (1994) Anti-obesity and anti-diabetic effects of CL 316,243, a highly specific β3-adrenoceptor agonist, in yellow KK mice. Life Sci 54:491–498
Acknowledgments
We thank Dr. Peter ten Dijke for providing plasmid. This work was supported by Kakenhi from The Japan Society for the Promotion of Science and a research project grant awarded by the Azabu University.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Murakami, M., Shirai, M., Ooishi, R. et al. Expression of Activin Receptor-like Kinase 7 in Adipose Tissues. Biochem Genet 51, 202–210 (2013). https://doi.org/10.1007/s10528-012-9555-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-012-9555-8