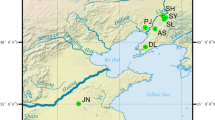
A total of 90 ESTs from normal and 157 from subtractive ovarian cDNA libraries of the giant tiger shrimp (Penaeus monodon) were sequenced. SSCP analysis of disulfide isomerase (DSl), zinc finger protein (ZFP), PMO920, and PMT1700 was carried out for population genetic studies of P. monodon in Thai waters. The number of codominant alleles per locus for overall samples was 6 for PMO920, 5 for PMT1700, and 12 for ZFP, and there were 19 dominant alleles for DSI. The observed heterozygosity of each geographic sample was 0.3043–0.5128 for PMO920, 0.3462–0.4643 for PMT1700, and 0.5000–0.8108 for ZFP. Linkage disequilibrium analysis indicated that genotypes of these loci segregate randomly (P > 0.05). Low genetic distance was found between pairs of geographic samples (0.0077–0.0178). The neighbor-joining tree constructed from the average genetic distance of overall loci allocated the Andaman samples (Satun, Trang, and Phangnga) into one cluster, and Chumphon and Trat into other clusters. Geographic differentiation between Satun-Trat and Satun-Phangnga was found only at the ZFP locus (P < 0.05), suggesting low degrees of genetic subdivision of Thai P. monodon.



Similar content being viewed by others
References
Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. (1990). Basic local alignment search tool. J. Mol. Biol. 215:403–410
Avise J. C. (1994). Molecular Markers, Natural History, and Evolution. Chapman and Hall, New York, pp. 511
Benzie J. A. H. (2000). Population genetic structure in penaeid prawns. Aquaculture Res. 31:95–119
Benzie J. A. H., Ballment E., Forbes A. T., Demetriades N. T., Suguma K. (2002). Mitochondrial DNA variation in Indo-Pacific populations of the giant tiger prawn, Penaeus monodon. Mol. Ecol. 11:2553–2569
Carvalho G. R., Hauser L. (1994). Molecular genetics and the stock concept in fisheries. Rev. Fish Biol. Fisheries 4:326–350
Crow J. F., Kimura M. (1965). Evolution in sexual and asexual populations. Am. Nature 99:439–450
Felsenstein, J. (1993). PHYLIP (Phylogenetic Inference Package) Version 3.5c. Seattle: Department of Genetics, University of Washington
Guo S. W., Thompson E. A. (1992). Performing the extract test of Hardy-Weinberg proportion for multiple alleles. Biometrics 48:361–372
Klinbunga S., Sodsuk S., Penman D. J., McAndrew B. J. (1996). An improved protocol for total DNA isolation and visualization of mtDNA-RFLP(s) in tiger prawn, Penaeus monodon. Thai J. Aquat. Sci. 3:36–41
Klinbunga S., Penman D. J., McAndrew B. J., Tassanakajon A. (1999). Mitochondrial DNA diversity in three populations of the giant tiger shrimp Penaeus monodon. Mar. Biotechnol. 1:113–121
Klinbunga S., Siludjai D., Wuthijinda W., Tassanakajon A., Jarayabhand A., Menasveta P. (2001). Genetic heterogeneity of the giant tiger shrimp (Penaeus monodon) in Thailand revealed by RAPD and mtDNA-RFLP analyses. Mar. Biotechnol. 3:428–438
Leelatanawit R., Klinbunga S., Puanglarp N., Tassanakajon A., Jarayabhand P., Hirono I., Aoki T., Menasveta P. (2004). Isolation and characterization of differentially expressed genes in ovaries and testes of the giant tiger shrimp (Penaeus monodon). Mar. Biotechnol. 6:S506-S510
Maggioni R., Rogers A. D., MaClean N., D’Incao F. (2001). Molecular phylogeny of western Atlantic Farfantepenaeus and Litopenaeus shrimp based on mitochondrial 16S partial sequences. Mol. Phylogenet. Evol. 18:66–73
Maniatis T., Fritsch E. F., Sambrook J. (1982). Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press, New York, pp. 545.
Nei M. (1987). Molecular Evolutionary Genetics, Columbia University Press, New York, pp. 512.
Nei M., Li W. H. (1979). Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc. Natl. Acad. Sci. USA 76:5269–5273
Raymond M., Rousset F. (1995). Genepop (Version 1.2): A population genetics software for exact tests and ecumenicism. J. Hered. 86:248–249
Rice W. R. (1989). Analyzing tables of statistical tests. Evolution 41:223–225
Saitou N., Nei M. (1987). The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4:406–425
Supungul P., Sootanan P., Klinbunga S., Kamonrat W., Jarayabhand P., Tassanakajon A. (2000). Microsatellite polymorphism and the population structure of the black tiger shrimp (Penaeus monodon) in Thailand. Mar. Biotechnol. 2:339–347
Tassanakajon A., Pongsomboon S., Jarayabhand P., Klinbunga S., Boonsaeng V. (1998). Genetic structure in wild populations of the black tiger shrimp (Penaeus monodon) using randomly amplified polymorphic DNA analysis. J. Mar. Biotechnol. 6:249–254
Ward R. D., Grewe P. M. (1994). Appraisal of molecular genetic techniques in fisheries. Rev. Fish Biol. Fisheries 4:300–325
Weir B. S., Cockerham C. C. (1984). Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Withyachumnarnkul, B., Boonsaeng, W., Flegel, T. W., Panyim, S., and Wongteerasupaya, C. (1998). Domestication and selective breeding of Penaeus monodon in Thailand. In Felgel, T. (ed.), Proceedings to the Special Session on Advances in Shrimp Biotechnology, Fifth Asian Fisheries Forum: International Conference on Fisheries and Food Security Beyond the Year 2000. 11–14 November 1998. Chiangmai, Thailand, pp. 73–77
Xu Z., Primavera J. H., de la Pena L. D., Pertit P., Belak J., Alcivar-Warren A. (2001). Genetic diversity of wild and cultured black tiger shrimp (Penaeus monodon) in the Philippines using microsatellites. Aquaculture 199:13–40
Acknowledgments
The authors thank two anonymous referees for their useful comments. This research was supported by the National Center for Genetic Engineering and Biotechnology (BIOTEC), Thailand, awarded to SK.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klinbunga, S., Preechaphol, R., Thumrungtanakit, S. et al. Genetic Diversity of the Giant Tiger Shrimp (Penaeus monodon) in Thailand Revealed by PCR-SSCP of Polymorphic EST-Derived Markers. Biochem Genet 44, 222–236 (2006). https://doi.org/10.1007/s10528-006-9027-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-006-9027-0