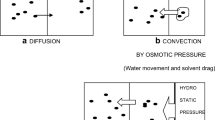
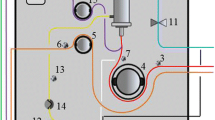
Risks inherent in the use of portable artificial kidney apparatuses in the out-of-hospital environment are analyzed. Technical solutions for increasing the reliability of the portable equipment are discussed.
Similar content being viewed by others
References
Gura V., Macy A.S., Beizai M., Ezon C., Golper T.A., Clin. J. Am. Soc. Nephrol., 4, 1441-1448 (2009).
Ronco C., Davenport A., Gura V., Nefrologia, 31, 9-16 (2011).
Davenport A., In: Advances in Peritoneal Dialysis, 28 (2012).
Lee D.B., Roberts M., Clin. Exp. Nephrol., 12, No. 3, 171-180 (2008).
Buffington D.A., Westover A.J., Johnston K.A., Humes H.D., Transl. Res., 163, No. 4, 342-351 (2014).
Wester M. et al., Artif. Organs, 38, No. 12, 998-1006 (2014).
GOST ISO 14971-2011. Medical Devices. Application of Risk Management to Medical Devices.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Meditsinskaya Tekhnika, Vol. 49, No. 6, Nov.-Dec., 2015, pp. 44-47.
Rights and permissions
About this article
Cite this article
Bazaev, N.A., Grinval’d, V.M. & Pozhar, K.V. Prospects for Use of Portable Equipment for Extracorporeal Blood Purification. Biomed Eng 49, 385–388 (2016). https://doi.org/10.1007/s10527-016-9572-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10527-016-9572-8