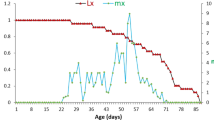
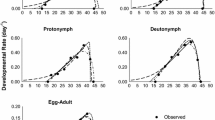
Abstract
Fecundity and feeding of two introduced sibling biological control species, Galerucella calmariensis and G. pusilla (Coleoptera: Chrysomelidae) on purple loosestrife, Lythrum salicaria L. (Lythraceae) were compared at constant temperatures of 12.5, 15, 20, 25, and 27.5 °C. Larval feeding was also carried out at 30 °C, but at this temperature, larvae developed only to the L2 stage and none pupated. Thus, data for this temperature were not used in the analysis. There were significant species × temperature interactions in fecundity. Of the two species, Galerucella pusilla laid more eggs. Although egg production of both species was lowest at 12.5 °C and increased to 20 °C, at higher temperatures, the two species reacted differently. From 25 to 27.5 °C, egg production decreased for G. pusilla, but G. calmariensis fecundity peaked at 27.5 °C. Significant temperature × species × life-stage interactions were also observed in feeding. For each species, the amount of feeding varied with temperature and stage of development. Galerucella pusilla adults consumed more foliage at 15, 20, and 27.5 °C. However, at 12.5 °C G. calmariensis adults fed more than G. pusilla. G. pusilla larvae consumed an average of 25% less foliage than G. calmariensis. The lower larval consumption of G. pusilla suggests that when food is limited, G. pusilla larvae may have a higher survival rate because of its ability to complete larval development with less food and produce more progeny due to its greater fecundity. When food is not limited neither species would have a competitive advantage and both species could coexist temporally and spatially. However, since G. calmariensis larvae consumed more leaf material, the larval stage of this species would have a greater impact on purple loosestrife than G. pusilla.
Similar content being viewed by others
References
Aarssen L.W. (1983). Ecological combining ability and competitive combining ability in plants: towards a general evolutionary theory of coexistence in systems of competition. Am. Nat. 122: 707–731
Ågren G.I., Fagerström T. (1984). Limiting dissimilarity in plants: randomness prevents exclusion of species with similar competitive abilities. Oikos 43:369–351
Atkinson W.D., Shorrocks B. (1981). Competition on a divided and ephemeral resource: a simulation model. J. Anim. Ecol. 50:461–471
Bartelt R.J., Cossé A.A., Zilowski B.W., Weislender D., Grode S.H., Wiedenmann R.N., Post S.L. (2006). Dimethylfuran-lactone pheromone from males of Galerucella calmariensis and Galerucella pusilla. J. Chem. Ecol. 32: 693–712
Beaver R.A. (1977). Non-equilibrium ‚island’ communities: Diptera breeding in dead snails. J. Anim. Ecol. 46:783–798
Blossey B. (1995). Coexistence of two leaf-beetles in the same fundamental niche. Distribution, adult phenology, and oviposition. Oikos 74:225–234
Dech J.P., Nosko P. (2002). Population establishment, dispersal, and impact of Galerucella pusilla and G. calmariensis, introduced to control purple loosestrife in central Ontario. Biol. Control. 23:228–0236
Goldberg D.E., Barton A.M. (1992). Patterns and consequences of interspecific competition in natural communities: a review of field experiments with plants. Am. Nat. 139:771–801
Grevstad F.S., Herzig A.L. (1996). Quantifying the effects of distance and conspecifics on colonization: experiments and models using the loosestrife leaf beetle, G. calmariensis. Oecologia 110:60–68
Gurevitch J.L., Morrow L., Wallace A., Walsh J.S. (1992). A meta-analysis of competition in field experiments. Am. Nat. 140:539–572
Hairston N.G., Smith F.E., Slobodkin L.B. (1960). Community structure, population control, and competition. Am. Nat. 140:421–425
Hassell M.P., Comins H.N. (1976). Discrete time models for two-species competition. Theor. Popul. Biol. 9:202–221
Hight S.D., Blossey B., Laing J., Declerck-Floate R. (1995). Establishment of insect biological control agents from Europe against Lythrum salicaria in North America. Environ. Entomol. 24:967–977
Inouye B. (1999). Integrating nested spatial scales: implications for the coexistence of competitors on a patchy resource. J. Anim. Ecology. 68:150–162
Iwao K., Rausher M.D. (1997). Evolution of plant resistance to multiple herbivores: quantifying diffuse coevolution. Am Nat. 149:316–335
Kok, L.T., T.J. McAvoy and W.T. Mays, 2000. Successful establishment of exotic agents for classical biological control of invasive weeds in Virginia. In: N. R. Spencer (ed), Proc. X Int. Symp. Biol. Contr. Weeds, July 4–10, 1999, Montana State University, Montana. pp. 59–65.
Landis D.A., Sebolt D.C., Haas M.J., Klepinger M. (2003). Establishment and impact of Galerucella calmariensis L. (Coleoptera: Chrysomelidae) on Lythrum salicaria L. and associated plant communities in Michigan. Biol. Control. 28:78–91
Lindgren C.J. (1997). Oviposition site preferences of Galerucella calmariensis and G. pusilla, biological control agents of purple loosestrife (Lythrum salicaria). Weed Technol. 11:824–827
Manguin S., White R., Blossey B., Hight S.D. (1993). Genetics, taxonomy, and ecology of certain species of Galerucella (Coleoptera: Chrysomelidae). Ann. Entomol. Soc. Am. 86:397–410
McAvoy T.J., Kok L.T., Mays W.T. (1997). Phenology of an established population of Galerucella calmariensis (L.) and G. pusilla (Duft.) (Coleoptera: Chrysomelidae) on purple loosestrife, Lythrum salicaria L. (Lythraceae), in southwest Virginia. Biol. Control. 9:106–111
McAvoy T.J., Kok L.T. (2004). Temperature dependent development and survival of two sympatric species, Galerucella calmariensis and G. pusilla, on purple loosestrife. BioControl 49:467–480
Nokkala S., Nokkala C. (1987). Chromosome numbers and chromosomal polymorphism in Finnish species of Galerucella Crotch (Coleoptera: Chrysomelidae). Hereditas 106:51–58
Piper G.L. (1996). Biological control of the wetlands weed purple loosestrife (Lythrum salicaria) in the Pacific northwestern United States. Hydrobiologia 340:291–294
SAS Institute, 2005. SAS User’s Guide: Statistics, Release 9.1. SAS Institute, Cary, NC.
Schmida A., Ellner S. (1984). Coexistence of plant species with similar niches. Vegetation 58:29–55
Schoener T.W. (1983). Field experiments on interspecific competition. Am. Nat. 122:240–285
Schooler S.S., McEvoy P.B. (2006). Relationship between insect density and plant damage for the golden loosestrife beetle, Galerucella pusilla, on purple loosestrife (Lythrum salicaria). Biol. Control. 36:100–105
Schooler, S.S., 1998. Biological control of purple loosestrife Lythrum salicaria by two chrysomelid beetles, Galerucella pusilla and G. calmariensis. MSc. Thesis, Oregon State University, Corvallis, OR.
Stamm Katovich E.J., Ragsdale D.W., Skinner L.C., Becker R.L. (200). Effect of Galerucella spp. feeding on seed production in purple loosestrife. Weed Sci. 49:190–194
Wiebe A.P., Obrycki J.J. (2004). Quantitative assessment of predation of eggs and larvae of Galerucella pusilla in Iowa. Biol. Control. 31:16–28
Acknowledgements
We thank Dr. Susanne Aref and Li Wang, Department of Statistics, Virginia Polytechnic Institute and State University for their assistance with the statistical analysis. Thanks also to two anonymous reviewers who provided constructive comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McAvoy, T.J., Kok, L.T. Fecundity and feeding of Galerucella calmariensis and G. pusilla on Lythrum salicaria . BioControl 52, 351–363 (2007). https://doi.org/10.1007/s10526-006-9042-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-006-9042-4