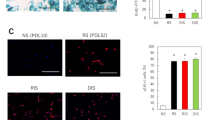
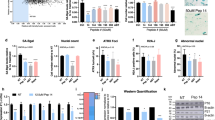
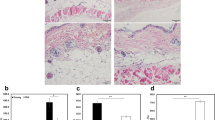
Abstract
Aging negatively affects the appearance and texture of the skin owing to the accumulation of senescent fibroblasts within the dermis. Senescent cells undergo abnormal remodeling of collagen and the extracellular matrix through an inflammatory histolytic senescence-associated secretory phenotype (SASP). Therefore, suppression of SASP in senescent cells is essential for the development of effective skin anti-aging therapies. Ectonucleotide pyrophosphatase/phosphodiesterase family member 5 (ENPP5), an extracellular signaling molecule, has been implicated in vascular aging and apoptosis; however, its role in SASP remains unclear. Therefore, this study aimed to investigate the role of ENPP5 in SASP and skin aging using molecular techniques. We investigated the effects of siRNA-mediated ENPP5 knockdown, human recombinant ENPP5 (rENPP5) treatment, and lentiviral overexpression of ENPP5 on SASP and aging in human skin fibroblasts. Additionally, we investigated the effect of siRNA-mediated ENPP5 knockdown on the skin of C57BL/6 mice. We found that ENPP5 was significantly expressed in replication-aged and otherwise DNA-damaged human skin fibroblasts and that treatment with human rENPP5 and lentiviral overexpression of ENPP5 promoted SASP and senescence. By contrast, siRNA-mediated knockdown of ENPP5 suppressed SASP and the expression of skin aging-related factors. Additionally, ENPP5 knockdown in mouse skin ameliorated the age-related reduction of subcutaneous adipose tissue, the panniculus carnosus muscle layer, and thinning of collagen fibers. Conclusively, these findings suggest that age-related changes may be prevented through the regulation of ENPP5 expression to suppress SASP in aging cells, contributing to the development of anti-aging treatments for the skin.







Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Adamus J, Aho S, Meldrum H, Bosko C, Lee JM (2014) p16INK4A influences the aging phenotype in the living skin equivalent. J Invest Dermatol 134:1131–1133. https://doi.org/10.1038/jid.2013.468
Alimbetov D, Davis T, Brook AJ, Cox LS, Faragher RG, Nurgozhin T, Zhumadilov Z, Kipling D (2016) Suppression of the senescence-associated secretory phenotype (SASP) in human fibroblasts using small molecule inhibitors of p38 MAP kinase and MK2. Biogerontology 17:305–315. https://doi.org/10.1007/s10522-015-9610-z
Baar MP, Brandt RMC, Putavet DA, Klein JDD, Derks KWJ, Bourgeois BRM, Stryeck S, Rijksen Y, van Willigenburg H, Feijtel DA, van der Pluijm I, Essers J, van Cappellen WA, van IJcken WF, Houtsmuller AB, Pothof J, de Bruin RWF, Madl T, Hoeijmakers JHJ, Campisi J, de Keizer PLJ (2017) Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169:132–147e16. https://doi.org/10.1016/j.cell.2017.02.031
Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, Kirkland JL, van Deursen JM (2011) Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479:232–236. https://doi.org/10.1038/nature10600
Childs BG, Durik M, Baker DJ, van Deursen JM (2015) Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med 21:1424–1435. https://doi.org/10.1038/nm.4000
Coppé JP, Desprez PY, Krtolica A, Campisi J (2010) The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 5:99–118. https://doi.org/10.1146%2Fannurev-pathol-121808-102144
Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR, Laberge RM, Vijg J, Van Steeg H, Dollé MET, Hoeijmakers JHJ, de Bruin A, Hara E, Campisi J (2014) An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell 31:722–733. https://doi.org/10.1016/j.devcel.2014.11.012
Demaria M, Desprez PY, Campisi J, Velarde MC (2015) Cell autonomous and non-autonomous effects of senescent cells in the skin. J Invest Dermatol 135:1722–1726. https://doi.org/10.1038/jid.2015.108
Fisher DI, Safrany ST, Strike P, McLennan AG, Cartwright JL (2002) Nudix hydrolases that degrade dinucleoside and diphosphoinositol polyphosphates also have 5-phosphoribosyl 1-pyrophosphate (PRPP) pyrophosphatase activity that generates the glycolytic activator ribose 1,5-bisphosphate. J Biol Chem 277:47313–47317. https://doi.org/10.1074/jbc.m209795200
Fitsiou E, Pulido T, Campisi J, Alimirah F, Demaria M (2021) Cellular senescence and the senescence-associated secretory phenotype as drivers of skin photoaging. J Invest Dermatol 141:1119–1126. https://doi.org/10.1016/j.jid.2020.09.031
Ghosh K, Capell BC (2016) The senescence-associated secretory phenotype: critical effector in skin cancer and aging. J Invest Dermatol 136:2133–2139. https://doi.org/10.1016%2Fj.jid.2016.06.621
Hao W, Shan W, Wan F, Luo J, Niu Y, Zhou J, Zhang Y, Xu N, Xie W (2023) Canagliflozin delays aging of HUVECs induced by palmitic acid via the ROS/p38/JNK pathway. Antioxid (Basel) 12:838. https://doi.org/10.3390/antiox12040838
Hayflick L, Moorhead PS (1961) The serial cultivation of human diploid cell strains. Exp Cell Res 25:585–621. https://doi.org/10.1016/0014-4827(61)90192-6
Higashiguchi M, Murakami H, Akita H, Kobayashi S, Takahama S, Iwagami Y, Yamada D, Tomimaru Y, Noda T, Gotoh K, Doki Y, Yamamoto T, Eguchi H (2023) The impact of cellular senescence and senescenceassociated secretory phenotype in cancerassociated fibroblasts on the malignancy of pancreatic cancer. Oncol Rep 49:98. https://doi.org/10.3892/or.2023.8535
Jin P, Li X, Xia Y, Li H, Li X, Yang ZY, Wang Z, Xu C, Fang T, Zhou D, Xiong X, Wang SY, Xu Sm Gao Q (2023) Bepotastine sensitizes ovarian cancer to PARP inhibitors through suppressing NF-κB-triggered SASP in cancer-associated fibroblasts. Mol Cancer Ther 22:447–458. https://doi.org/10.1158/1535-7163.mct-22-0396
Kim H, Jang J, Song MJ, Kim G, Park CH, Lee DH, Lee SH, Chung JH (2022) Attenuation of intrinsic ageing of the skin via elimination of senescent dermal fibroblasts with senolytic drugs. J Eur Acad Dermatol Venereol 36:1125–1135. https://doi.org/10.1111/jdv.18051
Krishnamurthy J, Torrice C, Ramsey MR, Kovalev GI, Al-Regaiey K, Su L, Sharpless NE (2004) Ink4a/Arf expression is a biomarker of aging. J Clin Invest 114:1299–1307. https://doi.org/10.1172/jci22475
Lago JC, Puzzi MB (2019) The effect of aging in primary human dermal fibroblasts. PLoS ONE 14:e0219165. https://doi.org/10.1371/journal.pone.0219165
Li H, Wang X, Lu X, Zhu H, Li S, Duan S, Zhao X, Zhang F, Alterovitz G, Wang F, Li Q, Tian XL, Xu M (2019) Co-expression network analysis identified hub genes critical to triglyceride and free fatty acid metabolism as key regulators of age-related vascular dysfunction in mice. Aging 11:7620–7638. https://doi.org/10.18632/aging.102275
Lovell CR, Smolenski KA, Duance VC, Light ND, Young S, Dyson M (1987) Type I and III collagen content and fibre distribution in normal human skin during ageing. Br J Dermatol 117:419–428. https://doi.org/10.1111/j.1365-2133.1987.tb04921.x
Puzzo D, Bizzoca A, Loreto C, Guida CA, Gulisano W, Frasca G, Bellomo M, Castorina S, Gennarini G, Palmeri A (2015) Role of F3/contactin expression profile in synaptic plasticity and memory in aged mice. Neurobiol Aging 36:1702–1715. https://doi.org/10.1016/j.neurobiolaging.2015.01.004
Ressler S, Bartkova J, Niederegger H, Bartek J, Scharffetter-Kochanek K, Jansen-Dürr P, Wlaschek M (2006) p16INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging Cell 5:379–389. https://doi.org/10.1111/j.1474-9726.2006.00231.x
Rognoni E, Watt FM (2018) Skin cell heterogeneity in development, wound healing, and cancer. Trends Cell Biol 28:709–722. https://doi.org/10.1016/j.tcb.2018.05.002
Schmitt CA, Wang B, Demaria M (2022) Senescence and cancer – role and therapeutic opportunities. Nat Rev Clin Oncol 19:619–636. https://doi.org/10.1038/s41571-022-00668-4
Sharma AK, Roberts RL, Benson RD Jr, Pierce JL, Yu K, Hamrick MW, McGee-Lawrence ME (2020) The senolytic drug navitoclax (ABT-263) causes trabecular bone loss and impaired osteoprogenitor function in aged mice. Front Cell Dev Biol 8:354. https://doi.org/10.3389/fcell.2020.00354
Sharma K, Sarkar J, Trisal A, Ghosh R, Dixit A, Singh AK (2023) Targeting mitochondrial dysfunction to salvage cellular senescence for managing neurodegeneration. Adv Protein Chem Struct Biol 136:309–337. https://doi.org/10.1016/bs.apcsb.2023.02.016
Song P, Duan JL, Ding J, Liu JJ, Fang ZQ, Xu H, Li ZW, Du W, Xu M, Ling YW, He F, Tao KS, Wang L (2023) Cellular senescence primes liver fibrosis regression through Notch-EZH2. MedComm https://doi.org/10.1002/mco2.346
Suda M, Shimizu I, Katsuumi G, Yoshida Y, Hayashi Y, Ikegami R, Matsumoto N, Yoshida Y, Mikawa R, Katayama A, Wada J, Seki M, Suzuki Y, Iwama A, Nakagami H, Nagasawa A, Morishita R, Sugimoto M, Okuda S, Tsuchida M, Ozaki K, Nakanishi-Matsui M, Minamino T (2021) Senolytic vaccination improves normal and pathological age-related phenotypes and increases lifespan in progeroid mice. Nat Aging 1:1117–1126. https://doi.org/10.1038/s43587-021-00151-2
Sun Y, Campisi J, Higano C, Beer TM, Porter P, Coleman I, True L, Nelson PS (2012) Treatment-induced damage to the tumor microenvironment promotes prostate cancer therapy resistance through WNT16B. Nat Med 18:1359–1368. https://doi.org/10.1038/nm.2890
Takaya K, Ishii T, Asou T, Kishi K (2023) Navitoclax (ABT-263) rejuvenates human skin by eliminating senescent dermal fibroblasts in a mouse/human chimeric model. Rejuvenation Res 26:9–20. https://doi.org/10.1089/rej.2022.0048
Toutfaire M, Bauwens E, Debacq-Chainiaux F (2017) The impact of cellular senescence in skin ageing: a notion of mosaic and therapeutic strategies. Biochem Pharmacol 142:1–12. https://doi.org/10.1016/j.bcp.2017.04.011
Treiber N, Maity P, Singh K, Kohn M, Keist AF, Ferchiu F, Sante L, Frese S, Bloch W, Kreppel F, Kochanek S, Sindrilaru A, Iben S, Högel J, Ohnmacht M, Claes LE, Ignatius A, Chung JH, Lee MJ, Kamenisch Y, Berneburg M, Nikolaus T, Braunstein K, Sperfeld AD, Ludolph AC, Briviba K, Wlaschek M, Scharffetter-Kochanek K (2011) Accelerated aging phenotype in mice with conditional deficiency for mitochondrial superoxide dismutase in the connective tissue. Aging Cell 10:239–254. https://doi.org/10.1111/j.1474-9726.2010.00658.x
Vollmayer P, Clair T, Goding JW, Sano K, Servos J, Zimmermann H (2003) Hydrolysis of diadenosine polyphosphates by nucleotide pyrophosphatases/phosphodiesterases. Eur J Biochem 270:2971–2978. https://doi.org/10.1046/j.1432-1033.2003.03674.x
Waaijer ME, Gunn DA, Adams PD, Pawlikowski JS, Griffiths CE, van Heemst D, Slagboom PE, Westendorp RGJ, Maier AB (2016) P16INK4a positive cells in human skin are indicative of local elastic fiber morphology, facial wrinkling, and perceived age. J Gerontol A Biol Sci Med Sci 71:1022–1028. https://doi.org/10.1093/gerona/glv114
Walker M (2022) Human skin through the ages. Int J Pharm 622:121850. https://doi.org/10.1016/j.ijpharm.2022.121850
Wlaschek M, Maity P, Makrantonaki E, Scharffetter-Kochanek K (2021) Connective tissue and fibroblast senescence in skin aging. J Invest Dermatol 141:985–992. https://doi.org/10.1016/j.jid.2020.11.010
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V, Cordon-Cardo C, Lowe SW (2007) Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445:656–660. https://doi.org/10.1038/nature05529
Yosef R, Pilpel N, Tokarsky-Amiel R, Biran A, Ovadya Y, Cohen S, Vadai E, Dassa L, Shahar E, Condiotti R, Ben-Porath I, Krizhanovsky V (2016) Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL. Nat Commun 7:11190. https://doi.org/10.1038/ncomms11190
Zimmermann H (2001) Ecto-nucleotidases. In: Abbracchio MP, Williams M (eds) Handbook of experimental pharmacology. Purinergic and pyrimidergic signalling. Springer-Verlag, Heidelberg, pp 209–250
Acknowledgements
The authors thank R. Toda for the technical assistance with the experiments and useful discussions. This study was supported by the JSPS KAKENHI (grant number JP 22K19589).
Author information
Authors and Affiliations
Contributions
Conceptualization: KT, KK; Investigation: KT; Animal work: KT; Data Analysis and interpretation: KT; Writing—original draft: KT; Supervision: KK. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest related to this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takaya, K., Kishi, K. Regulation of ENPP5, a senescence-associated secretory phenotype factor, prevents skin aging. Biogerontology (2024). https://doi.org/10.1007/s10522-024-10096-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10522-024-10096-9