Abstract
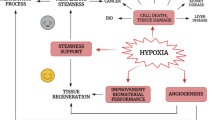
Brain hypoxia is involved in many diseases. The activation of angiogenesis is one of the major adaptive mechanisms to counteract the adverse effects of hypoxia. In a previous work, we have shown that the adult rat striatum promotes angiogenesis in response to hypoxia via upregulation of the most important proangiogenic factor, the vascular endothelial growth factor (VEGF). However, the effects of hypoxia on angiogenesis in the aged striatum remain unknown and constitute our aim. Here we show the upregulation of hypoxia-inducible factor-1α in the striatum of aged (24–25 months old) Wistar rats exposed to acute hypoxia and analysed during a reoxygenation period ranging from 0 h to 5 days. While the mRNA expression of the proangiogenic factors VEGF, transforming growth factor-β1 (TGF-β1), and adrenomedullin dropped at 0 h post-hypoxia compared to normoxic control, no changes were detected at the protein level, showing an impaired response of these proangiogenic factors to hypoxia in the aged striatum. However, the striatal blood vessel network increased at 24 h of reoxygenation, suggesting that mechanisms independent from these proangiogenic factors may be involved in hypoxia-induced angiogenesis in the striatum of aged rats. A thorough understanding of the factors involved in the response to hypoxia is essential to guide the design of therapies for hypoxia-related diseases in the aged brain.




Similar content being viewed by others
References
Ahluwalia A, Tarnawski AS (2012) Critical role of hypoxia sensor-HIF-1α in VEGF gene activation. Implications for angiogenesis and tissue injury healing. Curr Med Chem 19(1):90–97
Becker JB, Prendergast BJ, Liang JW (2016) Female rats are not more variable than male rats: a meta-analysis of neuroscience studies. Biol Sex Differ 26(7):34. doi:10.1186/s13293-016-0087-5
Benderro GF, Lamanna JC (2011) Hypoxia-induced angiogenesis is delayed in aging mouse brain. Brain Res 1389:50–60. doi:10.1016/j.brainres.2011.03.016
Brown WR, Thore CR (2011) Review: cerebral microvascular pathology in ageing and neurodegeneration. NeuropatholApplNeurobiol 37(1):56–74. doi:10.1111/j.1365-2990.2010.01139.x
Buga AM, Scholz CJ, Kumar S, Herndon JG, Alexandru D, Cojocaru GR, Dandekar T, Popa-Wagner A (2012) Identification of new therapeutic targets by genome-wide analysis of gene expression in the ipsilateral cortex of aged rats after stroke. PLoS ONE 7(12):e50985. doi:10.1371/journal.pone.0050985
Buga AM, Margaritescu C, Scholz CJ, Radu E, Zelenak C, Popa-Wagner A (2014a) Transcriptomics of post-stroke angiogenesis in the aged brain. Front Aging Neurosci 18(6):44. doi:10.3389/fnagi.2014.00044
Buga AM, Margaritescu C, Scholz CJ, Radu E, Zelenak C, Popa-Wagner A (2014b) Transcriptomics of post-stroke angiogenesis in the aged brain. Front Aging Neurosci 6:44. doi:10.3389/fnagi.2014.00044
Correia SC, Carvalho C, Cardoso S, Santos RX, Plácido AI, Candeias E, Duarte AI, Moreira PI (2013) Defective HIF signaling pathway and brain response to hypoxia in neurodegenerative diseases: not an “iffy” question! Curr Pharm Des 19(38):6809–6822
Di Ieva A, Grizzi F, Ceva-Grimaldi G, Russo C, Gaetani P, Aimar E, Levi D, Pisano P, Tancioni F, Nicola G, Tschabitscher M, Dioguardi N, Baena RR (2007) Fractal dimension as a quantitator of the microvasculature of normal and adenomatous pituitary tissue. J Anat 211(5):673–680
Donovan D, Harmey JH, Toomey D, Osborne DH, Redmond HP, Bouchier-Hayes DJ (1997) TGF beta-1 regulation of VEGF production by breast cancer cells. Ann SurgOncol 4(8):621–627
Efimenko A, Starostina E, Kalinina N, Stolzing A (2011) Angiogenic properties of aged adipose derived mesenchymal stem cells after hypoxic conditioning. J Transl Med 9:10. doi:10.1186/1479-5876-9-10
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9(6):669–676
Fong GH (2008) Mechanisms of adaptive angiogenesis to tissue hypoxia. Angiogenesis 11(2):121–140. doi:10.1007/s10456-008-9107-3
Gardoni F, Bellone C (2015) Modulation of the glutamatergic transmission by Dopamine: a focus on Parkinson, Huntington and Addiction diseases. Front Cell Neurosci 9:25. doi:10.3389/fncel.2015.00025
Gozal D, Row BW, Kheirandish L, Liu R, Guo SZ, Qiang F, Brittian KR (2003) Increased susceptibility to intermittent hypoxia in aging rats: changes in proteasomal activity, neuronal apoptosis and spatial function. J Neurochem 86(6):1545–1552
Grizzi F, Russo C, Colombo P, Franceschini B, Frezza EE, Cobos E, Chiriva-Internati M (2005) Quantitative evaluation and modeling of two-dimensional neovascular network complexity: the surface fractal dimension. BMC Cancer 8(5):14
Hung SP, Yang MH, Tseng KF, Lee OK (2013) Hypoxia-induced secretion of TGF-β1 in mesenchymal stem cell promotes breast cancer cell progression. Cell Transplant 22(10):1869–1882. doi:10.3727/096368912X657954
Hwang IS, Fung ML, Liong EC, Tipoe GL, Tang F (2007) Age-related changes in adrenomedullin expression and hypoxia-inducible factor-1 activity in the rat lung and their responses to hypoxia. J Gerontol A BiolSci Med Sci 62(1):41–49
Ignacak ML, Harbaugh SV, Dayyat E, Row BW, Gozal D, Czyzyk-Krzeska MF (2009) Intermittent hypoxia regulates RNA polymerase II in hippocampus and prefrontal cortex. Neuroscience 158(4):1436–1445. doi:10.1016/j.neuroscience.2008.11.025
Iimuro S, Shindo T, Moriyama N, Amaki T, Niu P, Takeda N, Iwata H, Zhang Y, Ebihara A, Nagai R (2004) Angiogenic effects of adrenomedullin in ischemia and tumor growth. Circ Res 95(4):415–423
Itoh Y, Arnold AP (2015) Are females more variable than males in gene expression? Meta-analysis of microarray datasets. Biol Sex Differ 6:18
Johnson AB, Denko N, Barton MC (2008) Hypoxia induces a novel signature of chromatin modifications and global repression of transcription. Mutat Res 640(1–2):174–179. doi:10.1016/j.mrfmmm.2008.01.001
Lam YT, Lecce L, Clayton Z, Simpson P, Karas R, Ng M (2016) Aging impairs ischemia-induced neovascularization by attenuating the mobilization of bone marrow-derived angiogenic cells. IJC MetabEndocr 12:19–29
Lawlor DA, Ebrahim S, Davey-Smith G (2002) Role of endogenous oestrogen in aetiology of coronary heart disease. BMJ 325:311–312
Li H, Witte K, August M, Brausch I, Godtel-Armbrust U, Habermeier A, Closs EI, Oelze M, Munzel T, Forstermann U (2006) Reversal of endothelial nitric oxide synthase uncoupling and up-regulation of endothelial nitric oxide synthase expression lowers blood pressure in hypertensive rats. J Am CollCardiol 47:2536–2544
Logan A, Berry M, Gonzalez AM, Frautschy SA, Sporn MB, Baird A (1994) Effects of transforming growth factor beta 1 on scar production in the injured central nervous system of the rat. Eur J Neurosci 6(3):355–363
Mancardi D, Varetto G, Bucci E, Maniero F, Guiot C (2008) Fractal parameters and vascular networks: facts & artifacts. Theor Biol Med Model 17(5):12. doi:10.1186/1742-4682-5-12
McMahon S, Charbonneau M, Grandmont S, Richard DE, Dubois CM (2006) Transforming growth factor beta1 induces hypoxia-inducible factor-1 stabilization through selective inhibition of PHD2 expression. J BiolChem 281(34):24171–24181
Molina F, Rus A, Peinado MA, Del Moral ML (2013) Short-term hypoxia/reoxygenation activates the angiogenic pathway in rat caudate putamen. J Biosci 38(2):363–371
Molina F, Peinado MA, del Moral ML, Rus A (2016) Response of Nitric Oxide system to hypobaric hypoxia in the aged striatum. Gerontology. doi:10.1159/000450607
Ndubuizu OI, Chavez JC, LaManna JC (2009) Increased prolyl 4-hydroxylase expression and differential regulation of hypoxia-inducible factors in the aged rat brain. Am J PhysiolRegulIntegr Comp Physiol 297(1):R158–R165. doi:10.1152/ajpregu.90829.2008
Ndubuizu OI, Tsipis CP, Li A, LaManna JC (2010) Hypoxia-inducible factor-1 (HIF-1)-independent microvascular angiogenesis in the aged rat brain. BrainRes 1366:101–109. doi:10.1016/j.brainres.2010.09.064
Patt S, Sampaolo S, Théallier-Jankó A, Tschairkin I, Cervós-Navarro J (1997) Cerebral angiogenesis triggered by severe chronic hypoxia displays regional differences. J Cereb Blood Flow Metab 17(7):801–806
Pisani A, Calabresi P, Bernardi G (1997) Hypoxia in striatal and cortical neurones: membrane potential and Ca2 + measurements. NeuroReport 8(5):1143–1147
Prabhakar NR (2001) Oxygen sensing during intermittent hypoxia: cellular and molecular mechanisms. J ApplPhysiol 90:1986–1994
Prendergast BJ, Onishi KG, Zucker I (2014) Female mice liberated for inclusion in neuroscience and biomedical research. Neurosci Biobehav Rev 40:1–5
Rich-Edwards JW, Manson JE, Hennekens CH, Buring JE (1995) The primary prevention of coronary heart disease in women. N Engl J Med 332:1758–1766
Rivard A, Berthou-Soulie L, Principe N, Kearney M, Curry C, Branellec D, Semenza GL, Isner JM (2000) Age-dependent defect in vascular endothelial growth factor expression is associated with reduced hypoxia-inducible factor 1 activity. J BiolChem 275(38):29643–29647
Sandu RE, Uzoni A, Ciobanu O, Moldovan M, Anghel A, Radu E, Coogan AN, Popa-Wagner A (2016) Post-stroke gaseous hypothermia increases vascular density but not neurogenesis in the ischemic penumbra of aged rats. Restor Neurol Neurosci 34(3):401–414. doi:10.3233/RNN-150600
Sullivan JM, Fowlkes LP (1996) The clinical aspects of estrogen and the cardiovascular system. Obstet Gynecol 87:36S–43S
Wang LN, Xu D, Gui QP, Zhu MW, Zhang HH, Hu YZ (2004) Morphological and quantatitive capillary changes in aging human brain. Zhongguo Yi XueKeXue Yuan XueBao 26(2):104–107
Wang Y, Zhang JS, Qian J, Huang GC, Chen Q (2006) Adrenomedullin regulates expressions of transforming growth factor-beta1 and beta1-induced matrix metalloproteinase-2 in hepatic stellate cells. Int J ExpPathol 87(3):177–184
Wang L, Bhatta A, Toque HA, Rojas M, Yao L, Xu Z, Patel C, Caldwell RB, Caldwell RW (2015) Arginase inhibition enhances angiogenesis in endothelial cells exposed to hypoxia. Microvasc Res 98:1–8. doi:10.1016/j.mvr.2014.11.002
Acknowledgements
We wish to thank to Dr. Rafael Lomas for his statistic assistance, and to Dra. M. Rosario Sepúlveda for critical reading of the manuscript.
Funding
This study was funded by Instituto de Salud Carlos III (PI081222) and University of Jaén (RFC/PP2008/UJA_08_16_20).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in animals were in accordance with the ethical standards of the University of Jaén (Spain) at which the study was conducted.
Rights and permissions
About this article
Cite this article
Molina, F., del Moral, M.L., Peinado, M.Á. et al. Angiogenesis is VEGF-independent in the aged striatum of male rats exposed to acute hypoxia. Biogerontology 18, 759–768 (2017). https://doi.org/10.1007/s10522-017-9709-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-017-9709-5