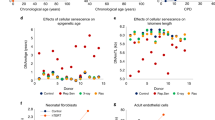
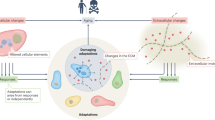
Abstract
We are accustomed to treating aging as a set of things that go wrong with the body. But for more than twenty years, there has been accumulating evidence that much of the process takes place under genetic control. We have seen that signaling chemistry can make dramatic differences in life span, and that single molecules can significantly affect longevity. We are frequently confronted with puzzling choices the body makes which benefit neither present health nor fertility nor long-term survival. If we permit ourselves a shift of reference frame and regard aging as a programmed biological function like growth and development, then these observations fall into place and make sense. This perspective suggests that aging proceeds under control of a master clock, or several redundant clocks. If this is so, we may learn to reset the clocks with biochemical interventions and make an old body behave like a young body, including repair of many of the modes of damage that we are accustomed to regard as independent symptoms of the senescent phenotype, and for which we have assumed that the body has no remedy.





Similar content being viewed by others
References
Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, Kirkland JL, van Deursen JM (2011) Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479(7372):232–236
Behl C (2000) Apoptosis and alzheimer’s disease. J Neural Trans 107(11):1325–1344
Bischoff C, Graakjaer J, Petersen HC, Jeune B, Bohr VA, Koelvraa S, Christensen K (2005) Telomere length among the elderly and oldest-old. Twin Res Hum Genet 8(5):425–432
Blagosklonny M (2010) Revisiting the antagonistic pleiotropy theory of aging. Cell Cycle 9(16):3151–3156
Bonduriansky R, Brassil CE (2002) Senescence: rapid and costly ageing in wild male flies. Nature 420(6914):377
Bouchard J, Villeda SA (2014) Aging and brain rejuvenation as systemic events. J Neurochem. doi:10.1111/jnc.12969
Bowles JT (1998) The evolution of aging: a new approach to an old problem of biology. Med Hypotheses 51(3):179–221
Bredesen DE (2004) The non-existent aging program: how does it work? Aging Cell 3(5):255–259
Bronikowski AM, Promislow DE (2005) Testing evolutionary theories of aging in wild populations. Trends Ecol Evol 20(6):271–273
Brouilette SW, Moore JS, McMahon AD, Thompson JR, Ford I, Shepherd J, Packard CJ, Samani NJ (2007) Telomere length, risk of coronary heart disease, and statin treatment in the West of Scotland Primary Prevention Study: a nested case-control study. Lancet 369(9556):107–114
Brümmendorf TH, Mak J, Sabo KM, Baerlocher GM, Dietz K, Abkowitz JL, Lansdorp PM (2002) Longitudinal studies of telomere length in feline blood cells: implications for hematopoietic stem cell turnover in vivo. Exp Hematol 30(10):1147–1152
Bryant MJ, Reznick D (2004) Comparative studies of senescence in natural populations of guppies. Am Nat 163(1):55–68
Calabrese EJ (2005) Toxicological awakenings: the rebirth of hormesis as a central pillar of toxicology. Toxicol Appl Pharmacol 204(1):1–8
Calabrese EJ, Baldwin LA (1998) Hormesis as a biological hypothesis. Environ Health Perspect 106(Supp 1):357–362
Campisi J (2013) Aging, cellular senescence, and cancer. Annu Rev Physiol 75:685
Cawthon RM, Smith KR, O’Brien E, Sivatchenko A, Kerber RA (2003) Association between telomere length in blood and mortality in people aged 60 years or older. Lancet 361(9355):393–395
Clark WR (1999) A means to an end: the biological basis of aging and death. Oxford University Press, New York, Oxford
Clark WR (2004) Reflections on an unsolved problem of biology: the evolution of senescence and death. Adv Gerontol 14:7–20
Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA (2005) Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 433(7027):760–764
Curtsinger JW, Fukui HH, Khazaeli AA, Kirscher A, Pletcher SD, Promislow DE, Tatar M (1995) Genetic variation and aging. Annu Rev Genet 29:553–575
de Magalhaes JP (2013) How ageing processes influence cancer. Nat Rev Cancer 13(5):357–365
de Magalhães JP (2012) Programmatic features of aging originating in development: aging mechanisms beyond molecular damage? FASEB J 26(12):4821–4826
de Magalhães JP, Church GM (2005) Genomes optimize reproduction: aging as a consequence of the developmental program. Physiology 20(4):252–259
de Magalhães JP, Curado J, Church GM (2009) Meta-analysis of age-related gene expression profiles identifies common signatures of aging. Bioinformatics 25(7):875–881
Ebling FJ (2005) The neuroendocrine timing of puberty. Reproduction 129(6):675–683
Elabd C, Cousin W, Upadhyayula P, Chen RY, Chooljian MS, Li J, Kung S, Jiang KP, Conboy IM (2014) Oxytocin is an age-specific circulating hormone that is necessary for muscle maintenance and regeneration. Nat Commun 5:4082
Fabrizio P, Battistella L, Vardavas R, Gattazzo C, Liou LL, Diaspro A, Dossen JW, Gralla EB, Longo VD (2004) Superoxide is a mediator of an altruistic aging program in Saccharomyces cerevisiae. J Cell Biol 166(7):1055–1067
Fisher RA (1930) The genetical theory of natural selection. The Clarendon Press, Oxford
Fitzpatrick AL, Kronmal RA, Gardner JP, Psaty BM, Jenny NS, Tracy RP, Walston J, Kimura M, Aviv A (2007) Leukocyte telomere length and cardiovascular disease in the cardiovascular health study. Am J Epidemiol 165(1):14–21
Flatt T (2009) Ageing: diet and longevity in the balance. Nature 462(7276):989–990
Fyhrquist F, Silventoinen K, Saijonmaa O, Kontula K, Devereux RB, de Faire U, Os I, Dahlof B (2011) Telomere length and cardiovascular risk in hypertensive patients with left ventricular hypertrophy: the LIFE study. J Hum Hypertens 25(12):711–718
Gavrilova NS, Gavrilov LA, Semyonova VG, Evdokushkina GN (2004) Does exceptional human longevity come with a high cost of infertility? Testing the evolutionary theories of aging. Ann N Y Acad Sci 1019:513–517
Goldsmith TC (2013) The evolution of aging. Annapolis, Azinet
Guarente L, Kenyon C (2000) Genetic pathways that regulate ageing in model organisms. Nature 408(6809):255–262
Hanson RW, Hakimi P (2008) Born to run; the story of the PEPCK-Cmus mouse. Biochimie 90(6):838–842
Hardin G (1968) The tragedy of the commons. Science 162:1243–1248
Harris SE, Deary IJ, Maclntyre A, Lamb KJ, Radhakrishnan K, Starr JM, Whalley LJ, Shiels PG (2006) The association between telomere length, physical health, cognitive ageing, and mortality in non-demented older people. Neurosci Lett 406(3):260–264
Horvath S (2013) DNA methylation age of human tissues and cell types. Genome Biol 14(10):R115
Johnson AA, Akman K, Calimport SR, Wuttke D, Stolzing A, de Magalhães JP (2012) The role of DNA methylation in aging, rejuvenation, and age-related disease. Rejuvenation Res 15(5):483–494
Jones MJ, Goodman SJ, Kobor MS (2015) DNA methylation and healthy human aging. Aging cell. (in press)
Jones OR, Scheuerlein A, Salguero-Gomez R, Camarda CG, Schaible R, Casper BB, Dahlgren JP, Ehrlen J, Garcia MB, Menges ES, Quintana-Ascencio PF, Caswell H, Baudisch A, Vaupel JW (2014) Diversity of ageing across the tree of life. Nature 505(7482):169–173
Katsimpardi L, Litterman NK, Schein PA, Miller CM, Loffredo FS, Wojtkiewicz GR, Chen JW, Lee RT, Wagers AJ, Rubin LL (2014) Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science 344(6184):630–634
Kenyon C (2001) A conserved regulatory system for aging. Cell 105(2):165–168
Kimura M, Hjelmborg, JVB, Hjelmborg JVB, Gardner JP, Bathum L, Brimacombe M, Lu X, Christiansen L, Vaupel JW, Aviv A, Christensen K (2008) Telomere length and mortality: a study of leukocytes in elderly Danish twins. Am J Epidemiol 167(7):799–806
Kirkwood T (1977) Evolution of aging. Nature 270:301–304
Kirkwood TB (2005) Understanding the odd science of aging. Cell 120(4):437–447
Klein DR (1968) The introduction, increase, and crash of reindeer on St Matthew Island. J Wildl Manag 32(2):350–367
Ma H, Zhou Z, Wei S, Liu Z, Pooley KA, Dunning AM, Svenson U, Roos G, Hosgood HD III, Shen M, Wei Q (2011) Shortened telomere length is associated with increased risk of cancer: a meta-analysis. PLoS ONE 6(6):e20466
Martins AC (2011) Change and aging senescence as an adaptation. PLoS ONE 6(9):e24328
Marzetti E, Leeuwenburgh C (2006) Skeletal muscle apoptosis, sarcopenia and frailty at old age. Exp Gerontol 41(12):1234–1238
Masoro EJ (2003) Subfield history: caloric restriction, slowing aging, and extending life. Science’s SAGE KE 2003(8):2
Masoro EJ (2005) Overview of caloric restriction and ageing. Mech Ageing Dev 126(9):913–922
Masoro EJ (2007) The role of hormesis in life extension by dietary restriction. Interdiscip Top Gerontol 35:1–17
Mayack SR, Shadrach JL, Kim FS, Wagers AJ (2010) Systemic signals regulate ageing and rejuvenation of blood stem cell niches. Nature 463(7280):495–500
McCay CM, Pope F, Lunsford W, Sperling G, Sambhavaphol P (1957) Parabiosis between old and young rats. Gerontology 1(1):7–17
McKevitt TP, Nasir L, Devlin P, Argyle DJ (2002) Telomere lengths in dogs decrease with increasing donor age. J Nutr 132(6):1604S–1606S
Medawar PB (1952) An unsolved problem of biology. Published for the college by H. K. Lewis, London
Merry BJ, Holehan AM (1981) Serum profiles of LH, FSH, testosterone and 5 alpha-DHT from 21 to 1000 days of age in ad libitum fed and dietary restricted rats. Exp Gerontol 16(6):431–444
Mitteldorf J (2001) Can experiments on caloric restriction be reconciled with the disposable soma theory for the evolution of senescence? Evol Int J Org Evol 55(9):1902–1905 discussion 1906
Mitteldorf J (2004) Aging selected for its own sake. Evol Ecol Res 6:1–17
Mitteldorf J (2006) Chaotic population dynamics and the evolution of aging: proposing a demographic theory of senescence. Evol Ecol Res 8:561–574
Mitteldorf J (2010a) Evolutionary origins of aging. Approaches to the control of aging: building a pathway to human life extension. In: Fahy GM, West MD, Coles LS, Harris SB (eds). New York, Springer, pp 87–126
Mitteldorf J (2010b) Female fertility and longevity. Age (Dordr) 32:79–84
Mitteldorf J (2013) Telomere biology: cancer firewall or aging clock? Biochem (Moscow) 78(9):1054–1060
Mitteldorf J (2016). Aging is a group-selected adaptation: theory, evidence, and medical implications, CRC Press
Mitteldorf J, Goodnight C (2012) Post-reproductive life span and demographic stability. Oikos 121(9):1370–1378
Mitteldorf J, Martins AC (2014) programmed life span in the context of evolvability. Am Nat 184(3):289–302
Mitteldorf J, Pepper J (2009) Senescence as an adaptation to limit the spread of disease. J Theor Biol 260(2):186–195
Nussey DH, Froy H, Lemaitre JF, Gaillard JM, Austad SN (2012) Senescence in natural populations of animals: widespread evidence and its implications for bio-gerontology. Ageing Res Rev 12(1):214–225
Pauliny A, Wagner RH, Augustin J, Szep T, Blomqvist D (2006) Age-independent telomere length predicts fitness in two bird species. Mol Ecol 15(6):1681–1687
Pepper J, Shelton D, Rashidi A, Durand P (2013) Are internal, death-promoting mechanisms ever adaptive. J Phylogen Evol Biol 1(113):2
Rando TA, Chang HY (2012) Aging, rejuvenation, and epigenetic reprogramming: resetting the aging clock. Cell 148(1):46–57
Ricklefs R (1998) Evolutionary theories of aging: confirmation of a fundamental prediction, with implications for the genetic basis and evolution of life span. Am Nat 152:24–44
Ricklefs RE, Cadena CD (2007) Lifespan is unrelated to investment in reproduction in populations of mammals and birds in captivity. Ecol Lett 10(10):867–872
Rode L, Nordestgaard BG, Bojesen SE (2015) Peripheral blood leukocyte telomere length and mortality among 64 637 individuals from the general population. J Natl Cancer Inst 107(6):djv074
Sager R (1991) Senescence as a mode of tumor suppression. Environ Health Perspect 93:59–62
Sharpe FRL, Lotka AJ (1911) A problem in age-distribution. Philos Mag 21:435–438
Spitze K (1991) Chaoborus predation and life history evolution in Daphnia pulex: temporal pattern of population diversity, fitness, and mean life history. Evolution 45(1):82–92
Stearns SC (2000) Life history evolution: successes, limitations, and prospects. Naturwissenschaften 87(11):476–486
Strandberg TE, Saijonmaa O, Tilvis RS, Pitkala KH, Strandberg AY, Miettinen TA, Fyhrquist F (2011) Association of telomere length in older men with mortality and midlife body mass index and smoking. J Gerontol A Biol Sci Med Sci 66(7):815–820
Weindruch R, Walford RL (1988) Retardation of aging and disease by dietary restriction. CC Thomas, Springfield
Willeit P, Willeit J, Kloss-Brandstatter A, Kronenberg F, Kiechl S (2011) Fifteen-year follow-up of association between telomere length and incident cancer and cancer mortality. JAMA 306(1):42–44
Williams G (1957) Pleiotropy, natural selection, and the evolution of senescence. Evolution 11:398–411
Yoon CK (2002) Looking back at the days of the locust. New York Times, New York
Zykovich A, Hubbard A, Flynn JM, Tarnopolsky M, Fraga MF, Kerksick C, Ogborn D, MacNeil L, Mooney SD, Melov S (2014) Genome-wide DNA methylation changes with age in disease-free human skeletal muscle. Aging Cell 13(2):360–366
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mitteldorf, J. An epigenetic clock controls aging. Biogerontology 17, 257–265 (2016). https://doi.org/10.1007/s10522-015-9617-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-015-9617-5