Abstract
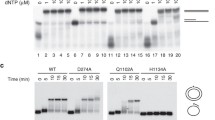
The premature human ageing Werner’s syndrome is caused by loss or mutation of the WRN helicase/exonuclease. We have recently identified the orthologue of the WRN exonuclease in flies, DmWRNexo, encoded by the CG7670 locus, and showed very high levels of mitotic recombination in a hypomorphic PiggyBac insertional mutant. Here, we report a novel allele of CG7670, with a point mutation resulting in the change of the conserved aspartate (229) to valine. Flies bearing this mutation show levels of mitotic recombination 20-fold higher than wild type. Molecular modelling suggests that D229 lies towards the outside of the molecule distant from the nuclease active site. We have produced recombinant protein of the D229V mutant, assayed its nuclease activity in vitro, and compared activity with that of wild type DmWRNexo and a D162A E164A double active site mutant we have created. We show for the first time that DmWRNexo has 3′–5′ exonuclease activity and that mutation within the presumptive active site disrupts exonuclease activity. Furthermore, we show that the D229V mutant has very limited exonuclease activity in vitro. Using Drosophila, we can therefore analyse WRN exonuclease from enzyme activity in vitro through to fly phenotype, and show that loss of exonuclease activity contributes to genome instability.




Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- DTT:
-
Dithiothreitol
- ECL:
-
Enhanced chemiluminescence
- HRP:
-
Horse radish peroxidase
- IPTG:
-
Isopropyl β-d-1-thiogalactopyranoside
- PBS:
-
Phosphate buffered saline
- TILLING:
-
Targeting Induced Local Lesions IN Genomes
- WS:
-
Werner syndrome
- WRN:
-
Protein mutated in Werner syndrome
References
Cheng WH, Muftuoglu M, Bohr VA (2007) Werner syndrome protein: functions in the response to DNA damage and replication stress in S-phase. Exp Gerontol 42:871–878. doi:10.1016/j.exger.2007.04.011
Courcelle J, Hanawalt PC (1999) RecQ and RecJ process blocked replication forks prior to the resumption of replication in UV-irradiated Escherichia coli. Mol Gen Genet 262:543–551. doi:10.1007/s004380051116
Cox LS, Faragher RG (2007) From old organisms to new molecules: integrative biology and therapeutic targets in accelerated human ageing. Cell Mol Life Sci 64:2620–2641. doi:10.1007/s00018-007-7123-x
Cox LS, Clancy DJ, Boubriak I, Saunders RD (2007) Modeling Werner Syndrome in Drosophila melanogaster: hyper-recombination in flies lacking WRN-like exonuclease. Ann N Y Acad Sci 1119:274–288. doi:10.1196/annals.1404.009
Davis T, Baird DM, Haughton MF, Jones CJ, Kipling D (2005) Prevention of accelerated cell aging in Werner syndrome using a p38 mitogen-activated protein kinase inhibitor. J Gerontol A Biol Sci Med Sci 60:1386–1393
Dhillon KK, Sidorova J, Saintigny Y, Poot M, Gollahon K, Rabinovitch PS, Monnat RJ Jr (2007) Functional role of the Werner syndrome RecQ helicase in human fibroblasts. Aging Cell 6:53–61. doi:10.1111/j.1474-9726.2006.00260.x
Faragher RG, Kill IR, Hunter JA, Pope FM, Tannock C, Shall S (1993) The gene responsible for Werner syndrome may be a cell division “counting” gene. Proc Natl Acad Sci USA 90:12030–12034. doi:10.1073/pnas.90.24.12030
Hartung F, Puchta H (2006) The RecQ gene family in plants. J Plant Physiol 163(3):287–296. doi:10.1016/j.jplph.2005.10.013
Hayflick L (1965) The limited in vitro lifetime of human diploid cell strains. Exp Cell Res 37:614–636. doi:10.1016/0014-4827(65)90211-9
Huang S, Li B, Gray MD, Oshima J, Mian IS, Campisi J (1998) The premature ageing syndrome protein, WRN, is a 3′→5′ exonuclease. Nat Genet 20:114–116. doi:10.1038/2410
Lawrence P, Johnston P, Morata G (1986) Methods of marking cells. In: Roberts DB (ed) Drosophila: a practical approach. IRL, Oxford, pp 229–242
Lebel M, Spillare EA, Harris CC, Leder P (1999) The Werner syndrome gene product co-purifies with the DNA replication complex and interacts with PCNA and topoisomerase I. J Biol Chem 274(53):37795–37799. doi:10.1074/jbc.274.53.37795
McCallum CM, Comai L, Greene EA, Henikoff S (2000) Targeting induced local lesions IN genomes (TILLING) for plant functional genomics. Plant Physiol 123:439–442. doi:10.1104/pp.123.2.439
McVey M, Andersen SL, Broze Y, Sekelsky J (2007) Multiple functions of Drosophila BLM helicase in maintenance of genome stability. Genetics 176:1979–1992. doi:10.1534/genetics.106.070052
Opresko PL, Laine JP, Brosh RM Jr, Seidman MM, Bohr VA (2001) Coordinate action of the helicase and 3′ to 5′ exonuclease of Werner syndrome protein. J Biol Chem 276:44677–44687. doi:10.1074/jbc.M107548200
Peitsch MC, Herzyk P, Wells TN, Hubbard RE (1996) Automated modelling of the transmembrane region of G-protein coupled receptor by Swiss-model. Receptors Channels 4:161–164
Perry JJ, Yannone SM, Holden LG, Hitomi C, Asaithamby A, Han S, Cooper PK, Chen DJ, Tainer JA (2006) WRN exonuclease structure and molecular mechanism imply an editing role in DNA end processing. Nat Struct Mol Biol 13:414–422. doi:10.1038/nsmb1088
Rodriguez-Lopez AM, Jackson DA, Iborra F, Cox LS (2002) Asymmetry of DNA replication fork progression in Werner’s syndrome. Aging Cell 1:30–39. doi:10.1046/j.1474-9728.2002.00002.x
Rodriguez-Lopez AM, Whitby MC, Borer CM, Bachler MA, Cox LS (2007) Correction of proliferation and drug sensitivity defects in the progeroid Werner’s syndrome by Holliday junction resolution. Rejuvenation Res 10:27–40. doi:10.1089/rej.2006.0503
Salk D, Au K, Hoehn H, Stenchever MR, Martin GM (1981) Evidence of clonal attenuation, clonal succession, and clonal expansion in mass cultures of aging Werner’s syndrome skin fibroblasts. Cytogenet Cell Genet 30:108–117. doi:10.1159/000131597
Saunders RD, Boubriak I, Clancy DJ, Cox LS (2008) Identification and characterization of a Drosophila ortholog of WRN exonuclease that is required to maintain genome integrity. Aging Cell 7:418–425. doi:10.1111/j.1474-9726.2008.00388.x
Schwede T, Kopp J, Guex N, Peitsch MC (2003) SWISS-MODEL: An automated protein homology-modeling server. Nucleic Acids Res 31:3381–3385. doi:10.1093/nar/gkg520
Sidorova JM, Li N, Folch A, Monnat RJ Jr (2008) The RecQ helicase WRN is required for normal replication fork progression after DNA damage or replication fork arrest. Cell Cycle 7:796–807
Till BJ, Colbert T, Tompa R, Enns LC, Codomo CA, Johnson JE, Reynolds SH, Henikoff JG, Greene EA, Steine MN, Comai L, Henikoff S (2003) High-throughput TILLING for functional genomics. Methods Mol Biol 236:205–220
Yu CE, Oshima J, Fu YH, Wijsman EM, Hisama F, Alisch R, Matthews S, Nakura J, Miki T, Ouais S, Martin GM, Mulligan J, Schellenberg GD (1996) Positional cloning of the Werner’s syndrome gene. Science 272:258–262. doi:10.1126/science.272.5259.258
Yu CE, Oshima J, Wijsman EM, Nakura J, Miki T, Piussan C, Matthews S, Fu YH, Mulligan J, Martin GM, Schellenberg GD (1997) Mutations in the consensus helicase domains of the Werner syndrome gene. Werner’s syndrome collaborative group. Am J Hum Genet 60:330–341
Acknowledgments
We thank Christine Borer for technical support, and Tony Willis for protein sequencing. This work was supported by the Biotechnology and Biological Sciences Research Council [grant numbers BB/E000924/1, BB/E002072/1 and BB/E016995/1].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boubriak, I., Mason, P.A., Clancy, D.J. et al. DmWRNexo is a 3′–5′ exonuclease: phenotypic and biochemical characterization of mutants of the Drosophila orthologue of human WRN exonuclease. Biogerontology 10, 267–277 (2009). https://doi.org/10.1007/s10522-008-9181-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-008-9181-3