Abstract
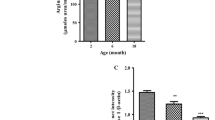
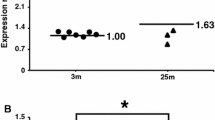
Dietary restriction (DR) influences several physiological processes, retards the incidences and severity of various age-related diseases and extends lifespan of various animal species. The effect of DR on the activities of malate–aspartate shuttle enzymes, viz. cytosolic and mitochondrial aspartate aminotransferase (c- and m-AsAT) and malate dehydrogenase (c- and m-MDH) was investigated in the liver and kidney of adult (5-months) and old (21-months) male mice. The results show that the activity (U/mg protein) of both c- and m-MDH and AsAT is decreased significantly in the liver and kidney of old mice compared to adult ones. However, DR in old mice reverses significantly the enzyme activities to a level closer to adult animals. Polyacrylamide gel electrophoresis (PAGE) and specific staining of c-AsAT, one of the selected isoenzymes of the shuttle, showed a similar pattern of activity expression as observed by activity measurements in both the tissues studied. Slot blot analysis of c-AsAT confirmed the lower protein content of this isoenzyme in old mice compared to adult ones and a higher level in old-dietary restricted mice. Thus, our results suggest that the late onset of DR in older mice reverses decline in malate–aspartate shuttle enzymes and that it may allow a better metabolic regulation in older animals.





Similar content being viewed by others
References
Bender K, Newsholme P, Brennan L, Maechter P (2006) The importance of redox shuttles to pancreatic β-cell energy metabolism and function. Biochem Soc Trans 34:811–814
Borst P (1963) In: Frunktionelle and morphologishen organization derzelle. Springer, Berlin, pp 137–158
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein using the principle of protein-dye binding. Anal Biochem 72:248–254
Cederbaum AI, Lieber CS, Beatic DS, Rubin E (1973) Characterization of shuttle mechanism for the transport of reducing equivalents into mitochondria. Arch Biochem Biophys 158:263–281
Colin S, Ricardo G, Sharon P, Suma K, Gustavo B, Christiaan L (2003) Short term caloric restriction and regulatory proteins of apoptosis in heart, skeletal muscle and kidney of Fischer 344 rats. Biogerontology 4:141–147
Davis BJ (1964) Native polyacrylamide gel electrophoresis. Ann N Y Acad Sci 121:404–427
Dhahbi JM, Mote PL, Wingo J, Tillman JB, Walford RL, Spindler SR (1999) Calories and aging alter gene expression for gluconeogenic, glycolytic, and nitrogen-metabolizing enzymes. Am J Physiol 277:E352–E360
Doonan S, Barra D, Bossa F, Porter PB, Wilkinson SM (1981) Interspecies comparisons of aspartate aminotransferases based on amino acid compositions. Comp Biochem Physiol 69:B747–B752
Dutta D, Sharma R (2004) Age-dependent dietary regulation of glucocorticoid receptors in the liver of mice. Biogerontology 5:177–184
Feuer RJ, Duffy PH, Leaky JA, Turturro A, Mittelstaedt PRA, Hart RW (1989) Effect of chronic caloric restriction on hepatic enzymes of intermediary metabolism in the male Fischer 344 rat. Mech Ageing Dev 48:179–189
Goto S, Takahashi R, Radak Z, Sharma R (2007) Beneficial biochemical outcomes of late onset dietary restriction in rodents. Ann N Y Acad Sci 1100:431–441
Goyary D, Sharma R (2005) Dietary restriction and triiodothyronine (T3) regulation of malate–aspartate shuttle enzymes in the liver and kidney of mice. Indian J Biochem Biophys 42:345–349
Hagopian K, Ramsey JJ, Weindruch R (2003) Caloric restriction increases gluconeogenic and transaminase enzyme activities in mouse liver. Exp Gerontol 38:267–278
Herzfeld A, Greengard O (1971) Aspartate aminotransferase in rate tissues: changes with growth and hormones. Biochim Biophys Acta 237:88–98
Hursting SD, Lavigne JA, Berrigan D, Perkins SN, Barret JC (2003) Calorie restriction, aging, and cancer prevention: mechanism of action and applicability to humans. Ann Rev Med 54:131–152
Kanungo MS (1980) In: Biochemistry of aging. Academic Press, London, pp 79–126
Karmen A (1955) A note on the spectrophotometric assay of glutamic oxalacetic transaminase in human blood serum. J Clin Invest 34:131–133
Kitto B (1969) In: Lowenstein JM (ed) Methods in enzymol V13. Acad. Press, New York, pp 106–107
Lardy HA, Poetkao V, Walker P (1965) Paths of carbon in gluconeogenesis and lipogenesis: the role of mitochondria in supplying precursors of phosphoenolpyruvate. Proc Natl Acad Sci 53:1410–1415
Lee J, Herman PJ, Mattson MP (2000) Dietary restriction selectively decreases GR expression in the hippocampus and cerebral cortex of rats. Exp Neurol 166:435–441
Lehninger AL (1951) Phosphorylation coupled to oxidation of dihydrodiphosphopyridine nucleotide. J Biol Chem 190:345–359
Lewis SEM, Goldspink DF, Philips JG, Merry BJ, Holehan AM (1985) The effects of aging and chronic dietary restriction on whole body growth and protein turnover in the rat. Exp Gerontol 20:253–263
Lin SJ, Defossez PA, Guarente L (2000) Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science 289:2126–2128
Lyngdoh HG, Sharma R (2001) Hydrocortisone and triiodothyronine regulation of malate-aspartate shuttle enzymes during postnatal development of chicken. Indian J Biochem Biophys 38:170–175
Lynn TF, Hart RW, Leaky JEA (1998) Caloric restriction as a mechanism mediating resistance to environmental disease. Environ Health Perspect 106:313–324
McKenna MC, Waagepetersen HS, Schousboe A, Sonnewald U (2006) Neuronal and astrocytic shuttle mechanisms for cytosolic-mitochondrial transfer of reducing equivalents: current evidence and pharmacological tools. Biochem Pharmacol 71:399–407
Merry BJ (1999) Dietary restriction in aging. In: Stenberg H, Timiras PS (eds) Studies of aging. Springer, New York, pp 143–163
Minarik P, Tomaskova N, Kollarova M, Antalik M (2002) Malate dehydrogenase-structure and function. Gen Physiol Biophys 21:257–265
Nisoli E, Tonello C, Cardile A, Cozzi V, Bracale R, Tedesco L, Falcone S, Valerio A, Cantoni O, Clementi E, Moncada S, Carruba MO (2005) Caloric restriction promotes mitochondrial biogenesis by inducing the expression of eNOS. Science 5746:314–317
Obled C, Arnal M (1991) Age-related changes in whole-body amino acid kinetics and protein turnover in rats. J Nutr 121:1990–1998
Pahlavani MA (2000) Caloric restriction and immunosenescence: a current perspective. Front Biosci 5:580–587
Ralphe JC, Bedell K, Segar JL, Scholz TD (2005) Correlation between myocardial malate/aspartate shuttle activity and EAAT1 protein expression in hyper- and hypothyroidism. Am J Physiol Heart Circ Physiol 288:H2521–H2526
Roth GS, Ingram DK, Black A, Lane MA (2000) Effects of reduced energy intake on the biology of aging: the primate model. Eur J Clin Nutr 54:S15–S20
Scholz TD, Stacia LK, Cynthia JT, Brian CS (1998) Ontogeny of malate-aspartate shuttle capacity and gene expression in cardiac mitochondria. Am J Physiol 274:C780–C788
Sharma R (1988) In: Timiras PS (ed) Physiological basis of aging and geriatrics. Mac Millan Inc., New York, pp 43–58
Sharma R (2004) Dietary restriction and its multifaceted effects. Curr Sci 87:1203–1210
Sharma R, Dey S, Verma R (1992) Age-specific development of malate-aspartate shuttle in the liver and kidney of mice. Biochem Int 27:1059–1066
Sharma R, Dutta D (2006) Age-dependent decrease in renal glucocorticoid receptor function is reversed by dietary restriction in mice. Ann N Y Acad Sci 1067:29–141
Sharma R, Patnaik SK (1982) Induction of rat liver cytosolic aspartate aminotransferase by hydrocortisone. Biochem Int 5:561–566
Spindler SR (2005) Rapid and reversible induction of the longevity, anticancer and genomic effects of caloric restriction. Mech Ageing Dev 126:960–966
Van Remmen H, Ward WF, Sabia RV, Richardson A (1995) In: Handbook of physiology of aging. Am Physiol Soc, Bethesda, MD, pp 171–234
Weindruch R (1996) Oxidative stress, caloric restriction and aging. Science 273:59–63
Weinert BT, Timiras PS (2003) Physiology of aging, invited review: theories of aging. J Appl Physiol 95:1706–1716
Willcox DC, Willcox BJ, Todoriki H, Curb JD, Suzuki M (2006) Calorie restriction and human longevity: what can we learn from Okinawans? Biogerontology 7:173–177
Acknowledgment
We are grateful to the North Eastern Hill University, Shillong for providing research facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goyary, D., Sharma, R. Late onset of dietary restriction reverses age-related decline of malate–aspartate shuttle enzymes in the liver and kidney of mice. Biogerontology 9, 11–18 (2008). https://doi.org/10.1007/s10522-007-9112-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-007-9112-8