Abstract
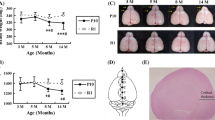
This study investigates characteristics of aging in the central nervous system of the senescence accelerated prone mice (SAMP8). We examined 3 and 10-months old senescence-accelerated-prone mice (SAMP8) for functional and molecular changes in their brains, specifically in the hippocampus and somatosensory cortex. There was no statistically significant increase in the apoptosis indicators as revealed by Western Blotting for BAD and TUNEL experiments. However, the functional magnetic resonance imaging showed an increase in the area of BOLD images from the 3-month old to the 10-months old SAMP mice upon the application of tail stimulus. These results demonstrated a lack of neuronal deaths but an increase in the activated brain area with age.






Similar content being viewed by others
References
Adams I, Jones DG (1982) Synaptic remodeling and astrocytic hypertrophy in rat cerebral cortex from early to late adulthood. Neurobiol Aging 3:179–186
Battaini F, Pascale A (2005) Protein kinase C signal transduction regulation in physiological and pathological aging. Ann N Y Acad Sci 1057:177–192
Bertoni-Freddari C, Fattoretti P, Meier-Ruge W, Ulrich J (1989) Computer-assisted morphometry of synaptic plasticity during aging and dementia. Pathol Res Pract 185:799–802
Bertoni-Freddari C, Fattoretti P, Casoli T, Pieroni M, Meier-Ruge W, Ulrich J (1991) Neurobiology of the aging brain: morphological alterations at synaptic regions. Arch Gerontol Geriatr 12:253–259
Dluzniewska J, Beresewicz M, Wojewodzka U, Gajkowska B, Zablocka B (2005) Transient cerebral ischemia induces delayed proapoptotic BAD translocation to mitochondria in CA1 sector of hippocampus. Brain Res Mol Brain Res 133:274–280
Driscoll I, Sutherland RJ (2005) The aging hippocampus: navigating between rat and human experiments. Rev Neurosci 16:87–121
Fang M, Li J, Lu G, Gong X, Yew DT (2005) A fMRI study of age-related differential cortical patterns during cued motor movement. Brain Topogr 17:127–137
Flood JF, Morley JE (1992) Early onset of age-related impairment of aversive and appetitive learning in the SAM-P/8 mouse. J Gerontol 47:B52–B59
Flood JF, Morley JE (1993) Age-related changes in footshock avoidance acquisition and retention in senescence accelerated mouse (SAM). Neurobiol Aging 14:153–157
Grigoriou V, Shapiro IM, Cavalcanti-Adam EA, Composto RJ, Ducheyne P, Adams CS (2005) Apoptosis and survival of osteoblast-like cells are regulated by surface attachment. J Biol Chem 280:1733–1739
Groh C, Ahrens D, Rosseler W (2006) Environment and age-dependent plasticity of synaptic complexes in the mushroom bodies of honeybee queens. Brain Behav Evol 68:1–14
Halpain S, Spencer K, Graber S (2005) Dynamics and pathology of dendritic spines. Prog Brain Res 147:29–37
Hof PR, Morrison JH (2004) The aging brain: morphomolecular senescence of cortical circuits. Trends Neurosci 27:607–613
Karasawa N, Nagatsu I, Sakai K, Nagatsu T, Watanabe K, Onozuka M (1997) Immunocytochemical study of catecholaminergic neurons in the senescence-accelerated mouse (SAM-P8) brain. J Neural Transm 104:1267–1275
Larner SF, McKinsey DM, Hayes RL, Wang KKW (2005) Caspase 7: increased expression and activation after traumatic brain injury in rats. J Neurochem 94:97–108
Li WP, Chan WY, Lai HW, Yew DT (1997) Terminal dUTP nick end labeling (TUNEL) positive cells in the different regions of the brain in normal aging and Alzheimer patients. J Mol Neurosci 8:75–82
Lores-Arnaiz S, Bustamante J, Arismendi M, Vilas S, Paglia N, Basso N, Capani F, Coirini H, Costa JJ, Arnaiz MR (2006) Extensive enriched environments protect old rats from the aging dependent impairment of spatial cognition, synaptic plasticity and nitric oxide production. Behav Brain Res 169:294–302
Marcilhac A (2004) Intracellular signaling pathways, apoptosis and neurodegenerative diseases. Psychol Neuropsychiatr Vieil 2:203–214
Miyamoto M (1997) Characteristics of age related behavioral changes in senescence accelerated mouse SAMP8 and SAMP10. Exp Gerontol 32:139–148
Miyamoto M, Kiyota Y, Yamazaki N, Nagaoka A, Matsuo T, Nagawa Y, Takeda T (1986) Age-related changes in learning and memory in the senescence-accelerated mouse (SAM). Physiol Behav 38:399–406
Miyamoto M, Kiyota Y, Nishiyama M, Nagaoka A (1992) Senescence-accelerated mouse (SAM): age related reduced anxiety-like behavior in the SAM-P/8 strain. Physiol Behav 51:979–985
Moolman DL, Vitolo OV, Vonsattel JP, Shelanski ML (2004) Dendrite and dendritic spine alterations in Alzheimer models. J Neurocytol 33:377–387
Morley JE, Kumar VB, Bernardo AE, Farr SA, Uezu K, Tumosa N, Flood JF (2000) Beta-amyloid precursor polypeptide in SAMP8 mice affects learning and memory. Peptides 21:1761–1767
Morley JE, Farr SA, Kumar VB, Banks WA (2002) Alzheimer’s disease through the eye of a mouse: acceptance lecture for the 2001 Gayle A. Olson and Richard D. Olson Prize Peptides 23:589–599
Ohta A, Hirano T, Yagi H, Tanaka S, Hosokawa M, Takeda T (1989) Behavioral characteristics of the SAM-P/8 strain in Sidman active avoidance task. Brain Res 498:195–198
Okuma Y, Nomura Y (1998) Senescence-accelerated mouse (SAM) as an animal model of senile dementia: pharmacological, neurochemical and molecular biological approach. Jpn J Pharmacol 78:399–404
Petrella JR, Townsend BA, Jha AP, Ziajko LA, Slavin MJ, Lustig C, Hart SJ, Doraiswamy PM (2005) Increasing memory load modulates regional brain activity in older adults as measured by fMRI. J Neuropsychiatry Clin Neurosci 17:75–83
Rombouts SA, Barkhof F, Goekoop R, Stam CJ, Scheltens P (2005) Altered resting state networks in mild cognitive impairment and mild Alzheimer’s disease: an fMRI study. Hum Brain Mapp 26:231–239
Sheng W, Wang G, Wang Y, Liang J, Wen J, Zheng PS, Wu Y, Lee V, Slingerland J, Dumont D, Yang BB (2005) The roles of versican V1 and V2 isoforms in cell proliferation and apoptosis. Mol Biol Cell 16:1330–1340
Sprott RL, Austad SN, Schneider EL, Rowe JW (1995) Handbook of the biology of aging. Academic, San Diego
Strong R, Reddy V, Morley JE (2003) Cholinergic deficits in the septal-hippocampal pathway of the SAM-P/8 senescence accelerated mouse. Brain Res 966:150–156
Sun X, Zhang X, Chen X, Zhang P, Bao M, Zhang D, Chen J, He S, Hu X (2005) Age-dependent brain activation during forward and backward digit recall revealed by fMRI. Neuroimage 26:36–47
Sureda FX, Gutierrez-Cuesta J, Romeu M, Mulero M, Canudas AM, Camins A, Mallol J, Pallas M (2006) Changes in oxidative stress parameters and neurodegeneration markers in the brain of the senescence-accelerated mice SAMP-8. Exp Gerontol 41:360–367
Takeda T, Hosokawa M, Takeshita S, Irino M, Higuchi K, Matsushita T, Tomita Y, Yasuhira K, Hamamoto H, Shimizu K, Ishii M, Yamamuro T (1981) A new murine model of accelerated senescence. Mech Ageing Dev 17(2):183–194
Takeda T, Hosokawa M, Higuchi K (1991) Senescence-accelerated mice (SAM): a novel murine model of accelerated senescence. J Am Geriatr Soc 39:911–919
Tessitore A, Hariri AR, Fera F, Smith WG, Das S, Weinberger DR, Mattay VS (2005) Functional changes in the activity of brain regions underlying emotion processing in the elderly. Psychiatry Res 139:9–18
Tonkikh A, Janus C, El-Beheiry H, Pennefather PS, Samoilova M, McDonald P, Ouanounou A, Carlen PL (2006) Calcium chelation improves spatial learning and synaptic plasticity in aged rats. Exp Neurol 197:291–300
Vantler M, Caglayan E, Zimmermann WH, Baumer AT, Rosenkranz S (2005) Systematic evaluation of anti-apoptotic growth factor signaling in vascular smooth muscle cells. Only phosphatidylinositol 3′-kinase is important. J Biol Chem 280:14168–14176
von Gunten A, Kovari E, Rivara CB, Bouras C, Hof PR, Giannakopoulos P (2005) Stereologic analysis of hippocampal Alzheimer’s disease pathology in the oldest-old: evidence for sparing of the entorhinal cortex and CA1 field. Exp Neurol 193:198–206
Wang J, Eslinger PJ, Smith MB, Yang QX (2005) Functional magnetic resonance imaging study of human olfaction and normal aging. J Gerontol A Biol Sci Med Sci 60:510–514
Wu Y, Zhang AQ, Wai MS, Lai HW, Wu SX, Yew DT (2006) Changes of apoptosis-related proteins in hippocampus of SAM mouse in development and aging. Neurobiol Aging 27:782.e1–782.e10
Yagi H, Katoh S, Akiguchi I, Takeda T (1988) Age-related deterioration of ability of acquisition in memory and learning in senescence accelerated mouse: SAM-P8 as an animal model of disturbances in recent memory. Brain Res 474:86–93
Zhang A, Lorke DE, Lai HW, Chu X, Wu Y, Yew DT (2004) Age-related alterations in cytochrome c-mediated caspase activation in rhesus macaque monkey (Macaca mulatta) brains. Brain Res Mol Brain Res 123:112–120
Zuo Y, Lin A, Chang P, Gan WB (2005) Development of long-term dendritic spine stability in diverse regions of cerebral cortex. Neuron 46:181–189
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Li, Q., Wolff, L.T. et al. Changes of brain activity in the aged SAMP mouse. Biogerontology 8, 81–88 (2007). https://doi.org/10.1007/s10522-006-9035-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-006-9035-9