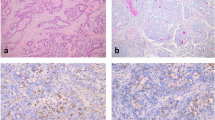
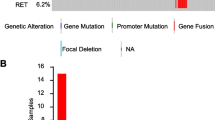
The standard for detecting chimeric genes of neurotrophic receptor tyrosine kinases (NTRK) is next generation sequencing (NGS). However, this analysis is expensive and takes several days. As a rapid screening method for the detection of NTRK3-dependent papillary thyroid cancer, an analysis of the expression imbalance between 5’ and 3’ NTRK3 mRNA fragments was used (5’/3’ RT-PCR). The reference method for detection of NTRK3 rearrangements was fluorescent in situ hybridization (FISH), and the most frequent rearrangements in papillary thyroid cancer were tested using reverse transcription PCR (RT-PCR). Using 5’/3’ RT-PCR, 18 samples of papillary thyroid cancer carrying chimeric transcripts of NTRK3 mRNA were detected. The sensitivity of the developed technique was 88.9% and specificity was 99.3%. Thus, a fast and cost-effective method of screening samples of papillary thyroid cancer in paraffin blocks is proposed with acceptable sensitivity and specificity.
Similar content being viewed by others
References
Amatu A, Sartore-Bianchi A, Siena S. NTRK gene fusions as novel targets of cancer therapy across multiple tumour types. ESMO Open. 2016;1(2):e000023. https://doi.org/10.1136/esmoopen-2015-000023
Khotskaya YB, Holla VR, Farago AF, Mills Shaw KR, Meric-Bernstam F, Hong DS. Targeting TRK family proteins in cancer. Pharmacol. Ther. 2017;173:58-66. https://doi.org/10.1016/j.pharmthera.2017.02.006
Cocco E, Scaltriti M, Drilon A. NTRK fusion-positive cancers and TRK inhibitor therapy. Nat. Rev. Clin. Oncol. 2018;15(12):731-747. https://doi.org/10.1038/s41571-018-0113-0
Doebele RC, Drilon A, Paz-Ares L, Siena S, Shaw AT, Farago AF, Blakely CM, Seto T, Cho BC, Tosi D, Besse B, Chawla SP, Bazhenova L, Krauss JC, Chae YK, Barve M, Garrido-Laguna I, Liu SV, Conkling P, John T, Fakih M, Sigal D, Loong HH, Buchschacher GL Jr, Garrido P, Nieva J, Steuer C, Overbeck TR, Bowles DW, Fox E, Riehl T, Chow-Maneval E, Simmons B, Cui N, Johnson A, Eng S, Wilson TR, Demetri GD; trial investigators. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1-2 trials. Lancet Oncol. 2020;21(2):271-282. https://doi.org/10.1016/S1470-2045(19)30691-6
Hong DS, DuBois SG, Kummar S, Farago AF, Albert CM, Rohrberg KS, van Tilburg CM, Nagasubramanian R, Berlin JD, Federman N, Mascarenhas L, Geoerger B, Dowlati A, Pappo AS, Bielack S, Doz F, McDermott R, Patel JD, Schilder RJ, Tahara M, Pfister SM, Witt O, Ladanyi M, Rudzinski ER, Nanda S, Childs BH, Laetsch TW, Hyman DM, Drilon A. Larotrectinib in patients with TRK fusion-positive solid tumours: a pooled analysis of three phase 1/2 clinical trials. Lancet Oncol. 2020;21(4):531-540. https://doi.org/10.1016/S1470-2045(19)30856-3
Connor A, Perez-Ordoñez B, Shago M, Skálová A, Weinreb I. Mammary analog secretory carcinoma of salivary gland origin with the ETV6 gene rearrangement by FISH: expanded morphologic and immunohistochemical spectrum of a recently described entity. Am. J. Surg. Pathol. 2012;36(1):27-34. https://doi.org/10.1097/PAS.0b013e318231542a
Skálová A, Vanecek T, Simpson RH, Laco J, Majewska H, Baneckova M, Steiner P, Michal M. Mammary analogue secretory carcinoma of salivary glands: molecular analysis of 25 ETV6 gene rearranged tumors with lack of detection of classical ETV6-NTRK3 fusion transcript by standard RT-PCR: report of 4 cases harboring ETV6-X gene fusion. Am. J. Surg. Pathol. 2016;40(1):3-13. https://doi.org/10.1097/PAS.0000000000000537
Zehir A, Benayed R, Shah RH, Syed A, Middha S, Kim HR, Srinivasan P, Gao J, Chakravarty D, Devlin SM, Hellmann MD, Barron DA, Schram AM, Hameed M, Dogan S, Ross DS, Hechtman JF, DeLair DF, Yao J, Mandelker DL, Cheng DT, Chandramohan R, Mohanty AS, Ptashkin RN, Jayakumaran G, Prasad M, Syed MH, Rema AB, Liu ZY, Nafa K, Borsu L, Sadowska J, Casanova J, Bacares R, Kiecka IJ, Razumova A, Son JB, Stewart L, Baldi T, Mullaney KA, Al-Ahmadie H, Vakiani E, Abeshouse AA, Penson AV, Jonsson P, Camacho N, Chang MT, Won HH, Gross BE, Kundra R, Heins ZJ, Chen HW, Phillips S, Zhang H, Wang J, Ochoa A, Wills J, Eubank M, Thomas SB, Gardos SM, Reales DN, Galle J, Durany R, Cambria R, Abida W, Cercek A, Feldman DR, Gounder MM, Hakimi AA, Harding JJ, Iyer G, Janjigian YY, Jordan EJ, Kelly CM, Lowery MA, Morris LGT, Omuro AM, Raj N, Razavi P, Shoushtari AN, Shukla N, Soumerai TE, Varghese AM, Yaeger R, Coleman J, Bochner B, Riely GJ, Saltz LB, Scher HI, Sabbatini PJ, Robson ME, Klimstra DS, Taylor BS, Baselga J, Schultz N, Hyman DM, Arcila ME, Solit DB, Ladanyi M, Berger MF. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat. Med. 2017;23(6):703-713. https://doi.org/10.1038/nm.4333
Solomon JP, Benayed R, Hechtman JF, Ladanyi M. Identifying patients with NTRK fusion cancer. Ann. Oncol. 2019;30(Suppl. 8):viii16-viii22. https://doi.org/10.1093/annonc/mdz384
Solomon JP, Hechtman JF. Detection of NTRK fusions: merits and limitations of current diagnostic platforms. Cancer Res. 2019;79(13):3163-3168. https://doi.org/10.1158/0008-5472.CAN-19-0372
Mitiushkina NV, Romanko AA, Preobrazhenskaya EV, Tiurin VI, Ermachenkova TI, Martianov AS, Mulkidjan RS, Sokolova TN, Kholmatov MM, Bizin IV, Ivantsov AO, Yatsuk OS, Zaitseva OA, Iyevleva AG, Kuligina ES, Imyanitov EN. Comprehensive evaluation of the test for 5’-/3’-end mRNA unbalanced expression as a screening tool for ALK and ROS1 fusions in lung cancer. Cancer Med. 2022;11(17):3226-3237. https://doi.org/10.1002/cam4.4686
Wang R, Pan Y, Li C, Hu H, Zhang Y, Li H, Luo X, Zhang J, Fang Z, Li Y, Shen L, Ji H, Garfield D, Sun Y, Chen H. The use of quantitative real-time reverse transcriptase PCR for 5’ and 3’ portions of ALK transcripts to detect ALK rearrangements in lung cancers. Clin. Cancer Res. 2012;18(17):4725-4732. https://doi.org/10.1158/1078-0432.CCR-12-0677
Nozaki Y, Yamamoto H, Iwasaki T, Sato M, Jiromaru R, Hongo T, Yasumatsu R, Oda Y. Clinicopathological features and immunohistochemical utility of NTRK-, ALK-, and ROS1-rearranged papillary thyroid carcinomas and anaplastic thyroid carcinomas. Hum. Pathol. 2020;106;82-92. https://doi.org/10.1016/j.humpath.2020.09.004
Ricarte-Filho JC, Li S, Garcia-Rendueles ME, Montero-Conde C, Voza F, Knauf JA, Heguy A, Viale A, Bogdanova T, Thomas GA, Mason CE, Fagin JA. Identification of kinase fusion oncogenes in post-Chernobyl radiation-induced thyroid cancers. J. Clin. Invest. 2013;123(11):4935-4944. https://doi.org/10.1172/JCI69766
Pitoia F. Complete response to larotrectinib treatment in a patient with papillary thyroid cancer harboring an ETV6-NTRK3 gene fusion. Clin. Case Rep. 2021;9(4):1905-1912. https://doi.org/10.1002/ccr3.3900
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 175, No. 2, pp. 203-209, February, 2023
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oskorbin, I.P., Ivanov, A.A., Smertina, M.A. et al. Identification of Chimeric NTRK3 Genes in Papillary Thyroid Cancer Cells by Analyzing the Imbalance of the Expression of 5’ and 3’ mRNA Fragments. Bull Exp Biol Med 175, 239–244 (2023). https://doi.org/10.1007/s10517-023-05842-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05842-x