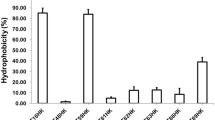
The aim of this study was the development of a methodology for the integral study of the antagonistic activity of normal human microbiota against Staphylococcus aureus to enable direct selection (without prior isolation of pure cultures) of potentially highly efficient probiotic preparations. The selection of bacterial representatives of normal human nasal microbiota capable of antagonizing S. aureus was carried out using two complimentary methods: replica-plating and deferred antagonism procedures. The material of the anterior nares from healthy human subjects was plated onto the surface of different nutrient media agar plates followed by incubation under appropriate conditions. The grown bacterial colonies were replica-plated to Petri dishes with nutrient agar overlayed with a thin layer of a soft agar which contained the culture of an indicator S. aureus strain. This agar also supported the growth of potential probiotic strains. The potential probiotic strains were selected by their ability to suppress the growth of S. aureus around their colonies. Most active strains-inhibitors may be used to develop probiotic preparations with targeted activity against S. aureus.
Similar content being viewed by others
References
Khamash DF, Voskertchian A, Milstone AM. Manipulating the microbiome: evolution of a strategy to prevent S. aureus disease in children. J. Perinatol. 2018;38(2):105-109. https://doi.org/10.1038/jp.2017.155
Kluytmans J, van Belkum A, Verbrugh H. Nasal carriage of Staphylococcus aureus: epidemiology, underlying mechanisms, and associated risks. Clin. Microbiol. Rev. 1997;10(3):505-520. https://doi.org/10.1128/CMR.10.3.505
Tan RA, Corren J. The relationship of rhinitis and asthma, sinusitis, food allergy, and eczema. Immunol. Allergy Clin. North Am. 2011;31(3):481-491. https://doi.org/10.1016/j.iac.2011.05.010
Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S, Calder PC, Sanders ME. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014;11(8):506-514. https://doi.org/10.1038/nrgastro.2014.66
Uehara Y, Nakama H, Agematsu K, Uchida M, Kawakami Y, Abdul Fattah AS, Maruchi N. Bacterial interference among nasal inhabitants: eradication of Staphylococcus aureus from nasal cavities by artificial implantation of Corynebacterium sp. J. Hosp. Infect. 2000;44(2):127-133. https://doi.org/10.1053/jhin.1999.0680
Kiryukhina NV, Melnikov VG, Suvorov AV, Morozova YA, Ilyin VK. Use of Corynebacterium pseudodiphtheriticum for elimination of Staphylococcus aureus from the nasal cavity in volunteers exposed to abnormal microclimate and altered gaseous environment. Probiotics Antimicrob. Proteins. 2013;5(4):233-238. https://doi.org/10.1007/s12602-013-9147-x
Primak TD, Ehrdyneeva BS. Patent RU No. 2341273. Method of rhinopharynx staphylococcal carrier state treatment. Bull. No. 35. Published December 20, 2008.
Shokryazdan P, Faseleh Jahromi M, Liang JB, Ho YW. Probiotics: from isolation to application. J. Am. Coll. Nutr. 2017;36(8):666-676. https://doi.org/10.1080/07315724.2017.1337529
Hjelm M, Bergh O, Riaza A, Nielsen J, Melchiorsen J, Jensen S, Duncan H, Ahrens P, Birkbeck H, Gram L. Selection and identification of autochthonous potential probiotic bacteria from turbot larvae (Scophthalmus maximus) rearing units. Syst. Appl. Microbiol. 2004;27(3):360-371. https://doi.org/10.1078/0723-2020-00256
Muriana PM, Klaenhammer TR. Conjugal transfer of plasmid-encoded determinants for bacteriocin production and immunity in Lactobacillus acidophilus 88. Appl. Environ. Microbiol. 1987;53(3):553-560. https://doi.org/10.1128/aem.53.3.553-560.1987
Pikina AP, Smeianov VV, Efimov BA, Baĭnov NA, Brook I, Reeves G, Korshunov VM. The primary screening of bifidobacteria and lactobacilli strains to develop effective probiotic preparations based on them. Zh. Mikrobiol. Epidemiol. Immunobiol. 1999;(6):34-38.
Jorgensen JH, Turnidge JD. Susceptibility test methods: dilution and disk diffusion methods. Manual of clinical microbiology. Jorgensen JH, Pfaller MA, eds. Washington, 2015. P. 1253-1273.
Biavati B, Mattarelli P. Genus I. Bifidobacterium. Bergey’s manual of systematic bacteriology. Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki K, Ludwig W, Whitman WB, eds. London; New York, 2009. P. 171-206.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 175, No. 2, pp. 192-196, February, 2023
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uzdenov, M.B., Uzdenova, A.I., Katchieva, P.K. et al. Methodology for Integral Study of Antagonistic Activity of Normal Nasal Microbiota to Select Potential Probiotics Efficient in Eradication of Staphylococcus aureus. Bull Exp Biol Med 175, 229–233 (2023). https://doi.org/10.1007/s10517-023-05840-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05840-z