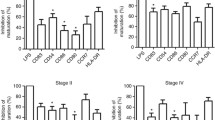
We compared the ability of SW837, SW480, HT-29, Caco-2, and HCT116 colorectal cancer lines and cancer-associated fibroblasts obtained from a colorectal adenocarcinoma biopsy specimen to modulate differentiation and maturation of dendritic cells in co-culture. The expression of surface markers of dendritic cell differentiation (CD1a) and maturation (CD83), as well as the expression of CD14 monocyte marker was evaluated by flow cytometry. Cancer-associated fibroblasts completely suppressed dendritic cell differentiation from peripheral blood monocytes induced by granulocyte-macrophage CSF and IL-4, but had no significant effect on their maturation under the influence of bacterial LPS. On the contrary, tumor cell lines did not interfere with monocyte differentiation, although some of them significantly reduced the level of CD1a expression. In contrast to cancer-associated fibroblasts, tumor cell lines and conditioned medium from primary tumor cell culture suppressed LPS-induced maturation of dendritic cells. These results suggest that tumor cells and cancer-associated fibroblasts can modulate different stages of the antitumor immune response.
Similar content being viewed by others
References
Blankenstein T, Coulie PG, Gilboa E, Jaffee EM. The determinants of tumour immunogenicity. Nat. Rev. Cancer. 2012;12(4):307-313. doi: https://doi.org/10.1038/nrc3246
Tie Y, Tang F, Wei YQ, Wei XW. Immunosuppressive cells in cancer: mechanisms and potential therapeutic targets. J. Hematol. Oncol. 2022;15(1):61. doi: https://doi.org/10.1186/s13045-022-01282-8
Lan T, Chen L, Wei X. Inflammatory cytokines in cancer: comprehensive understanding and clinical progress in gene therapy. Cells. 2021;10(1):100. doi: https://doi.org/10.3390/cells10010100
Goswami S, Anandhan S, Raychaudhuri D, Sharma P. Myeloid cell-targeted therapies for solid tumours. Nat. Rev. Immunol. 2022. Jun 13. doi: https://doi.org/10.1038/s41577-022-00737-w
SECTION 2 Adaptive Immune Responses. Immunology. Male D, Peebles S, Male V, eds. Elsevier, 2020. P. 393.
Lupatov AYu, Karalkin PA, Boyko AA, Yarygin KN. Autotransplantation of T-lymphocytes as a tool for antigen-specific immunotherapy of oncological diseases. Vestn. Transplantol. Iskusstv. Organov. 2018;20(3):95-104. Russian. doi: https://doi.org/10.15825/1995-1191-2018-3-95-104
Lau P, Shen M, Ma F, Chen Y, Zhang J, Su J, Chen X, Liu H. A Bayesian network meta-analysis of comparison of cancer therapeutic vaccines for melanoma. J. Eur. Acad. Dermatol. Venereol. 2021;35(10):1976-1986. doi: https://doi.org/10.1111/jdv.17437
Morisaki T, Morisaki T, Kubo M, Morisaki S, Nakamura Y, Onishi H. Lymph nodes as anti-tumor immunotherapeutic tools: intranodal-tumor-specific antigen-pulsed dendritic cell vaccine immunotherapy. Cancers (Basel). 2022;14(10):2438. doi: https://doi.org/10.3390/cancers14102438
Sadeghi Najafabadi SA, Bolhassani A, Aghasadeghi MR. Tumor cell-based vaccine: an effective strategy for eradication of cancer cells. Immunotherapy. 2022;14(8):639-654. doi: https://doi.org/10.2217/imt-2022-0036
Cernadas M, Lu J, Watts G, Brenner MB. CD1a expression defines an interleukin-12 producing population of human dendritic cells. Clin. Exp. Immunol. 2009;155(3):523-533. doi: https://doi.org/10.1111/j.1365-2249.2008.03853.x
Gogolak P, Rethi B, Szatmari I, Lanyi A, Dezso B, Nagy L, Rajnavolgyi E. Differentiation of CD1a— and CD1a+ monocyte-derived dendritic cells is biased by lipid environment and PPARgamma. Blood. 2007;109(2):643-652. doi: https://doi.org/10.1182/blood-2006-04-016840
Lupatov AY, Saryglar RY, Vtorushina VV, Poltavtseva RA, Bystrykh OA, Chuprynin VD, Krechetova LV, Pavlovich SV, Yarygin KN, Sukhikh GT. Mesenchymal stromal cells isolated from ectopic but not eutopic endometrium display pronounced immunomodulatory activity in vitro. Biomedicines. 2021;9(10):1286. doi: https://doi.org/10.3390/biomedicines9101286
Lupatov AY, Poltavtseva RA, Bystrykh OA, Yarygin KN, Sukhikh GT. Neural stem/progenitor cells maintained in vitro under different culture conditions alter differentiation capacity of monocytes to generate dendritic cells. J. Stem Cells Regen. Med. 2017;13(2):54-61. doi: https://doi.org/10.46582/jsrm.1302009
Berthier-Vergnes O, Gaucherand M, Péguet-Navarro J, Plouet J, Pageaux JF, Schmitt D, Staquet MJ. Human melanoma cells inhibit the earliest differentiation steps of human Langerhans cell precursors but failed to affect the functional maturation of epidermal Langerhans cells. Br. J. Cancer. 2001;85(12):1944-1951. doi: https://doi.org/10.1054/bjoc.2001.2183
Schetters STT, Rodriguez E, Kruijssen LJW, Crommentuijn MHW, Boon L, Van den Bossche J, Den Haan JMM, Van Kooyk Y. Monocyte-derived APCs are central to the response of PD1 checkpoint blockade and provide a therapeutic target for combination therapy. J. Immunother. Cancer. 2020;8(2):e000588. doi: https://doi.org/10.1136/jitc-2020-000588
Morelli AE, Thomson AW. Dendritic cells: regulators of alloimmunity and opportunities for tolerance induction. Immunol. Rev. 2003;196:125-146. doi: https://doi.org/10.1046/j.1600-065x.2003.00079.x
Ning Y, Shen K, Wu Q, Sun X, Bai Y, Xie Y, Pan J, Qi C. Tumor exosomes block dendritic cells maturation to decrease the T cell immune response. Immunol. Lett. 2018;199:36-43. doi: https://doi.org/10.1016/j.imlet.2018.05.002
Andersen MM, Larsen J, Hansen M, Pedersen AE, Gad M. Development of an In vitro assay to assess pharmacological compounds and reversion of tumor-derived immunosuppression of dendritic cells. Immunol. Invest. 2021;50(5):527-543. doi: https://doi.org/10.1080/08820139.2020.1778024
Michielsen AJ, Hogan AE, Marry J, Tosetto M, Cox F, Hyland JM, Sheahan KD, O’Donoghue DP, Mulcahy HE, Ryan EJ, O’Sullivan JN. Tumour tissue microenvironment can inhibit dendritic cell maturation in colorectal cancer. PLoS One. 2011;6(11):e27944. doi: https://doi.org/10.1371/journal.pone.0027944
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 4, pp. 222-227, December 2022
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saryglar, R.Y., Lupatov, A.Y. & Yarygin, K.N. Colorectal Cancer and Tumor Stromal Cells Have Different Effects on the Differentiation and Maturation of Dendritic Cells In Vitro. Bull Exp Biol Med 174, 533–537 (2023). https://doi.org/10.1007/s10517-023-05743-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05743-z