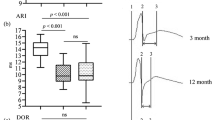
To investigate the consequences and mechanisms of myocardium remodeling of aging left atrium, we analyzed the main cardiac electrophysiological parameters such as rest membrane potential, action potential amplitude, maximum rate of action potential increase (max dV/dt), action potential plateau, and 30, 50, and 90% action potential duration (APD30, APD50, and APD90, respectively), as well as the inducibility and duration of atrial arrhythmias in adult and aging rabbits. L-type calcium current was also recorded. The collagen content in the myocardium and ultrastructure of left atrial cells were also studied. Significant changes were detected in the electrophysiological parameters and structure in aged left atrium, which can contribute to atrial susceptibility to arrhythmia in aged rabbits.
Similar content being viewed by others
References
Heijman J, Voigt N, Nattel S, Dobrev D. Cellular and molecular electrophysiology of atrial fibrillation initiation, maintenance, and progression. Circ. Res. 2014;114(9):1483-1499. https://doi.org/10.1161/CIRCRESAHA.114.302226
Yamashita A, Asada Y. Pathology and Mechanisms Underlying Thrombus Formation in Stroke. Brain Nerve. 2021;73(9):965-974
Honarbakhsh S, Schilling RJ, Orini M, Providencia R, Keating E, Finlay M, Sporton S, Chow A, Earley MJ, Lambiase PD, Hunter RJ. Structural remodeling and conduction velocity dynamics in the human left atrium: Relationship with reentrant mechanisms sustaining atrial fibrillation. Heart Rhythm. 2019;16(1):18-25. https://doi.org/10.1016/j.hrthm.2018.07.019
Corradi D, Callegari S, Maestri R, Ferrara D, Mangieri D, Alinovi R, Mozzoni P, Pinelli S, Goldoni M, Privitera YA, Bartoli V, Astorri E, Macchi E, Vaglio A, Benussi S, Alfieri O. Differential structural remodeling of the left-atrial posterior wall in patients affected by mitral regurgitation with or without persistent atrial fibrillation: a morphological and molecular study. J. Cardiovasc. Electrophysiol. 2012 Mar;23(3):271-9. https://doi.org/10.1111/j.1540-8167.2011.02187.x
Gan TY, Qiao W, Xu GJ, Zhou XH, Tang BP, Song JG, Li YD, Zhang J, Li FP, Mao T, Jiang T. Aging-associated changes in L-type calcium channels in the left atria of dogs. Exp. Ther. Med. 2013;6(4):919-924. https://doi.org/10.3892/etm.2013.1266
Mesquita TRR, Zhang R, de Couto G, Valle J, Sanchez L, Rogers RG, Holm K, Liu W, Marbán E, Cingolani E. Mechanisms of atrial fibrillation in aged rats with heart failure with preserved ejection fraction. Heart Rhythm. 2020;17(6):1025-1033. https://doi.org/10.1016/j.hrthm.2020.02.007
Qin M, Huang H, Wang T, Hu H, Liu Y, Gu Y, Cao H, Li H, Huang C. Atrial tachyarrhythmia in Rgs5-null mice. PLoS One. 2012;7(11):e46856. https://doi.org/10.1371/journal.pone.0046856
Huang Y, Liu T, Wang D, Wang X, Li R, Chen Y, Tang Y, Wang T, Huang C. Calmodulin kinase II inhibitor regulates calcium homeostasis changes caused by acute β-adrenergic receptor agonist stimulation in mouse ventricular myocytes. In Vitro Cell Dev. Biol. Anim. 2016;52(2):156-162. https://doi.org/10.1007/s11626-015-9967-y
Andrade J, Khairy P, Dobrev D, Nattel S. The clinical profile and pathophysiology of atrial fibrillation: relationships among clinical features, epidemiology, and mechanisms. Circ. Res. 2014;114(9):1453-1468. https://doi.org/10.1161/CIRCRESAHA.114.303211
Dun W, Boyden PA. Aged atria: electrical remodeling conducive to atrial fibrillation. J. Interv. Card Electrophysiol. 2009n;25(1):9-18. https://doi.org/10.1007/s10840-008-9358-3
Davies L, Jin J, Shen W, Tsui H, Shi Y, Wang Y, Zhang Y, Hao G, Wu J, Chen S, Fraser JA, Dong N, Christoffels V, Ravens U, Huang CL, Zhang H, Cartwright EJ, Wang X, Lei M. Mkk4 is a negative regulator of the transforming growth factor beta 1 signaling associated with atrial remodeling and arrhythmogenesis with age. J. Am. Heart Assoc. 2014;3(2):e000340. https://doi.org/10.1161/JAHA.113.000340
Barana A, Matamoros M, Dolz-Gaitón P, Pérez-Hernández M, Amorós I, Núñez M, Sacristán S, Pedraz Á, Pinto Á, Fernández-Avilés F, Tamargo J, Delpón E, Caballero R. Chronic atrial fibrillation increases microRNA-21 in human atrial myocytes decreasing L-type calcium current. Circ. Arrhythm. Electrophysiol. 2014;7(5):861-868. https://doi.org/10.1161/CIRCEP.114.001709
Yalcin MU, Gurses KM, Kocyigit D, Canpinar H, Canpolat U, Evranos B, Yorgun H, Sahiner ML, Kaya EB, Hazirolan T, Tokgozoglu L, Oto MA, Ozer N, Guc D, Aytemir K. The Association of Serum Galectin-3 Levels with Atrial Electrical and Structural Remodeling. J. Cardiovasc. Electrophysiol. 2015;26(6):635-640. https://doi.org/10.1111/jce.12637
Biliczki P, Boon RA, Girmatsion Z, Bukowska A, Ördög B, Kaess BM, Hohnloser SH, Goette A, Varró A, Moritz A, Nattel S, Ehrlich JR. Age-related regulation and region-specific distribution of ion channel subunits promoting atrial fibrillation in human left and right atria. Europace. 2019;21(8):1261-1269. https://doi.org/10.1093/europace/euz135
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 174, No. 8, pp. 257-264, August, 2022
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, T., Huang, Y., Wang, ZM. et al. Consequences and Mechanisms of Left Atrium Remodeling in Aging Rabbits. Bull Exp Biol Med 174, 283–289 (2022). https://doi.org/10.1007/s10517-023-05691-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05691-8