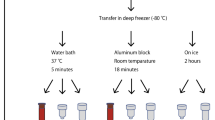
Contamination of the sample with DNA is a problem when isolating RNA by phenol-chloroform extraction, and DNase treatment introduces an additional error in the analysis of gene expression. Bearing in mind the ability of LiCl to precipitate RNA selectively, we studied the possibility and advantage of using LiCl as a precipitation agent in the protocol for RNA precipitation [3] from frozen segments of large vessels. Combined use of LiCl with isopropanol as precipitating agents (optimal ratio 2.5 M and 40%, respectively) significantly eliminates negative effects of using this salt (inhibition of reverse transcription and low RNA yield).
Similar content being viewed by others
References
Ahmad J, Baig MA, Ali AA, Al-Huqail A, Ibrahim MM, Qureshi MI. Comparative assessment of four RNA extraction methods and modification to obtain high-quality RNA from Parthenium hysterophorus leaf. 3 Biotech. 2017;7(6):373. doi: https://doi.org/10.1007/s13205-017-1003-3
Bookout AL, Cummins CL, Mangelsdorf DJ, Pesola JM, Kramer MF. High-throughput real-time quantitative reverse transcription PCR. Curr. Protoc. Mol. Biol. 2006; 73(1). doi: https://doi.org/10.1002/0471142727.mb1508s73
Chomczynski P, Sacchi N. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: twenty-something years on. Nat. Protoc. 2006;1(2):581-585. doi: https://doi.org/10.1038/nprot.2006.83
Farrell R. RNA Methodologies. A Laboratory Guide for Isolation and Characterization. London, 2010.
Ma XB, Yang J. An optimized preparation method to obtain high-quality RNA from dry sunflower seeds. Genet. Mol. Res. 2011;10(1):160-168. doi: https://doi.org/10.4238/vol10-1gmr979
Mironov KS, Shumskaya M, Los DA. Construction of prokaryotic strand-specific primary-transcripts saturated RNASeq library by controlled heat magnesium-dependent mRNA degradation. Biochimie. 2020;177:63-67. doi: https://doi.org/10.1016/j.biochi.2020.08.001
Mohammadi MR, Vadamalai G, Joseph H. An optimized method for extraction and detection of Coconut cadang-cadang viroid(CCCVd) from oil palm. Commun. Agric. Appl. Biol. Sci. 2010;75(4):777-781.
Nilsen TW. Selective precipitation of large RNAs. Cold Spring Harb. Protoc. 2012;2012(12):pdb.prot072322. doi: https://doi.org/10.1101/pdb.prot072322
Oldak B, Cruz-Rivera M, Flisser A, Mendlovic F. RNA purity, Real-Time PCR sensitivity, and colon segment influence mRNA relative expression in murine dextran sodium sulfate experimental colitis. J. Biomol. Tech. 2018;29(3):61-70. doi: https://doi.org/10.7171/jbt.18-2903-001
Pulleyblank D, Michalak M, Daisley SL, Glick R. A method for the purification of E. coli plasmid DNA by homogeneous lysis and polyethylene glycol precipitation. Mol. Biol. Rep. 1983;9(3):191-195. doi: https://doi.org/10.1007/BF00775367
Smetanina MA, Kel AE, Sevost’ianova KS, Maiborodin IV, Shevela AI, Zolotukhin IA, Stegmaier P, Filipenko ML. DNA methylation and gene expression profiling reveal MFAP5 as a regulatory driver of extracellular matrix remodeling in varicose vein disease. Epigenomics. 2018;10(8):1103-1119. doi: https://doi.org/10.2217/epi-2018-0001
Sommer SS, Cohen JE. The size distributions of proteins, mRNA, and nuclear RNA. J. Mol. Evol. 1980;15(1):37-57. doi: https://doi.org/10.1007/BF01732582
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 173, No. 3, pp. 384-389, March, 2022
Rights and permissions
About this article
Cite this article
Korolenya, V.A., Gavrilov, K.A., Sevost’ianova, K.S. et al. Evaluation of Advantages of Lithium Chloride as a Precipitating Agent in RNA Isolation from Frozen Vein Segments. Bull Exp Biol Med 173, 384–389 (2022). https://doi.org/10.1007/s10517-022-05554-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-022-05554-8