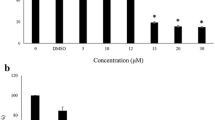
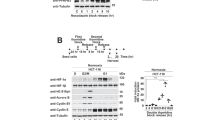
The effect of endogenous oxidative stress induced by γ-glutamyl cysteinesynthetase inhibitor D,L-buthionine sulfoximine (BSO) on the functioning of hypoxia-induced factor 1α (HIF-1α) was studied on Caco-2 cells. BSO was added for 24 h in concentrations of 5, 10, 50, 100, and 500 μM. It was shown that BSO in concentrations of 10, 50, and 100 μM induced endogenous oxidative stress and increased the content of HIF-1α; this effect was regulated through nuclear factor of erythroid origin 2 (Nrf2). Activation of HIF-1α had an independent protective effect, as evidenced by the decrease in cell viability after HIF-1α inhibition under these conditions. When the concentration of BSO was increased to 500 μM the content of HIF-1α did not change, and cell viability decreased.
Similar content being viewed by others
References
Abalenikhina YV, Sudakova EA, Seidkulieva AA, Shchulkin AV, Yakusheva EN. Functioning of pregnan X receptor under conditions of nitrosative stress. Biomed Khim. 2021;67(5):394-401. Russian. doi: https://doi.org/10.18097/PBMC20216705394
Kalinin RE, Suchkov IA, Mzhavanadze ND, Korotkova NV, Nikiforov AA, Surov IYu, Ivanova PYu, Bozhenova AD, Strelnikova EA. Comparison of cytotoxicity of vascular prostheses in vitro. 2020;28(2):183-192. Russian. doi: https://doi.org/10.23888/PAVLOVJ2020282183-192
Iakovlev AA, Guliaeva NV. Using thioGlo-1 for the determination of thiols in brain tissue. Biomed Khim. 2004 Jul-Aug;50(4):390-7. Russian.
Bollong MJ, Yun H, Sherwood L, Woods AK, Lairson LL, Schultz PG. A small molecule inhibits deregulated NRF2 transcriptional activity in cancer. ACS Chem. Biol. 2015;10(10):2193-2198. doi: https://doi.org/10.1021/acschembio.5b00448
Chan SY, Zhang YY, Hemann C, Mahoney CE, Zweier JL, Loscalzo J. MicroRNA-210 controls mitochondrial metabolism during hypoxia by repressing the iron-sulfur cluster assembly proteins ISCU1/2. Cell Metab. 2009;10(4):273-284. doi: https://doi.org/10.1016/j.cmet.2009.08.015
Huang C, Han Y, Wang Y, Sun X, Yan S, Yeh ET, Chen Y, Cang H, Li H, Shi G, Cheng J, Tang X, Yi J. SENP3 is responsible for HIF-1 transactivation under mild oxidative stress via p300 de-SUMOylation. EMBO J. 2009;28(18):2748-2762. doi: https://doi.org/10.1038/emboj.2009.210
Ke Q, Costa M. Hypoxia-inducible factor-1 (HIF-1). Mol. Pharmacol. 2006;70(5):1469-1480. doi: https://doi.org/10.1124/mol.106.027029
Moine L, Rivoira M, Díaz de Barboza G, Pérez A, Tolosa de Talamoni N. Glutathione depleting drugs, antioxidants and intestinal calcium absorption. World J Gastroenterol. 2018;24(44):4979-4988. doi: https://doi.org/10.3748/wjg.v24.i44.4979
Narita T, Yin S, Gelin CF, Moreno CS, Yepes M, Nicolaou KC, Van Meir EG. Identification of a novel small molecule HIF-1alpha translation inhibitor. Clin. Cancer Res. 2009;15(19):6128-6136. doi: https://doi.org/10.1158/1078-0432.CCR-08-3180
Schofield CJ, Ratcliffe PJ. Signalling hypoxia by HIF hydroxylases. Biochem. Biophys. Res. Commun. 2005;338(1): 617-626. doi: https://doi.org/10.1016/j.bbrc.2005.08.111
Semenza GL. HIF-1: upstream and downstream of cancer metabolism. Curr. Opin Genet. Dev. 2010;20(1):51-56. doi: https://doi.org/10.1016/j.gde.2009.10.009
Semenza GL. Hypoxia-inducible factor 1: regulator of mitochondrial metabolism and mediator of ischemic preconditioning. Biochim. Biophys. Acta. 2011;1813(7):1263-1268. doi: https://doi.org/10.1016/j.bbamcr.2010.08.006
Semenza GL. Hypoxia-inducible factors in physiology and medicine. Cell. 2012;148(3):399-408. doi: https://doi.org/10.1016/j.cell.2012.01.021
Wang J, Wu H, Zhou Y, Pang H, Liu Y, Oganezov G, Lv T, Li J, Xu J, Xiao Z, Dong X. HIF-1α inhibits mitochondria-mediated apoptosis and improves the survival of human adipose-derived stem cells in ischemic microenvironments. J. Plast. Reconstr. Aesthet. Surg. 2021;74(8):1908-1918. doi: https://doi.org/10.1016/j.bjps.2020.11.041
Wen Z, Liu W, Li X, Chen W, Liu Z, Wen J, Liu Z. A protective role of the NRF2-Keap1 pathway in maintaining intestinal barrier function. Oxid. Med. Cell. Longev. 2019;2019:1759149. doi: https://doi.org/10.1155/2019/1759149
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 173, No. 3, pp. 301-306, March, 2022
Rights and permissions
About this article
Cite this article
Abalenikhina, Y.V., Myl’nikov, P.Y., Shchul’kin, A.V. et al. Regulation and Role of Hypoxia-Induced Factor 1α (HIF-1α) under Conditions of Endogenous Oxidative Stress In Vitro. Bull Exp Biol Med 173, 312–316 (2022). https://doi.org/10.1007/s10517-022-05540-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-022-05540-0