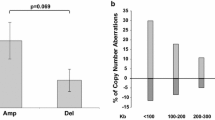
We analyzed the peculiarities of the copy number variation of genes that regulate apoptosis, DNA repair, cell proliferation, metabolism, and estrogen reception in tumor and normal cells of high-grade and low-grade serous adenocarcinoma of the ovaries. Using real-time qPCR method, the relative copy number of 34 genes (BAX, BCL2, TP53, MDM2, CASP9, CASP3, CASP7, CASP8, PRKCI, SOX2, OCT4, PIK3, PTEN, C-MYC, SOX18, AKT1, NOTCH1, BRCA1, BRCA2, EXO1, SCNN1A, KRAS, EGFR, BRAF, CYP1A1, CYP1A2, CYP1B1, CYP19A, ESR1, ESR2, GPER, STS, SULT1A, and SULT1E1) was determined in normal and tumor cells of the ovaries extracted by contactless capture laser microsection from FFPE-blocks of 200 patients. The most typical molecular markers of ovarian serous adenocarcinoma cells were identified: copy number of PIK3CA, BCL2, BAX, CASP3, and CASP8 genes. Based on the differences in the gene copy number variation, two molecular subtypes of serous adenocarcinoma were identified, corresponding to two histological subtypes: high-grade (MDM2, SOX2, ESR1, CYP1B1, SULT1E1, TP53, BRCA2) and low-grade (PIK3CA, PTEN, BCL2, BAX, and CASP3). Each of these subtypes was also characterized by molecular heterogeneity and can be subdivided into several subgroups: 3 subgroups for high-grade and 4 subgroups for low-grade serous adenocarcinoma. These findings extend our understanding of the molecular mechanisms of carcinogenesis in the ovarian tissue, confirm molecular difference between the two histological subtypes of serous adenocarcinoma probably underlying their different clinical course.
Similar content being viewed by others
References
Kolesnikov EN, Maksimov AYu, Kit OI, Kutilin DS. Dependence of overall and relapse-free patients survival from molecular genetic subtype of esophageal squamous cell cancer. Vopr. Onkol. 2019;65(5):691-700. Russian.
Kutilin DS, Airapetova TG, Anistratov PA, Pyltsin SP, Leiman IA, Karnaukhov NS, Kit OI. Copy Number Variation in Tumor Cells and Extracellular DNA in Patients with Lung Adenocarcinoma. Bull. Exp. Biol. Med. 2019;167(6):771-778. https://doi.org/10.1007/s10517-019-04620-y
Kutilin DS, Dimitriadi SN, Vodolazhsky DI, Frantsiyants HM, Kit OI. Effect of thermal ischemia-reperfusion on expression of apoptosis-regulating genes in the renal tissue of patients with renal cell carcinoma. Nefrologiya. 2017;21(1):80-86. Russian.
Tsandekova MR, Porkhanova NV, Kutilin DS. Molecular characteristic of serous ovarian adenocarcinoma: implications for diagnosis and treatment. Sovremen. Probl. Nauki Obrazovania. 2020;(1):55. Russian.
Beltrame L, Di Marino M, Fruscio R, Calura E, Chapman B, Clivio L, Sina F, Mele C, Iatropoulos P, Grassi T, Fotia V, Romualdi C, Martini P, Noris M, Paracchini L, Craparotta I, Petrillo M, Milani R, Perego P, Ravaggi A, Zambelli A, Ronchetti E, D’Incalci M, Marchini S. Profiling cancer gene mutations in longitudinal epithelial ovarian cancer biopsies by targeted next-generation sequencing: A retrospective study. Ann. Oncol. 2015;26(7):1363-1371.
Blagden SP. Harnessing pandemonium: the clinical implications of tumor heterogeneity in ovarian cancer. Front. Oncol. 2015;5:149. https://doi.org/10.3389/fonc.2015.00149
Bouska A, Eischen CM. Mdm2 affects genome stability independent of p53. Cancer Res. 2009;69(5):1697-1701.
Gajjar K, Martin-Hirsch PL, Martin FL. CYP1B1 and hormone-induced cancer. Cancer Lett. 2012;324(1):13-30.
He J, Chang W, Feng C, Cui M, Xu T. Endometriosis malignant transformation: epigenetics as a probable mechanism in ovarian tumorigenesis. Int. J. Genomics. 2018;2018:1465348. https://doi.org/10.1155/2018/1465348
Katzenellenbogen BS. Estrogen receptors: bioactivities and interactions with cell signaling pathways. Biol. Reprod. 2001;54(2):287-293.
Krishnan A, Zhang R, Yao V, Theesfeld CL, Wong AK, Tadych A, Volfovsky N, Packer A, Lash A, Troyanskaya OG. Genome-wide prediction and functional characterization of the genetic basis of autism spectrum disorder. Nat. Neurosci. 2016;19(11):1454-1462.
Kurman RJ. Origin and molecular pathogenesis of ovarian high-grade serous carcinoma. Ann. Oncol. 2013;24(Suppl. 10):x16-x21.
Kurman RJ, Shih IeM. The dualistic model of ovarian carcinogenesis: Revisited, revised, and expanded. Am. J. Pathol. 2016;186(4):733-747.
Wang JYJ. Cell death response to DNA damage. Yale J. Biol. Med. 2019;92(4):771-779.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 170, No. 9, pp. 325-333, September, 2020
Rights and permissions
About this article
Cite this article
Kutilin, D.S., Tsandekova, M.R. & Porkhanova, N.V. Features of the Copy Number Variation of Certain Genes in Tumor Cells in Patients with Serous Ovarian Adenocarcinoma. Bull Exp Biol Med 170, 332–339 (2021). https://doi.org/10.1007/s10517-021-05062-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05062-1