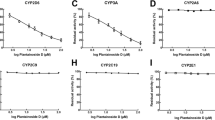
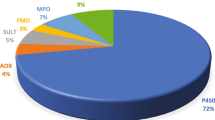
Cytochrome p450-mediated metabolism of GRS (indolinone antiaggregant) and its effects on activities of cytochrome p450 isoenzymes were studied. Inhibition of 6 isomers of cytochrome p450 in human liver microsomes was studied with the use of specific substrates. It was found that human liver cytochrome p450 enzymes could not induce degradation of GRS and that GRS was not an inductor or inhibitor of cytochrome p450 family members 1A2, 2C9, 2C19, 2D6, 2C8, and 3A4. Hence, clinical use of the prospective antiaggregant would not involve the risk of uncontrolled fluctuations in GRS concentrations in the organism because of interactions between the drugs.
Similar content being viewed by others
References
Baranov VS, Baranova EV, Ivashchenko TE, Aseev MV. Human Genome and Genes of Predispodition. Introduction into Predictive Medicine. St. Petersburg, 2000. Russian.
Basics of Toxicology. Kutsenko SA, ed. St. Petersburg, 2004. Russian.
Spitsyn VA, Makarov SV, Pai GV, Bychkovskaya LS. Polymorphism in human genes associated with biotransformation of xenobiotics. Vestn. Vavil. Obshchestva Gen. Selekts. 2006;10(1):97-105. Russian.
Sychev DA. Clinical Pharmacogenetics. Clinical Pharmacokinetics. Kukes VG, ed. Moscow, 2004. P. 154-167. Russian.
Brewer L, Williams D. Clinically Relevant Drug-Drug and Drug-Food Interactions: Underlying Mechanisms and Regulatory Requirements for Drug Licensing. Pharm. Med. 2013;27(1):9-23. https://doi.org/10.1007/s40290-013-0008-4
Curial M, Nath E, Lang E. Novel antiplatelet agent use for acute coronary syndrome in the emergency department: a review. Cardiol. Res. Pract. 2013;2013. 127270. https://doi.org/10.1155/2013/127270
Frye RF. Probing the world of cytochrome P450 enzymes. Mol. Interv. 2004;4(3):157-162.
Granik VG, Ryabova SYu, Grigoriev NB. Exogenous nitric oxide donors and inhibitors of its formation (the chemical aspects). Rus. Chem. Rev. 1997;66(8):717-731.
Guengerich FP. Cytochrome P450s and other enzymes in drug metabolism and toxicity. AAPS J. 2006;8(1):E101-E111.
Scott SA, Sangkuhl K, Stein CM, Hulot JS, Mega JL, Roden DM, Klein TE, Sabatine MS, Johnson JA, Shuldiner AR; Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium guidelines for CYP2C19 genotype and clopidogrel therapy: 2013 update. Clin. Pharmacol. Ther. 2013;94(3):317-323.
Tomaszewski P, Kubiak-Tomaszewska G, Pachecka J. Cytochrome P450 polymorphism--molecular, metabolic, and pharmacogenetic aspects. II. Participation of CYP isoenzymes in the metabolism of endogenous substances and drugs. Acta Pol. Pharm. 2008;65(3):307-318.
Williams JA, Hyland R, Jones BC, Smith DA, Hurst S, Goosen TC, Peterkin V, Koup JR, Ball SE. Drug-drug interactions for UDP-glucuronosyltransferase substrates: a pharmacokinetic explanation for typically observed low exposure (AUCi/AUC) ratios. Drug Metab. Dispos. 2004;32(11):1201-1208.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 168, No. 12, pp. 698-701, December, 2019
Rights and permissions
About this article
Cite this article
Bykov, V.V., Leonov, K.A., Serebrov, V.Y. et al. Metabolism of a New Antiaggregant, Indolinone Derivative. Bull Exp Biol Med 168, 739–742 (2020). https://doi.org/10.1007/s10517-020-04792-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-020-04792-y