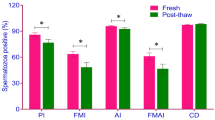
We analyzed the effect of cryopreservation on the expression of glutathione peroxidase (GPX1) and glutathione reductase (GSR) genes in human sperm cells (15 sperm samples from fertile donors and 10 samples from infertile patients). The relative expression of GPX1 and GSR genes was determined by real-time PCR. The rate of post-thaw recovery was 2.1 times higher in the group of fertile donors. A significant increase in the expression of GPX1, but not GSR, was observed in sperm samples from infertile patients, while in patients with infertility, GPX1 expression significantly decreased after cryopreservation/thawing, in samples from fertile donors after the same procedure it increased to the level observed in the sperm samples from infertile patients. A positive correlation was revealed between GPX1 expression and sperm cryotolerance.
Similar content being viewed by others
Change history
21 June 2018
The second author’s name should read A. Yu. Romanov.
References
Aitken RJ, Baker MA, Nixon B. Are sperm capacitation and apoptosis the opposite ends of a continuum driven by oxidative stress? Asian J. Androl. 2015;17(4):633-639.
Aitken RJ, Curry BJ. Redox regulation of human sperm function: from the physiological control of sperm capacitation to the etiology of infertility and DNA damage in the germ line. Antioxid. Redox Signal. 2011;14(3):367-381.
Alvarez JG, Storey BT. Role of glutathione peroxidase in protecting mammalian spermatozoa from loss of motility caused by spontaneous lipid peroxidation. Gamete Res. 1989;23(1):77-90.
Braga DP, Setti AS, Figueira RC, Iaconelli A Jr, Borges E Jr. The negative influence of sperm cryopreservation on the quality and development of the embryo depends on the morphology of the oocyte. Andrology. 2015;3(4):723-728.
Cho C, Jung-Ha H, Willis WD, Goulding EH, Stein P, Xu Z, Schultz RM, Hecht NB, Eddy EM Protamine 2 deficiency leads to sperm DNA damage and embryo death in mice. Biol. Reprod. 2003;69(1):211-217.
Cowan DB, Weisel RD, Williams WG, Mickle DA. The regulation of glutathione peroxidase gene expression by oxygen tension in cultured human cardiomyocytes. J. Mol. Cell. Cardiol. 1992;24(4):423-433.
Cowan DB, Weisel RD, Williams WG, Mickle DA. Identification of oxygen responsive elements in the 5’-flanking region of the human glutathione peroxidase gene. J. Biol. Chem. 1993;268(36):26 904-26 910.
Gandini L, Lombardo F, Lenzi A, Spanò M, Dondero F. Cryopreservation and sperm DNA integrity. Cell Tissue Bank. 2006;7(2):91-98.
Irvine DS, Twigg JP, Gordon EL, Fulton N, Milne PA, Aitken RJ. DNA integrity in human spermatozoa: relationships with semen quality. J. Androl. 2000;21(1):33-44.
Jornot L, Junod AF. Differential regulation of glutathione peroxidase by selenomethionine and hyperoxia in endothelial cells. Biochem. J. 1995;306(Pt 2):581-587.
Jornot L, Junod AF. Hyperoxia, unlike phorbol ester, induces glutathione peroxidase through a protein kinase C-independent mechanism. Biochem J. 1997;326(Pt 1):117-123.
Linfor JJ, Meyers SA. Detection of DNA damage in response to cooling injury in equine spermatozoa using single-cell gel electrophoresis. J. Androl. 2002;23(1):107-113.
Lobascio AM, De Felici M, Anibaldi M, Greco P, Minasi MG, Greco E. Involvement of seminal leukocytes, reactive oxygen species, and sperm mitochondrial membrane potential in the DNA damage of the human spermatozoa. Andrology. 2015;3(2):265-270.
McCarthy MJ, Baumber J, Kass PH, Meyers SA. Osmotic stress induces oxidative cell damage to rhesus macaque spermatozoa. Biol. Reprod. 2010;82(3):644-651.
McCarthy MJ, Meyers SA. Antioxidant treatment in the absence of exogenous lipids and proteins protects rhesus macaque sperm from cryopreservation-induced cell membrane damage. Theriogenology. 2011;76(1):168-176.
Meseguer M, Garrido N, Simón C, Pellicer A, Remohí J. Concentration of glutathione and expression of glutathione peroxidases 1 and 4 in fresh sperm provide a forecast of the outcome of cryopreservation of human spermatozoa. J. Androl. 2004;25(5):773-780.
Sridharan TB, Vickram AS. Evolving trends in cryopreservation and parameters influencing semen extender preparation — a prospective review. Cryo Lett. 2016;37(3):196-205.
Wehbi E, Meriano J, Laskin C, Jarvi KA, Lo KC. Adverse IVF/ICSI outcomes associated with higher levels of sperm dna fragmentation. J. Urol. 2009;181(4, Suppl):688.
World Health Organization, Department of Reproductive Health and Research. WHO laboratory manual for the examination and processing of human semen. WHO, 2010.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 1, pp. 58-62, January, 2018
Rights and permissions
About this article
Cite this article
Makarova, N.P., Romanov, Y.A., Dolgushina, N.V. et al. Comparative Analysis of the Expression of Glutathione Peroxidase and Glutathione Reductase Genes in Human Sperm after Cryopreservation. Bull Exp Biol Med 165, 166–170 (2018). https://doi.org/10.1007/s10517-018-4121-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-018-4121-y