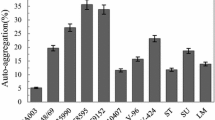
We analyzed the formation of biofilms by 7 strains of Campylobacter genus bacteria and 18 strains of Enterobacteriaceae genus bacteria that were isolated from plant and animal raw materials, from finished products, and swabs from the equipment of the food industry. Biofilm formation on glass plates, slides and coverslips, microtubes made of polymeric materials and Petri dishes, and polystyrene plates of different profiles were analyzed. When studying the process of films formation, different effects on bacterial populations were simulated, including variation of growth factor composition of culture media, technique of creating of anaerobiosis, and biocide treatment (active chlorine solutions in a concentration of 100 mg/dm3). The formation of biofilms by the studied cultures was assessed by the formation of extracellular matrix stained with aniline dyes on glass and polystyrene surfaces after incubation; 0.1% crystal violet solution was used as the dye. The presence and density of biomatrix were assessed by staining intensity of the surfaces of contact with broth cultures or by optical density of the stained inoculum on a spectrophotometer. Biofilms were formed by 57% Campylobacter strains and 44% Enterobacteriaceae strains. The intensity of the film formation depended on culturing conditions and protocols, species and genus of studied isolates, and largely on adhesion properties of abiotic surfaces. In 30% of Enterobacteriaceae strains, the biofilm formation capacity tended to increase under the influence of chlorine-containing biocide solutions. Thus, we developed and tested under laboratory conditions a plate version of in vitro chromogenic model for evaluation of biofilm formation capacity of C. jejuni strains and studied stress responses to negative environmental factors.
Similar content being viewed by others
References
Bukharin OV. From persistence to symbiosis of microorganisms. Zh. Mikrobiol., Epidemiol. Immunobiol. 2012;(4):4-9.
Efimochkina NR, Bykova IB, Korotkevich YuV, Markova YuM, Minaeva LP, Sheveleva SA. Study of tolerance of Enterobacteria to chlorine-based biocides in experimental models using chromogenic indicator tests. Analiz Riska Zdorov’yu. 2015;(3):73-82. Russian.
Litvin VYu, Gintsburg AL, Pushkareva VI, Romanova YuI, Boev BV. Epidemiological aspects of ecology of bacteria. Moscow, 1997. Russian.
Lyamin AV, Botkin EA, Zhestkov AV. Medical problems associated with bacterial biofilms. Klin. Mikrobiol. Antimikrob. Khimioter. 2012;14(4):268-275. Russian.
Romanova YuM, Alekseeva NV, Gintsburg AL. Nonculturable status of pathogenic bacteria as illustrated by Salmonella typhimurium: phenomenon and genetic control. Zh. Mikrobiol, Epidemiol. Immunobiol. 1997;(4):35-41. Russian.
Chernukha MI, Kovtun VP, Nikolaeva TN, Shaginian IA, Gintsburg AL. Development of the controlled model of persisting infection, caused by Pseudomonas aeruginosa and bacteria of the complex Burkholderia cepacia. Zh. Mikrobiol, Epidemiol. Immunobiol. 2004;(2):14-20. Russian.
Asakura H, Yamasaki M, Yamamoto S, Igimi S. Deletion of peb4 gene impairs cell adhesion and biofilm formation in Campylobacter jejuni. FEMS Microbiol. Lett. 2007;275(2):278-285.
Brown HL, Reuter M, Salt LJ, Cross KL, Betts RP, van Vliet AH. Chicken juice enhances surface attachment and biofilm formation of Campylobacter jejuni. Appl. Environ. Microbiol. 2014;80(22):7053-7060.
Gunther NW 4th, Chen CY. The biofilm forming potential of bacterial species in the genus Campylobacter. Food Microbiol. 2009;26(1):44-51.
Ica T, Caner V, Istanbullu O, Nguyen HD, Ahmed B, Call DR, Beyenal H. Characterization of mono- and mixed-culture Campylobacter jejuni biofilms. Appl. Environ. Microbiol. 2012;78(4):1033-1038.
Oliver JD. Viable but nonculturable bacteria in food. Foodborne pathogens. Microbiology and Molecular Biology. Fratamico PM, Bhunia AK, Smith JL, eds. Norfolk, 2005. P. 99-112.
Reeser RJ, Medler RT, Billington SJ, Jost BH, Joens LA. Characterization of Campylobacter jejuni biofilms under defined growth conditions. Appl. Environ. Microbiol. 2007;73(6):1908-1913.
Reuter M, Mallett A, Pearson BM, van Vliet AH. Biofilm formation by Campylobacter jejuni is increased under aerobic conditions. Appl. Environ. Microbiol. 2010;76(7):2122-2128.
Sanders SQ, Boothe DH, Frank JF, Arnold JW. Culture and detection of Campylobacter jejuni within mixed microbial populations of biofilms on stainless steel. J. Food Prot. 2007;70(6):1379-1385.
Teh AH, Lee SM, Dykes GA. Does Campylobacter jejuni form biofilms in food-related environment? Appl. Environ. Microbiol. 2014;80(17):5154-5160.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 162, No. 10, pp. 470-475, October, 2016
Rights and permissions
About this article
Cite this article
Efimochkina, N.R., Bykova, I.B., Markova, Y.M. et al. Formation of Biofilms by Foodborne Pathogens and Development of Laboratory In Vitro Model for the Study of Campylobacter Genus Bacteria Based on These Biofilms. Bull Exp Biol Med 162, 474–478 (2017). https://doi.org/10.1007/s10517-017-3643-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-017-3643-z